Abstract
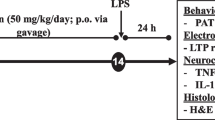
Recent studies show that morphine possesses protective preconditioning effects in different ischemia/reperfusion models. However, there is very little information about the antineuroinflammatory role of morphine and its protective effect against memory deficit. In the present study, we evaluated the role of morphine preconditioning in a model of mild neuroinflammation induced by intraperitoneal lipopolysaccharide (LPS) injection (1 mg/kg). Rats were trained on passive avoidance apparatus and challenged with LPS 20 h later. Four hours after LPS, rats were subjected to passive avoidance testing and then for the assessments of inflammatory and apoptotic cell death mediators in the hippocampus. LPS significantly increased the nuclear NF-κB and expression of COX-2, IL-1β, and TNF-α, augmented the activity of caspase-3 and PARP cleavage, and in parallel shortened the latencies to enter the dark compartment. Although morphine injection in a noninflammatory context was able to induce a neuroinflammatory response and memory loss, morphine preconditioning at the dose of 4 mg/kg significantly prevented the LPS-induced neuroinflammation and memory deficit. Morphine preconditioning was abolished by naloxone and, therefore, is dependent on opioid receptors. These results suggest that acute morphine injection, in spite of the induction of a neuroinflammatory response and amnesia per se, exerts an antineuroinflammatory role and protects from cell death and memory deficit in an inflammatory context.






Similar content being viewed by others
References
Albensi BC, Mattson MP (2000) Evidence for the involvement of TNF and NF B in hippocampal synaptic plasticity. Synapse 35:151–159
Askari N, Mahboudi F, Haeri Rohani A et al (2008) Effects of single administration of morphine on G protein mRNA level in the presence and absence of inflammation in the rat spinal cord. Scand J Immunol 67:47–52
Atici S, Cinel L, Cinel I et al (2004) Opioid neurotoxicity: comparison of morphine and tramadol in an experimental rat model. Int J Neurosci 114:1001–1011
Bao L, Zhu Y, Elhassan AM et al (2001) Adjuvant-induced arthritis: IL-1 [beta], IL-6 and TNF-[alpha] are up-regulated in the spinal cord. Neuroreport 12:3905
Barry U, Zuo Z (2005) Opioids: old drugs for potential new applications. Curr Pharm Des 11:1343–1350
Boronat MA, García-Fuster MJ, García-Sevilla JA (2001) Chronic morphine induces up–regulation of the pro–apoptotic Fas receptor and down–regulation of the anti-apoptotic Bcl-2 oncoprotein in rat brain. Br J Pharmacol 134:1263–1270
Brown GC, Bal-Price A (2003) Inflammatory neurodegeneration mediated by nitric oxide, glutamate, and mitochondria. Mol Neurobiol 27:325–355
Candelario-Jalil E, Mhadu N, González-Falcón A et al (2005) Effects of the cyclooxygenase-2 inhibitor nimesulide on cerebral infarction and neurological deficits induced by permanent middle cerebral artery occlusion in the rat. J Neuroinflammation 2:3
Chakravarty S, Herkenham M (2005) Toll-like receptor 4 on nonhematopoietic cells sustains CNS inflammation during endotoxemia, independent of systemic cytokines. J Neurosci 25:1788
Chen Q, Cui J, Zhang Y, Yu LC (2008) Prolonged morphine application modulates Bax and Hsp70 levels in primary rat neurons. Neurosci Lett 441:311–314
Comstock KL, Krown KA, Page MT et al (1998) LPS-induced TNF-[alpha] release from and apoptosis in rat cardiomyocytes: obligatory role for CD14 in mediating the LPS response. J Mol Cell Cardiol 30:2761–2775
Cunningham C, Wilcockson DC, Campion S, Lunnon K, Perry VH (2005) Central and systemic endotoxin challenges exacerbate the local inflammatory response and increase neuronal death during chronic neurodegeneration. J Neurosci 25:9275
D’Acquisto F, Iuvone T, Rombolą L, Sautebin L, Di Rosa M, Carnuccio R (1997) Involvement of NF-[kappa] B in the regulation of cyclooxygenase-2 protein expression in LPS-stimulated J774 macrophages. FEBS Lett 418:175–178
Dash PK, Orsi SA, Moore AN (2005) Sequestration of serum response factor in the hippocampus impairs long term spatial memory. J Neurochem 93:269–278
