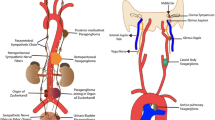
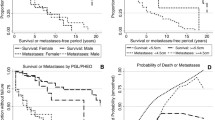
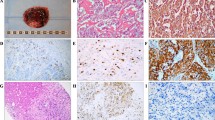
Abstract
The laboratory workup of patients with pheochromocytoma and extra-adrenal paraganglioma (PPGLs) has traditionally focused on biochemical measurements of tumor secretory products or their metabolites, with ultimate diagnosis resting on routine histopathology and immunohistochemistry. While such testing remains important, the needs to distinguish potentially metastatic from benign tumors and to identify tumors with a hereditary basis have stimulated searches for additional means to stratify patients according to risk of metastasis or presence of a particular mutation. Biomarkers based on traditional biochemical tests, such as profiles of catecholamine metabolites and granin-derived peptides, provide utility for both purposes, while novel biomarkers are being identified by proteomic and transcriptomic studies, the latter including microRNA expression profiling. Histopathological scoring methods for predicting metastatic potential, such as the Pheochromocytoma of the Adrenal Gland Scaled Score (PASS), are limited by poor interobserver concordance, discrepant results between studies and incomplete knowledge of how scores relate to genotype. Immunohistochemical staining for succinate dehydrogenase (SDH) subunit B to triage patients for genetic testing of SDH subunit genes illustrates the growing importance of pathology as an adjunct to genetic testing for disease stratification. Although considerable effort has been expended on microarray-based platforms to identify biomarkers of malignancy, as yet, none of those proposed have been demonstrated to reliably discriminate malignant from benign disease any better than the PASS. Because of the heterogeneity of PPGLs and variable time between first appearance of tumors and identification of metastases, any prospective study to establish prognostic efficacy requires large numbers of patients and extended follow-up.



Similar content being viewed by others
References
Eisenhofer G, Goldstein DS, Kopin IJ, Crout JR: Pheochromocytoma: rediscovery as a catecholamine-metabolizing tumor. Endocr Pathol 14:193–212, 2003
Eisenhofer G, Kopin IJ, Goldstein DS: Catecholamine metabolism: a contemporary view with implications for physiology and medicine. Pharmacol Rev 56:331–49, 2004
Eisenhofer G, Lenders JW, Linehan WM, Walther MM, Goldstein DS, Keiser HR: Plasma normetanephrine and metanephrine for detecting pheochromocytoma in von Hippel–Lindau disease and multiple endocrine neoplasia type 2. N Engl J Med 340:1872–9, 1999
Lenders JW, Pacak K, Walther MM, et al.: Biochemical diagnosis of pheochromocytoma: which test is best? Jama 287:1427–34, 2002
Pacak K, Eisenhofer G, Ahlman H, et al.: Pheochromocytoma: recommendations for clinical practice from the First International Symposium. Nat Clin Pract Endocrinol Metab 3:92–102, 2007
Raber W, Raffesberg W, Bischof M, et al.: Diagnostic efficacy of unconjugated plasma metanephrines for the detection of pheochromocytoma. Arch Intern Med 160:2957–63, 2000
Sawka AM, Jaeschke R, Singh RJ, Young WF, Jr.: A comparison of biochemical tests for pheochromocytoma: Measurement of fractionated plasma metanephrines compared with the combination of 24-hour urinary metanephrines and catecholamines. J Clin Endocrinol Metab 88:553–8, 2003
