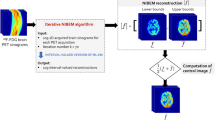
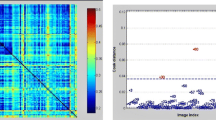
Abstract
18F–fluoro-deoxy-glucose Positron Emission Tomography (FDG-PET) allows early identification of neurodegeneration in dementia. The use of an optimized method based on the SPM software package highly improves diagnostic accuracy. However, the impact of different scanners for data acquisition on the SPM results and the effects of different pools of healthy subjects on the statistical comparison have not been investigated yet. Images from 144 AD patients acquired using six different PET scanners were analysed with an optimized single-subject SPM procedure to identify the typical AD hypometabolism pattern at single subject level. We compared between-scanners differences on the SPM outcomes in a factorial design. Single-subject SPM comparison analyses were also performed against a different group of healthy controls from the ADNI initiative. The concordance between the two analyses (112 vs. 157 control subjects) was tested using Dice scores. In addition, we applied the optimized single-subject SPM procedure to the FDG-PET data acquired with 3 different scanners in 57 MCI subjects, in order to assess for tomograph influence in early disease phase. All the patients showed comparable AD-like hypometabolic patterns, also in the prodromal phase, in spite of being acquired with different PET scanners. SPM statistical comparisons performed with the two different healthy control databases showed a high degree of concordance (76% average pattern volume overlap and 90% voxel-wise agreement in AD-related brain structures). The validated optimized SPM-based single-subject procedure is influenced neither by the scanners used for image acquisition, nor by differences in healthy control groups, thus implying a great reliability of this method for longitudinal and multicentre studies.




Similar content being viewed by others
References
Adam, L. E., Zaers, J., Ostertag, H., Trojan, H., Bellemann, M. E., & Brix, G. (1997). Performance evaluation of the whole-body PET scanner ECAT EXACT HR+ following the IEC standard. IEEE Transactions on Nuclear Science, 44(3 PART 2), 1172–1179. doi:10.1109/23.596983.
Alzheimer’s Disease Neuroimaging Initiative PET Technical Procedures Manual Version 9.5. (2006). http://adni.loni.usc.edu/wp-content/uploads/2010/09/PET-Tech_Procedures_Manual_v9.5.pdf
Anchisi, D., Borroni, B., Franceschi, M., Kerrouche, N., Kalbe, E., Beuthien-Beumann, B., et al. (2005). Heterogeneity of brain glucose metabolism in mild cognitive impairment and clinical progression to Alzheimer disease. Archives of Neurology, 62(11), 1728. doi:10.1001/archneur.62.11.1728.
Armstrong, M. J., Litvan, I., Lang, A. E., Bak, T. H., Bhatia, K. P., Borroni, B., et al. (2013). Criteria for the diagnosis of corticobasal degeneration. Neurology, 80(5), 496–503. doi:10.1212/WNL.0b013e31827f0fd1.
Bateman, R. J., Xiong, C., Benzinger, T. L. S. S., Fagan, A. M., Goate, A., Fox, N. C., et al. (2012). Clinical and biomarker changes in dominantly inherited Alzheimer’s disease. The New England Journal of Medicine, 367(9), 795–804. doi:10.1056/NEJMoa1202753.
Bettinardi, V., Danna, M., Savi, A., Lecchi, M., Castiglioni, I., Gilardi, M. C., et al. (2004). Performance evaluation of the new whole-body PET/CT scanner: discovery ST. European Journal of Nuclear Medicine and Molecular Imaging, 31(6), 867–881. doi:10.1007/s00259-003-1444-2.
Bettinardi, V., Presotto, L., Rapisarda, E., Picchio, M., Gianolli, L., & Gilardi, M. C. (2011). Physical performance of the new hybrid PET/CT discovery-690. Medical Physics, 38(10), 5394–5411. doi:10.1118/1.3635220.
Bohnen, N. I., Djang, D. S. W., Herholz, K., Anzai, Y., & Minoshima, S. (2012). Effectiveness and safety of 18F-FDG PET in the evaluation of dementia: a review of the recent literature. Journal of Nuclear Medicine, 53(1), 59–71. doi:10.2967/jnumed.111.096578.