El-Hage N, Bruce-Keller AJ, Yakovleva T et al (2008) Morphine exacerbates HIV-1 Tat-induced cytokine production in astrocytes through convergent effects on [Ca2+]i, NF-κB trafficking and transcription. PLoS One 3:e4093
Frässdorf J, Weber NC, Obal D et al (2005) Morphine induces late cardioprotection in rat hearts in vivo: the involvement of opioid receptors and nuclear transcription factor B. Anesth Analg 101:934
Gidday JM, Fitzgibbons JC, Shah AR, Park T (1994) Neuroprotection from ischemic brain injury by hypoxic preconditioning in the neonatal rat. Neurosci Lett 168:221–224
Giovannini M, Scali C, Prosperi C, Bellucci A, Pepeu G, Casamenti F (2003) Experimental brain inflammation and neurodegeneration as model of Alzheimer’s disease: protective effects of selective COX-2 inhibitors. Int J Immunopathol Pharmacol 16:31
Gwak MS, Li L, Zuo Z (2010) Morphine preconditioning reduces lipopolysaccharide and interferon-[gamma]-induced mouse microglial cell injury via [delta] 1 opioid receptor activation. Neuroscience 167:256–260
Hoshino K, Tsutsui H, Kawai T et al (1999) Cutting edge: generation of IL-18 receptor-deficient mice: evidence for IL-1 receptor-related protein as an essential IL-18 binding receptor. J Immunol 162:5041
Hu S, Sheng WS, Lokensgard JR, Peterson PK (2002) Morphine induces apoptosis of human microglia and neurons. Neuropharmacology 42:829–836
Hutchinson MR, Bland ST, Johnson KW, Rice KC, Maier SF, Watkins LR (2007) Opioid-induced glial activation: mechanisms of activation and implications for opioid analgesia, dependence, and reward. Sci World 98:111
Hutchinson M, Coats B, Lewis S et al (2008) Proinflammatory cytokines oppose opioid-induced acute and chronic analgesia. Brain, Behav, Immun 22:1178–1189
Hutchinson MR, Loram LC, Zhang Y et al (2010a) Evidence that tricyclic small molecules may possess toll-like receptor and myeloid differentiation protein 2 activity. Neuroscience 168:551–563
Hutchinson MR, Zhang Y, Shridhar M et al (2010b) Evidence that opioids may have toll-like receptor 4 and MD-2 effects. Brain, Behav, Immun 24:83–95
Jänicke RU, Sprengart ML, Wati MR, Porter AG (1998) Caspase-3 is required for DNA fragmentation and morphological changes associated with apoptosis. J Biol Chem 273:9357
Joshi VD, Kalvakolanu DV, Cross AS (2003) Simultaneous activation of apoptosis and inflammation in pathogenesis of septic shock: a hypothesis1. FEBS Lett 555:180–184
Kao TK, Ou YC, Liao SL et al (2008) Opioids modulate post-ischemic progression in a rat model of stroke. Neurochem Int 52:1256–1265
Kassed C, Willing A, Garbuzova-Davis S, Sanberg P, Pennypacker K (2002) Lack of NF-[kappa] B p50 exacerbates degeneration of hippocampal neurons after chemical exposure and impairs learning. Exp Neurol 176:277–288
Kieffer BL (1995) Recent advances in molecular recognition and signal transduction of active peptides: receptors for opioid peptides. Cell Mol Neurobiol 15:615–635
Kim MS, Cheong YP, So HS et al (2001) Protective effects of morphine in peroxynitrite-induced apoptosis of primary rat neonatal astrocytes: potential involvement of G protein and phosphatidylinositol 3-kinase (PI3 kinase). Biochem Pharmacol 61:779–786
Kreutzberg GW (1996) Microglia: a sensor for pathological events in the CNS. Trends Neurosci 19:312–318
Lacroix S, Feinstein D, Rivest S (1998) The bacterial endotoxin lipopolysaccharide has the ability to target the brain in upregulating its membrane CD14 receptor within specific cellular populations. Brain Pathol 8:625–640
Laflamme N, Rivest S (2001) Toll-like receptor 4: the missing link of the cerebral innate immune response triggered by circulating gram-negative bacterial cell wall components. FASEB J 15:155
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC (1994) Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE. Nature 371:346–347
Liao S, Chen W, Raung S, Chen C (2003) Neuroprotection of naloxone against ischemic injury in rats: role of mu receptor antagonism. Neurosci Lett 345:169–172