Unger N, Pitt C, Schmidt IL, et al.: Diagnostic value of various biochemical parameters for the diagnosis of pheochromocytoma in patients with adrenal mass. Eur J Endocrinol 154:409–17, 2006
Vaclavik J, Stejskal D, Lacnak B, et al.: Free plasma metanephrines as a screening test for pheochromocytoma in low-risk patients. J Hypertens 25:1427–31, 2007
Hickman PE, Leong M, Chang J, Wilson SR, McWhinney B: Plasma free metanephrines are superior to urine and plasma catecholamines and urine catecholamine metabolites for the investigation of phaeochromocytoma. Pathology 41:173–7, 2009
Gao YC, Lu HK, Luo QY, Chen LB, Ding Y, Zhu RS: Comparison of free plasma metanephrines enzyme immunoassay with (131)I-MIBG scan in diagnosis of pheochromocytoma. Clin Exp Med 8:87–91, 2008
Sarathi V, Pandit R, Jagtap V, et al.: Performance of fractionated plasma free metanephrines by enzyme immunoassay in the diagnosis of pheochromocytoma and paraganglioma. Endocr Pract 17:759–65, 2011
Marney LC, Laha TJ, Baird GS, Rainey PM, Hoofnagle AN: Isopropanol protein precipitation for the analysis of plasma free metanephrines by liquid chromatography-tandem mass spectrometry. Clin Chem 54:1729–32, 2008
Whiting MJ: Simultaneous measurement of urinary metanephrines and catecholamines by liquid chromatography with tandem mass spectrometric detection. Ann Clin Biochem 46:129–36, 2009
Peaston RT, Graham KS, Chambers E, van der Molen JC, Ball S: Performance of plasma free metanephrines measured by liquid chromatography-tandem mass spectrometry in the diagnosis of pheochromocytoma. Clin Chim Acta 411:546–52, 2010
Marrington R, Johnston J, Knowles S, Webster C: Measurement of urinary metadrenaline and normetadrenaline by liquid chromatography tandem mass spectrometry for the diagnosis of phaeochromocytoma. Ann Clin Biochem 47:467–75, 2010
Gabler J, Miller A, Wang S: A simple liquid chromatography-tandem mass spectrometry method for measuring metanephrine and normetanephrine in urine. Clin Chem Lab Med 49:1213–6, 2011
He X, Gabler J, Yuan C, Wang S, Shi Y, Kozak M: Quantitative measurement of plasma free metanephrines by ion-pairing solid phase extraction and liquid chromatography-tandem mass spectrometry with porous graphitic carbon column. J Chromatogr B Analyt Technol Biomed Life Sci 879:2355–9, 2011
Pillai D, Ross HA, Kratzsch J, et al.: Proficiency test of plasma free and total metanephrines: report from a study group. Clin Chem Lab Med 47:786–90, 2009
Pillai D, Callen S: Pilot quality assurance programme for plasma metanephrines. Ann Clin Biochem 47:137–42, 2010
Mullins F, O’Shea P, Fitzgerald R, Tormey W: Enzyme-linked immunoassay for plasma-free metanephrines in the biochemical diagnosis of phaeochromocytoma in adults is not ideal. Clin Chem Lab Med 2011 (in press)
Boyle JG, Davidson DF, Perry CG, Connell JM: Comparison of diagnostic accuracy of urinary free metanephrines VMA, and catecholamines and plasma catecholamines for diagnosis of pheochromocytoma. J Clin Endocrinol Metab 92:4602–8, 2007
Douwes Dekker PB, Corver WE, Hogendoorn PC, van der Mey AG, Cornelisse CJ: Multiparameter DNA flow-sorting demonstrates diploidy and SDHD wild-type gene retention in the sustentacular cell compartment of head and neck paragangliomas: chief cells are the only neoplastic component. J Pathol 202:456–62, 2004
Tischler AS: Pheochromocytoma: time to stamp out “malignancy”? Endocr Pathol 19:207–8, 2008
Linnoila RI, Keiser HR, Steinberg SM, Lack EE: Histopathology of benign versus malignant sympathoadrenal paragangliomas: clinicopathologic study of 120 cases including unusual histologic features. Hum Pathol 21:1168–80, 1990
