Abstract
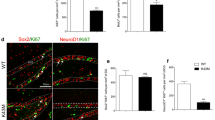
Although stem cells can proliferate and differentiate through the completion of cell cycle progression, little is known about the genes and molecular mechanisms controlling this process. Here, we investigated the effect of the inhibition of cell cycle by cyclin D1 gene knockout on proliferation and differentiation of neural stem cells (NSCs). Knockout of cyclin D1 induced the cultured neural stem cells arrested at the G0/G1 phase as detected by flow cytometry. Cyclin D1 knockout led to the apoptosis of NSCs and inhibited the differentiation into astrocytes without affecting the differentiation into neurons. We further demonstrated that a significant reduction of BrdU+ cells in the subgranular zone of the dentate gyrus and subventricular zone was found in cyclin D1 gene knockout (cyclin D1−/−) mice compared with cyclin D1+/+ and cyclin D1+/− mice. These observations demonstrated that cyclin D1 plays essential roles in the proliferation and differentiation of neural stem cells.






Similar content being viewed by others
Abbreviations
- BrdU:
-
5-Bromo-2′-deoxyuridine
- TuJI:
-
β-Tubulin III
- NSC:
-
Neural stem cell
- PI:
-
Propidium iodide
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP nick-end labeling
- SVZ:
-
The subventricular zone
- DG:
-
The subgranular zone of the dentate gyrus
References
Baldin V, Lukas J et al (1993) Cyclin D1 is a nuclear protein required for cell cycle progression in G1. Genes Dev 7(5):812–21
Bates S, Bonetta L et al (1994) CDK6 (PLSTIRE) and CDK4 (PSK-J3) are a distinct subset of the cyclin-dependent kinases that associate with cyclin D1. Oncogene 9(1):71–9
Bordey A (2006) Adult neurogenesis, basic concepts of signaling. Cell Cycle 5(7):722–8
Chen Y, Dong C (2009) Abeta40 promotes neuronal cell fate in neural progenitor cells. Cell Death Differ 16(3):386–94
Dyer MA, Cepko CL (2000) p57(Kip2) regulates progenitor cell proliferation and amacrine interneuron development in the mouse retina. Development 127(16):3593–3605
Fantl V, Stamp G et al (1995) Mice lacking cyclin D1 are small and show defects in eye and mammary gland development. Genes Dev 9(19):2364–2372
Ferguson KL, Slack RS (2001) The Rb pathway in neurogenesis. Neuroreport 12(9):A55–A62
Gavrieli Y, Sherman Y et al (1992) Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119(3):493–501
Hayes TE, Valtz NL et al (1991) Downregulation of CDC2 upon terminal differentiation of neurons. New Biol 3(3):259–269
He F, Ge W et al (2005) A positive autoregulatory loop of Jak-STAT signaling controls the onset of astrogliogenesis. Nat Neurosci 8(5):616–625
Kohn KW (1999) Molecular interaction map of the mammalian cell cycle control and DNA repair systems. Mol Biol Cell 10(8):2703–2734
Kuwabara T, Hsieh J et al (2004) A small modulatory dsRNA specifies the fate of adult neural stem cells. Cell 116(6):779–793
Lukas J, Pagano M et al (1994) Cyclin D1 protein oscillates and is essential for cell cycle progression in human tumour cell lines. Oncogene 9(3):707–718
Matsushime H, Quelle DE et al (1994) D-type cyclin-dependent kinase activity in mammalian cells. Mol Cell Biol 14(3):2066–2076
Meijer L, Borgne A et al (1997) Biochemical and cellular effects of roscovitine, a potent and selective inhibitor of the cyclin-dependent kinases cdc2, cdk2 and cdk5. Eur J Biochem 243:527–536
Ohnuma S, Philpott A et al (1999) p27Xic1, a Cdk inhibitor, promotes the determination of glial cells in Xenopus retina. Cell 99(5):499–510
Okano HJ, Pfaff DW et al (1993) RB and Cdc2 expression in brain: correlations with 3H-thymidine incorporation and neurogenesis. J Neurosci 13(7):2930–2938
Pines J (2006) Mitosis: a matter of getting rid of the right protein at the right time. Trends Cell Biol 16(1):55–63
Platel JC, Lacar B et al (2007) GABA and glutamate signaling: homeostatic control of adult forebrain neurogenesis. J Mol Hist 38:303–311
Sicinski P, Donaher JL et al (1995) Cyclin D1 provides a link between development and oncogenesis in the retina and breast. Cell 82(4):621–630
Sumrejkanchanakij P, Tamamori-Adachi M et al (2003) Role of cyclin D1 cytoplasmic sequestration in the survival of postmitotic neurons. Oncogene 22(54):8723–8730
Swanton C (2004) Cell-cycle targeted therapies. Lancet Oncol 5(1):27–36
Tamaru T, Okada M et al (1994) Differential expression of D type cyclins during neuronal maturation. Neurosci Lett 168(1–2):229–232
van Grunsven LA, Thomas A et al (1996) Nerve growth factor-induced accumulation of PC12 cells expressing cyclin D1: evidence for a G1 phase block. Oncogene 12(4):855–862
Wang W, Bu B et al (2009) Neural cell cycle dysregulation and central nervous system diseases. Prog Neurobiol 89:1–17
Xiong Y, Zhang H et al (1992) D type cyclins associate with multiple protein kinases and the DNA replication and repair factor PCNA. Cell 71(3):505–514
Xiong W, Pestell R et al (1997a) Role of cyclins in neuronal differentiation of immortalized hippocampal cells. Mol Cell Biol 17(11):6585–6597
Xiong W, Pestell RG et al (1997b) Cyclin D1 is required for S phase traversal in bovine tracheal myocytes. Am J Physiol 272(6 Pt 1):L1205–L1210
Yan GZ, Ziff EB (1995) NGF regulates the PC12 cell cycle machinery through specific inhibition of the Cdk kinases and induction of cyclin D1. J Neurosci 15(9):6200–6212
Yang Y, Mufson EJ et al (2003) Neuronal cell death is preceded by cell cycle events at all stages of Alzheimer's disease. J Neurosci 23(7):2557–2563
Zezula J, Casaccia-Bonnefil P et al (2001) p21cip1 is required for the differentiation of oligodendrocytes independently of cell cycle withdrawal. EMBO Rep 2(1):27–34
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (nos. 30725019, 30971007, and 30800341).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ma J and Yu Z contribute equally to this work.
Rights and permissions
About this article
Cite this article
Ma, J., Yu, Z., Qu, W. et al. Proliferation and Differentiation of Neural Stem Cells Are Selectively Regulated by Knockout of Cyclin D1. J Mol Neurosci 42, 35–43 (2010). https://doi.org/10.1007/s12031-010-9362-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-010-9362-9