Lim YJ, Zheng S, Zuo Z (2004) Morphine preconditions Purkinje cells against cell death under in vitro simulated ischemia–reperfusion conditions. Anesthesiology 100:562
Lin HY, Huang CC, Chang KF (2009) Lipopolysaccharide preconditioning reduces neuroinflammation against hypoxic ischemia and provides long-term outcome of neuroprotection in neonatal rat. Pediatr Res 66:254
Lin S, Tsai R, Tai Y et al (2010) Ultra-low dose naloxone upregulates interleukin-10 expression and suppresses neuroinflammation in morphine-tolerant rat spinal cords. Behav Brain Res 207:30–36
Liu SF, Malik AB (2006) NF-κB activation as a pathological mechanism of septic shock and inflammation. Am J Physiol-Lung Cell Mol Physiol 290:L622–L645
Lynn W, Golenbock D (1992) Lipopolysaccharide antagonists. Immunol Today 13:271–276
Madera-Salcedo IK, Cruz SL, Gonzalez-Espinosa C (2011) Morphine decreases early peritoneal innate immunity responses in Swiss-Webster and C57BL6/J mice through the inhibition of mast cell TNF-[alpha] release. J Neuroimmunol 232:101–107
Mao J, Sung B, Ji RR, Lim G (2002) Neuronal apoptosis associated with morphine tolerance: evidence for an opioid-induced neurotoxic mechanism. J Neurosci 22:7650
Mattson MP, Culmsee C, Yu ZF, Camandola S (2000) Roles of nuclear factor B in neuronal survival and plasticity. J Neurochem 74:443–456
McPherson BC, Yao Z (2001) Morphine mimics preconditioning via free radical signals and mitochondrial KATP channels in myocytes. Circulation 103:290–295
Merrill JE, Benveniste EN (1996) Cytokines in inflammatory brain lesions: helpful and harmful. Trends Neurosci 19:331–338
Minami M, Satoh M (1995) Molecular biology of the opioid receptors: structures, functions and distributions. Neurosci Res 23:121–145
Mucke L, Eddleston M (1993) Astrocytes in infectious and immune-mediated diseases of the central nervous system. FASEB J 7:1226
Nandagopal K, Dawson TM, Dawson VL (2001) Critical role for nitric oxide signaling in cardiac and neuronal ischemic preconditioning and tolerance. J Pharmacol Exp Ther 297:474
Nasiraei-Moghadam S, Kazeminezhad B, Dargahi L, Ahmadiani A (2010) Maternal oral consumption of morphine increases Bax/Bcl-2 ratio and caspase 3 activity during early neural system development in rat embryos. J Mol Neurosci 41:156–164
Okubo S, Tanabe Y, Takeda K et al (2004) Ischemic preconditioning and morphine attenuate myocardial apoptosis and infarction after ischemia–reperfusion in rabbits: role of delta-opioid receptor. Am J Physiol Heart Circ Physiol 287:H1786
Olin MR, Roy S, Molitor T (2010) In vivo morphine treatment synergistically increases LPS-induced caspase activity in immune organs. J Neuroimmune Pharm 5:546–552
Pak T, Cadet P, Mantione KJ, Stefano GB (2005) Morphine via nitric oxide modulates beta-amyloid metabolism: a novel protective mechanism for Alzheimer’s disease. Med Sci Monit 11:BR357–BR366
Pakpour B, Ahmadi S, Nayer-Nouri T, Oryan S, Zarrindast MR (2010) Inhibitory avoidance memory deficit induced by scopolamine: interaction with glutamatergic system in the nucleus accumbens. Behav Pharmacol 21:719
Pateliya BB, Singh N, Jaggi AS (2008) Possible role of opioids and K ATP channels in neuroprotective effect of postconditioning in mice. Biol Pharm Bull 31:1755–1760
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinate, sixth ed. Academic press, New York
Piliponsky AM, Chen CC, Grimbaldeston MA et al (2010) Mast cell-derived TNF can exacerbate mortality during severe bacterial infections in C57BL/6-KitW-sh/W-sh mice. Am J Pathol 176:926
Pourpak Z, Ahmadiani A, Alebouyeh M (2004) Involvement of interleukin 1 in systemic morphine effects on paw oedema in a mouse model of acute inflammation. Scand J Immunol 59:273–277
Pradillo JM, Romera C, Hurtado O et al (2005) TNFR1 upregulation mediates tolerance after brain ischemic preconditioning. J Cereb Blood Flow Metab 25:193–203
Qian L, Tan K, Wei S et al (2007) Microglia-mediated neurotoxicity is inhibited by morphine through an opioid receptor-independent reduction of NADPH oxidase activity. J Immunol 179:1198
Qureshi S, Larivière L, Leveque G et al (1999) Endotoxin-tolerant mice have mutations in Toll-like receptor 4 (Tlr4). The J Exp Med 189:615