Kimura N, Watanabe T, Noshiro T, Shizawa S, Miura Y: Histological grading of adrenal and extra-adrenal pheochromocytomas and relationship to prognosis: a clinicopathological analysis of 116 adrenal pheochromocytomas and 30 extra-adrenal sympathetic paragangliomas including 38 malignant tumors. Endocr Pathol 16:23–32, 2005
Thompson LD: Pheochromocytoma of the Adrenal gland Scaled Score (PASS) to separate benign from malignant neoplasms: a clinicopathologic and immunophenotypic study of 100 cases. Am J Surg Pathol 26:551–66, 2002
Gao B, Meng F, Bian W, et al.: Development and validation of pheochromocytoma of the adrenal gland scaled score for predicting malignant pheochromocytomas. Urology 68:282–6, 2006
Strong VE, Kennedy T, Al-Ahmadie H, et al.: Prognostic indicators of malignancy in adrenal pheochromocytomas: clinical, histopathologic, and cell cycle/apoptosis gene expression analysis. Surgery 143:759–68, 2008
Wu D, Tischler AS, Lloyd RV, et al.: Observer variation in the application of the Pheochromocytoma of the Adrenal Gland Scaled Score. Am J Surg Pathol 33:599–608, 2009
Agarwal A, Mehrotra PK, Jain M, et al.: Size of the tumor and pheochromocytoma of the adrenal gland scaled score (PASS): can they predict malignancy? World J Surg 34:3022–8, 2010
Favier J, Plouin PF, Corvol P, Gasc JM: Angiogenesis and vascular architecture in pheochromocytomas: distinctive traits in malignant tumors. Am J Pathol 161:1235–46, 2002
Eisenhofer G, Lenders JW, Siegert G, et al.: Plasma methoxytyramine: a novel biomarker of metastatic pheochromocytoma and paraganglioma in relation to established risk factors of tumor size, location and SDHB mutation status. Eur J Cancer 2011 (in press)
Remine W, Chong G, van Heerden J, Sheps S, Harrison EJ: Current management of pheochromocytoma. Ann Surg 179:740–8, 1974
Shen WT, Sturgeon C, Clark OH, Duh QY, Kebebew E: Should pheochromocytoma size influence surgical approach? A comparison of 90 malignant and 60 benign pheochromocytomas. Surgery 136:1129–37, 2004
van der Harst E, Bruining HA, Jaap Bonjer H, et al.: Proliferative index in phaeochromocytomas: does it predict the occurrence of metastases? J Pathol 191:175–80, 2000
Ayala-Ramirez M, Feng L, Johnson MM, et al.: Clinical risk factors for malignancy and overall survival in patients with pheochromocytomas and sympathetic paragangliomas: primary tumor size and primary tumor location as prognostic indicators. J Clin Endocrinol Metab 96:717–25, 2011
Feng F, Zhu Y, Wang X, et al.: Predictive factors for malignant pheochromocytoma: analysis of 136 patients. J Urol 185:1583–90, 2011
John H, Ziegler WH, Hauri D, Jaeger P: Pheochromocytomas: can malignant potential be predicted? Urology 53:679–83, 1999
Park J, Song C, Park M, et al.: Predictive characteristics of malignant pheochromocytoma. Korean J Urol 52:241–6, 2011
Clarke MR, Weyant RJ, Watson CG, Carty SE: Prognostic markers in pheochromocytoma. Hum Pathol 29:522–6, 1998
Medeiros LJ, Wolf BC, Balogh K, Federman M: Adrenal pheochromocytoma: a clinicopathologic review of 60 cases. Hum Pathol 16:580–9, 1985
Plouin PF, Chatellier G, Fofol I, Corvol P: Tumor recurrence and hypertension persistence after successful pheochromocytoma operation. Hypertension 29:1133–9, 1997
Proye C, Vix M, Goropoulos A, Kerlo P, Lecomte-Houcke M: High incidence of malignant pheochromocytoma in a surgical unit. 26 cases out of 100 patients operated from 1971 to 1991. J Endocrinol Invest 15:651–63, 1992