Bonanni, L., Thomas, A., & Onofrj, M. (2006). Diagnosis and management of dementia with Lewy bodies: third report of the DLB consortium. Neurology, 66(9), 1455 . doi:10.1212/01.wnl.0000224698.67660.45.author reply 1455
Buchert, R., Wilke, F., Chakrabarti, B., Martin, B., Brenner, W., Mester, J., & Clausen, M. (2005). Adjusted scaling of FDG positron emission tomography images for statistical evaluation in patients with suspected Alzheimer’s disease. Journal of Neuroimaging : Official Journal of the American Society of Neuroimaging, 15(4), 348–355. doi:10.1177/1051228405280169.
Cerami, C., Della Rosa, P. A., Magnani, G., Santangelo, R., Marcone, A., Cappa, S. F., & Perani, D. (2015). Brain metabolic maps in mild cognitive impairment predict heterogeneity of progression to dementia. NeuroImage: Clinical, 7, 187–194. doi:10.1016/j.nicl.2014.12.004.
Cerami, C., Dodich, A., Lettieri, G., Iannaccone, S., Magnani, G., Marcone, A., et al. (2016). Different FDG-PET metabolic patterns at single-subject level in the behavioral variant of fronto-temporal dementia. Cortex, 83, 101–112. doi:10.1016/j.cortex.2016.07.008.
Chételat, G., Desgranges, B., de la Sayette, V., Viader, F., Eustache, F., & Baron, J.-C. (2003). Mild cognitive impairment: can FDG-PET predict who is to rapidly convert to Alzheimer’s disease? Neurology, 60(8), 1374–1377 http://www.ncbi.nlm.nih.gov/pubmed/12707450.
Chetelat, G., Desgranges, B., Landeau, B., Mezenge, F., Poline, J. B., de la Sayette, V., et al. (2007). Direct voxel-based comparison between grey matter hypometabolism and atrophy in Alzheimer’s disease. Brain, 131(1), 60–71. doi:10.1093/brain/awm288.
de Leon, M. J., Convit, A., Wolf, O. T., Tarshish, C. Y., DeSanti, S., Rusinek, H., et al. (2001). Prediction of cognitive decline in normal elderly subjects with 2-[(18)F]fluoro-2-deoxy-D-glucose/poitron-emission tomography (FDG/PET). Proceedings of the National Academy of Sciences of the United States of America, 98(19), 10966–10971. doi:10.1073/pnas.191044198.
Della Rosa, P. A., Cerami, C., Gallivanone, F., Prestia, A., Caroli, A., Castiglioni, I., et al. (2014). A standardized [18F]-FDG-PET template for spatial normalization in statistical parametric mapping of dementia. Neuroinformatics, 12(4), 575–593. doi:10.1007/s12021-014-9235-4.
Dubois, B., Feldman, H. H., Jacova, C., Hampel, H., Molinuevo, J. L., Blennow, K., et al. (2014). Advancing research diagnostic criteria for Alzheimer’s disease: the IWG-2 criteria. The Lancet Neurology, 13(6), 614–629. doi:10.1016/S1474-4422(14)70090-0.
Dukart, J., Mueller, K., Horstmann, A., Vogt, B., Frisch, S., Barthel, H., et al. (2010). Differential effects of global and cerebellar normalization on detection and differentiation of dementia in FDG-PET studies. NeuroImage, 49(2), 1490–1495. doi:10.1016/j.neuroimage.2009.09.017.
Eriksson, L., Wienhard, K., Eriksson, M., Casey, M. E., Knoess, C., Bruckbauer, T., et al. (2002). The ECAT HRRT: NEMA NEC evaluation of the HRRT system, the new high-resolution research tomograph. IEEE Transactions on Nuclear Science, 49 I(5), 2085–2088. doi:10.1109/TNS.2002.803784.
Foster, N. L., Heidebrink, J. L., Clark, C. M., Jagust, W. J., Arnold, S. E., Barbas, N. R., et al. (2007). FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer’s disease. Brain : a Journal of Neurology, 130(Pt 10), 2616–2635. doi:10.1093/brain/awm177.
Frisoni, G. B., Bocchetta, M., Chételat, G., Rabinovici, G. D., De Leon, M. J., Kaye, J., et al. (2013). Imaging markers for Alzheimer disease: which vs how. Neurology, 81(5), 487–500. doi:10.1212/WNL.0b013e31829d86e8.