Rambhia S, Mantione KJ, Stefano GB, Cadet P (2005) Morphine modulation of the ubiquitin–proteasome complex is neuroprotective. Med Sci Monit: Int Med J Exp Clin Res 11:BR386
Rehni AK, Singh TG, Jaggi AS, Singh N (2008) Pharmacological preconditioning of the brain: a possible interplay between opioid and calcitonin gene related peptide transduction systems. Pharmacol Rep 60:904–913
Roy S, Wang J, Kelschenbach J, Koodie L, Martin J (2006) Modulation of immune function by morphine: implications for susceptibility to infection. J Neuroimmune Pharmacol 1:77–89
Singhal PC, Sharma P, Kapasi AA, Reddy K, Franki N, Gibbons N (1998) Morphine enhances macrophage apoptosis. J Immunol 160:1886
Slee EA, Adrain C, Martin SJ (2001) Executioner caspase-3, -6, and -7 perform distinct, non-redundant roles during the demolition phase of apoptosis. J Biol Chem 276:7320
Spain JW, Newsom GC (1991) Chronic opioids impair acquisition of both radial maze and Y-maze choice escape. Psychopharmacology 105:101–106
Stevens SL, Ciesielski TM, Marsh BJ et al (2008) Toll-like receptor 9: a new target of ischemic preconditioning in the brain. J Cereb Blood Flow Metab 28:1040–1047
Sugimoto K, Iadecola C (2003) Delayed effect of administration of COX-2 inhibitor in mice with acute cerebral ischemia. Brain Res 960:273–276
Supajatura V, Ushio H, Nakao A, Okumura K, Ra C, Ogawa H (2001) Protective roles of mast cells against enterobacterial infection are mediated by Toll-like receptor 4. J Immunol 167:2250
Takeuchi O, Akira S (2001) Toll-like receptors; their physiological role and signal transduction system. Int Immunopharmacol 1:625–635
Turrin NP, Rivest S (2006) Molecular and cellular immune mediators of neuroprotection. Mol Neurobiol 34:221–242
Ulevitch R, Tobias P (1995) Receptor-dependent mechanisms of cell stimulation by bacterial endotoxin. Annu Rev Immunol 13:437–457
Vartanian KB, Stevens SL, Marsh BJ, Williams-Karnesky R, Lessov NS, Stenzel-Poore MP (2011) LPS preconditioning redirects TLR signaling following stroke: TRIF-IRF3 plays a seminal role in mediating tolerance to ischemic injury. J Neuroinflammation 8:140
Wang Z, Kang J, Li Y, Yuan Z, Liu S, Sun L (2006) The effects of dexamethasone on rat brain cortical nuclear factor kappa B (NF-[kappa] B) in endotoxic shock. Toxicol Appl Pharmacol 214:263–269
Wang Z, Zhao H, Peng J et al (2009) Anti-apoptotic effect of morphine postconditioning on myocardial ischemia–reperfusion injury and its anti-oxidative stress mechanism. J Sun Yat-Sen Univ (Med Sci) 5
Watkins LR, Hutchinson MR, Rice KC, Maier SF (2009) The “toll” of opioid-induced glial activation: improving the clinical efficacy of opioids by targeting glia. Trends Pharmacol Sci 30:581–591
Wright SD (1999) Toll, a new piece in the puzzle of innate immunity. The J Exp Med 189:605
Yin D, Mufson RA, Wang R, Shi Y (1999) Fas-mediated cell death promoted by opioids. Nature 397:218
Yirmiya R, Goshen I (2011) Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain, Behav, Immun 25:181–213
Zarrindast MR, Fazli-Tabaei S, Ahmadi S, Yahyavi SH (2006) Effect of lithium on morphine state-dependent memory of passive avoidance in mice. Physiol Behav 87:409–415
Zhao P, Huang Y, Zuo Z (2006) Opioid preconditioning induces opioid receptor-dependent delayed neuroprotection against ischemia in rats. J Neuropathol Exp Neurol 65:945
Zujovic V, Schussler N, Jourdain D, Duverger D, Taupin V (2001) In vivo neutralization of endogenous brain fractalkine increases hippocampal TNF [alpha] and 8-isoprostane production induced by intracerebroventricular injection of LPS. J Neuroimmunol 115:135–143
Acknowledgments
The authors are grateful to the Neuroscience Research Center of Shahid Beheshti University of Medical Sciences for the financial support of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rostami, F., Oryan, S., Ahmadiani, A. et al. Morphine Preconditioning Protects Against LPS-Induced Neuroinflammation and Memory Deficit. J Mol Neurosci 48, 22–34 (2012). https://doi.org/10.1007/s12031-012-9726-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9726-4