Goldstein RE, O’Neill JA, Jr., Holcomb GW, 3rd, et al.: Clinical experience over 48 years with pheochromocytoma. Ann Surg 229:755–64; discussion 64–6, 1999
Mannelli M, Ianni L, Cilotti A, Conti A: Pheochromocytoma in Italy: a multicentric retrospective study. Eur J Endocrinol 141:619–24, 1999
Edstrom Elder E, Hjelm Skog AL, Hoog A, Hamberger B: The management of benign and malignant pheochromocytoma and abdominal paraganglioma. Eur J Surg Oncol 29:278–83, 2003
Amar L, Servais A, Gimenez-Roqueplo AP, Zinzindohoue F, Chatellier G, Plouin PF: Year of diagnosis, features at presentation, and risk of recurrence in patients with pheochromocytoma or secreting paraganglioma. J Clin Endocrinol Metab 90:2110–6, 2005
Chapman DB, Lippert D, Geer CP, et al.: Clinical, histopathologic, and radiographic indicators of malignancy in head and neck paragangliomas. Otolaryngol Head Neck Surg 143:531–7, 2010
Ghayee HK, Havekes B, Corssmit EP, et al.: Mediastinal paragangliomas: association with mutations in the succinate dehydrogenase genes and aggressive behavior. Endocr Relat Cancer 16:291–9, 2009
Amar L, Bertherat J, Baudin E, et al.: Genetic testing in pheochromocytoma or functional paraganglioma. J Clin Oncol 23:8812–8, 2005
Brouwers FM, Eisenhofer G, Tao JJ, et al.: High frequency of SDHB germline mutations in patients with malignant catecholamine-producing paragangliomas: implications for genetic testing. J Clin Endocrinol Metab 91:4505–9, 2006
Ricketts CJ, Forman JR, Rattenberry E, et al.: Tumor risks and genotype-phenotype-proteotype analysis in 358 patients with germline mutations in SDHB and SDHD. Hum Mutat 31:41–51, 2010
King KS, Prodanov T, Kantorovich V, et al.: Metastatic Pheochromocytoma/paraganglioma related to primary tumor development in childhood or adolescence: significant link to SDHB mutations. J Clin Oncol 29:4137–42, 2011
Eisenhofer G, Huynh TT, Pacak K, et al.: Distinct gene expression profiles in norepinephrine- and epinephrine-producing hereditary and sporadic pheochromocytomas: activation of hypoxia-driven angiogenic pathways in von Hippel–Lindau syndrome. Endocrine-Related Cancer 11:897–911, 2004
Dahia PL, Ross KN, Wright ME, et al.: A HIF1alpha regulatory loop links hypoxia and mitochondrial signals in pheochromocytomas. PLoS Genet 1:72–80, 2005
Favier J, Briere JJ, Burnichon N, et al.: The Warburg effect is genetically determined in inherited pheochromocytomas. PLoS One 4:e7094, 2009
Lopez-Jimenez E, Gomez-Lopez G, Leandro-Garcia LJ, et al.: Research resource: Transcriptional profiling reveals different pseudohypoxic signatures in SDHB and VHL-related pheochromocytomas. Mol Endocrinol 24:2382–91, 2010
Burnichon N, Vescovo L, Amar L, et al.: Integrative genomic analysis reveals somatic mutations in pheochromocytoma and paraganglioma. Hum Mol Genet 20:3974–3985, 2011
Eisenhofer G, Lenders JW, Timmers H, et al.: Measurements of plasma methoxytyramine, normetanephrine, and metanephrine as discriminators of different hereditary forms of pheochromocytoma. Clin Chem 57:411–20, 2011
Eisenhofer G, Pacak K, Huynh TT, et al.: Catecholamine metabolomic and secretory phenotypes in phaeochromocytoma. Endocr Relat Cancer 18:97–111, 2011
Eisenhofer G, Timmers H, Lenders JW, et al.: Age at diagnosis of pheochromocytoma differs according to catecholamine phenotype and tumor location. Journal of Clinical Endocrinology and Metabolism 96:375–84, 2011