Friston, K. J. (2002). Statistics I. In Brain Mapping: The Methods (pp. 605–631). London: Elsevier. doi:10.1016/B978-012693019-1/50024-1
Gallivanone, F., Della Rosa, P., Perani, D., Gilardi, M. C., & Castiglioni, I. (2014). The impact of different 18FDG PET Healthy Subject scans for comparison with single patient in SPM analysis. The quarterly journal of nuclear medicine and molecular imaging : official publication of the Italian Association of Nuclear Medicine (AIMN) [and] the International Association of Radiopharmacology (IAR), [and] Section of the Society of... http://www.ncbi.nlm.nih.gov/pubmed/25479418
Gorno-Tempini, M. L., Hillis, A. E., Weintraub, S., Kertesz, A., Mendez, M., Cappa, S. F., et al. (2011). Classification of primary progressive aphasia and its variants. Neurology, 76(11), 1006–1014. doi:10.1212/WNL.0b013e31821103e6.
Herholz, K. (2014). The role of PET quantification in neurological imaging: FDG and amyloid imaging in dementia. Clinical Translational Imaging, 2(4), 321–330. doi:10.1007/s40336-014-0073-z.
Herholz, K., Salmon, E., Perani, D., Baron, J.-C. C., Holthoff, V., Frölich, L., et al. (2002). Discrimination between Alzheimer dementia and controls by automated analysis of multicenter FDG PET. NeuroImage, 17(1), 302–316. doi:10.1006/nimg.2002.1208.
Iaccarino, L., Crespi, C., Della Rosa, P. A., Catricalà, E., Guidi, L., Marcone, A., et al. (2015). The semantic variant of primary progressive aphasia: clinical and neuroimaging evidence in single subjects. PloS One, 10(3), e0120197. doi:10.1371/journal.pone.0120197.
Iatrou, M., Ross, S. G., Manjeshwar, R. M., & Stearns, C. W. (2004). A fully 3D iterative image reconstruction algorithm incorporating data corrections. In IEEE Symposium Conference Record Nuclear Science 2004. (Vol. 4, pp. 2493–2497). New York: IEEE. doi:10.1109/NSSMIC.2004.1462761
Iatrou, M., Manjeshwar, R. M., Ross, S. G., Thielemans, K., & Stearns, C. W. (2006). 3D implementation of Scatter Estimation in 3D PET. In 2006 I.E. Nuclear Science Symposium Conference Record (pp. 2142–2145). IEEE. doi:10.1109/NSSMIC.2006.354338
Ishii, K., Kono, A. K., Sasaki, H., Miyamoto, N., Fukuda, T., Sakamoto, S., & Mori, E. (2006). Fully automatic diagnostic system for early- and late-onset mild Alzheimer’s disease using FDG PET and 3D-SSP. European Journal of Nuclear Medicine and Molecular Imaging, 33(5), 575–583. doi:10.1007/s00259-005-0015-0.
Jack, C. R., Knopman, D. S., Jagust, W. J., Petersen, R. C., Weiner, M. W., Aisen, P. S., et al. (2013). Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurology, 12(2), 207–216. doi:10.1016/S1474-4422(12)70291-0.
Jakoby, B. W., Bercier, Y., Watson, C. C. C., Rappoport, V., Young, J., Bendriem, B., & Townsend, D. W. W. (2006). Physical Performance and Clinical Workflow of a new LSO HI-REZ PET/CT Scanner. In 2006 I.E. Nuclear Science Symposium Conference Record (Vol. 5, pp. 3130–3134). New York: IEEE. doi:10.1109/NSSMIC.2006.356538
Joshi, A., Koeppe, R. A., & Fessler, J. A. (2009). Reducing between scanner differences in multi-center PET studies. NeuroImage, 46(1), 154–159. doi:10.1016/j.neuroimage.2009.01.057.
Landau, S. M., Harvey, D., Madison, C. M., Reiman, E. M., Foster, N. L., Aisen, P. S., et al. (2010). Comparing predictors of conversion and decline in mild cognitive impairment. Neurology, 75(3), 230–238. doi:10.1212/WNL.0b013e3181e8e8b8.