Robinson R, Smith P, Whittaker SR: Secretion of catecholamines in malignant phaeochromocytoma. Br Med J 1:1422–4, 1964
Tippett PA, McEwan AJ, Ackery DM: A re-evaluation of dopamine excretion in phaeochromocytoma. Clin Endocrinol (Oxf) 25:401–10, 1986
Rao F, Keiser HR, O’Connor DT: Malignant pheochromocytoma. Chromaffin granule transmitters and response to treatment. Hypertension 36:1045–52, 2000
Januszewicz W, Wocial B, Januszewicz A, Gryglas P, Prejbisz A: Dopamine and dopa urinary excretion in patients with pheochromocytoma—diagnostic implications. Blood Press 10:212–6, 2001
van der Harst E, de Herder WW, de Krijger RR, et al.: The value of plasma markers for the clinical behaviour of phaeochromocytomas. Eur J Endocrinol 147:85–94, 2002
Yon L, Guillemot J, Montero-Hadjadje M, et al.: Identification of the secretogranin II-derived peptide EM66 in pheochromocytomas as a potential marker for discriminating benign versus malignant tumors. J Clin Endocrinol Metab 88:2579–85, 2003
Guerin M, Guillemot J, Thouennon E, et al.: Granins and their derived peptides in normal and tumoral chromaffin tissue: implications for the diagnosis and prognosis of pheochromocytoma. Regul Pept 165:21–9, 2010
Szalat A, Fraenkel M, Doviner V, Salmon A, Gross DJ: Malignant pheochromocytoma: predictive factors of malignancy and clinical course in 16 patients at a single tertiary medical center. Endocrine 39:160–6, 2011
Timmers HJ, Pacak K, Huynh TT, et al.: Biochemically silent abdominal paragangliomas in patients with mutations in the Sdhb Gene. J Clin Endocrinol Metab 93:4826–4832, 2008
Eisenhofer G, Lenders JW, Goldstein DS, et al.: Pheochromocytoma catecholamine phenotypes and prediction of tumor size and location by use of plasma free metanephrines. Clin Chem 51:735–44, 2005
van Nederveen FH, Gaal J, Favier J, et al.: An immunohistochemical procedure to detect patients with paraganglioma and phaeochromocytoma with germline SDHB, SDHC, or SDHD gene mutations: a retrospective and prospective analysis. Lancet Oncol 10:764–71, 2009
Korpershoek E, Favier J, Gaal J, et al.: SDHA immunohistochemistry detects germline SDHA gene mutations in apparently sporadic paragangliomas and pheochromocytomas. J Clin Endocrinol Metab 96:E1472–6, 2011
Gill AJ, Benn DE, Chou A, et al.: Immunohistochemistry for SDHB triages genetic testing of SDHB, SDHC, and SDHD in paraganglioma–pheochromocytoma syndromes. Hum Pathol 41:805–14, 2010
Gill AJ, Chou A, Vilain R, et al.: Immunohistochemistry for SDHB divides gastrointestinal stromal tumors (GISTs) into 2 distinct types. Am J Surg Pathol 34:636–44, 2011
Gaal J, Stratakis CA, Carney JA, et al.: SDHB immunohistochemistry: a useful tool in the diagnosis of Carney–Stratakis and Carney triad gastrointestinal stromal tumors. Mod Pathol 24:147–51, 2011
Janeway KA, Kim SY, Lodish M, et al.: Defects in succinate dehydrogenase in gastrointestinal stromal tumors lacking KIT and PDGFRA mutations. Proc Natl Acad Sci U S A 108:314–8, 2011
Miettinen M, Wang ZF, Sarlomo-Rikala M, Osuch C, Rutkowski P, Lasota J: Succinate dehydrogenase-deficient GISTs: a clinicopathologic, immunohistochemical, and molecular genetic study of 66 gastric GISTs with predilection to young age. Am J Surg Pathol 35:1712–21, 2011
Gill AJ, Chou A, Vilain RE, Clifton-Bligh RJ: “Pediatric-type” gastrointestinal stromal tumors are SDHB negative (“type 2”) GISTs. Am J Surg Pathol 35:1245–7; author reply 7–8, 2011
Gill AJ, Pachter NS, Clarkson A, et al.: Renal tumors and hereditary pheochromocytoma-paraganglioma syndrome type 4. N Engl J Med 364:885–6, 2011
Gill AJ, Pachter NS, Chou A, et al.: Renal tumors associated with germline SDHB mutation show distinctive morphology. Am J Surg Pathol 35:1578–85, 2011
Sandgren J, Andersson R, Rada-Iglesias A, et al.: Integrative epigenomic and genomic analysis of malignant pheochromocytoma. Exp Mol Med 42:484–502, 2010
Brouwers FM, Petricoin EF, 3rd, Ksinantova L, et al.: Low molecular weight proteomic information distinguishes metastatic from benign pheochromocytoma. Endocr Relat Cancer 12:263–72, 2005
Brouwers FM, Elkahloun AG, Munson PJ, et al.: Gene expression profiling of benign and malignant pheochromocytoma. Ann N Y Acad Sci 1073:541–56, 2006
Suh I, Shibru D, Eisenhofer G, et al.: Candidate genes associated with malignant pheochromocytomas by genome-wide expression profiling. Ann Surg 250:983–90, 2009
Bjorklund P, Cupisti K, Fryknas M, et al.: Stathmin as a marker for malignancy in pheochromocytomas. Exp Clin Endocrinol Diabetes 118:27–30, 2010
Thouennon E, Pierre A, Tanguy Y, et al.: Expression of trophic amidated peptides and their receptors in benign and malignant pheochromocytomas: high expression of adrenomedullin RDC1 receptor and implication in tumoral cell survival. Endocr Relat Cancer 17:637–51, 2010
Waldmann J, Fendrich V, Holler J, et al.: Microarray analysis reveals differential expression of benign and malignant pheochromocytoma. Endocr Relat Cancer 17:743–56, 2010
Thouennon E, Elkahloun AG, Guillemot J, et al.: Identification of potential gene markers and insights into the pathophysiology of pheochromocytoma malignancy. J Clin Endocrinol Metab 92:4865–72, 2007
Eisenhofer G, Bornstein SR, Brouwers FM, et al.: Malignant pheochromocytoma: current status and initiatives for future progress. Endocr Relat Cancer 11:423–36, 2004
Chrisoulidou A, Kaltsas G, Ilias I, Grossman AB: The diagnosis and management of malignant phaeochromocytoma and paraganglioma. Endocr Relat Cancer 14:569–85, 2007
Tombol Z, Eder K, Kovacs A, et al.: MicroRNA expression profiling in benign (sporadic and hereditary) and recurring adrenal pheochromocytomas. Mod Pathol 23:1583–95, 2010
Meyer-Rochow GY, Jackson NE, Conaglen JV, et al.: MicroRNA profiling of benign and malignant pheochromocytomas identifies novel diagnostic and therapeutic targets. Endocr Relat Cancer 17:835–46, 2010
Grossman A, Pacak K, Sawka A, et al.: Biochemical diagnosis and localization of pheochromocytoma: can we reach a consensus? Ann N Y Acad Sci 1073:332–47, 2006
Michaud GY: The role of standards in the development and implementation of clinical laboratory tests: a domestic and global perspective. Cancer Biomark 1:209–16, 2005
Hall JA, Brown R, Paul J: An exploration into study design for biomarker identification: issues and recommendations. Cancer Genomics Proteomics 4:111–9, 2007
Mischak H, Allmaier G, Apweiler R, et al.: Recommendations for biomarker identification and qualification in clinical proteomics. Sci Transl Med 2:46ps2, 2010
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eisenhofer, G., Tischler, A.S. & de Krijger, R.R. Diagnostic Tests and Biomarkers for Pheochromocytoma and Extra-adrenal Paraganglioma: From Routine Laboratory Methods to Disease Stratification. Endocr Pathol 23, 4–14 (2012). https://doi.org/10.1007/s12022-011-9188-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12022-011-9188-1