Lewellen, T. K., Kohlmyer, S. G., Miyaoka, R. S., Kaplan, M. S., Stearns, C. W., & Schubert, S. F. (1996). Investigation of the performance of the General Electric ADVANCE positron emission tomograph in 3D mode. IEEE Transactions on Nuclear Science, 43(4), 2199–2206. doi:10.1109/23.531882.
Liu, X., Comtat, C., Michel, C., Kinahan, P., Defrise, M., & Townsend, D. (2001). Comparison of 3-D reconstruction with 3D-OSEM and with FORE + OSEM for PET. IEEE Transactions on Medical Imaging, 20(8), 804–814. doi:10.1109/42.938248.
Manjeshwar, R. M., Ross, S. G., Iatrou, M., Deller, T. W., & Stearns, C. W. (2007). Fully 3D PET iterative reconstruction using distance-driven projectors and native scanner geometry. IEEE Nuclear Science Symposium Conference Record, 5, 2804–2807. doi:10.1109/NSSMIC.2006.356461.
Martino, M. E., de Villoria, J. G., Lacalle-Aurioles, M., Olazarán, J., Cruz, I., Navarro, E., et al. (2013). Comparison of different methods of spatial normalization of FDG-PET brain images in the voxel-wise analysis of MCI patients and controls. Annals of Nuclear Medicine, 27(7), 600–609. doi:10.1007/s12149-013-0723-7.
McKeith, I. G., Dickson, D. W., Lowe, J., Emre, M., O’Brien, J. T., Feldman, H., et al. (2005). Diagnosis and management of dementia with Lewy bodies: third report of the DLB consortium. Neurology, 65(12), 1863–1872. doi:10.1212/01.wnl.0000187889.17253.b1.
McKhann, G. M., Knopman, D. S., Chertkow, H., Hyman, B. T., Jack, C. R., Kawas, C. H., Klunk, W. E., Koroshetz, W. J., Manly, J. J., Mayeux, R., Mohs, R. C., Morris, J. C., Rossor, M. N., Scheltens, P., Carrillo, M. C., Thies, B., Weintraub, S., Phelps, C. H., et al. (2011a). The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement, 7(3), 270–279. doi:10.1016/j.jalz.2011.03.005.
McKhann, G. M., Knopman, D. S., Chertkow, H., Hyman, B. T., Jack, C. R., Kawas, C. H., Klunk, W. E., Koroshetz, W. J., Manly, J. J., Mayeux, R., Mohs, R. C., Morris, J. C., Rossor, M. N., Scheltens, P., Carrillo, M. C., Thies, B., Weintraub, S., & Phelps, C. H. (2011b). The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement, 7(3), 263–269. doi:10.1016/j.jalz.2011.03.005.
Minoshima, S., Frey, K. A., Koeppe, R. A., Foster, N. L., & Kuhl, D. E. (1995). A diagnostic approach in Alzheimer’s disease using three-dimensional stereotactic surface projections of fluorine-18-FDG PET. Journal of Nuclear Medicine: Official Publication, Society of Nuclear Medicine, 36(7), 1238–1248.
Minoshima, S., Foster, N. L., Sima, A. A. F., Frey, K. A., Albin, R. L., & Kuhl, D. E. (2001). Alzheimer’s disease versus dementia with Lewy bodies: cerebral metabolic distinction with autopsy confirmation. Annals of Neurology, 50(3), 358–365. doi:10.1002/ana.1133.
Morbelli, S., Garibotto, V., Van De Giessen, E., Arbizu, J., Chételat, G., Drezgza, A., et al. (2015a). A Cochrane review on brain [18F]FDG PET in dementia: limitations and future perspectives. European Journal of Nuclear Medicine and Molecular Imaging, 42(10), 1487–1491. doi:10.1007/s00259-015-3098-2.
Morbelli, S., Brugnolo, A., Bossert, I., Buschiazzo, A., Frisoni, G. B., Galluzzi, S., et al. (2015b). Visual versus semi-quantitative analysis of 18F-FDG-PET in amnestic MCI: an European Alzheimer’s disease consortium (EADC) project. Journal of Alzheimer's Disease, 44(3), 815–826. doi:10.3233/JAD-142229.
Mosconi, L., Mistur, R., Switalski, R., Tsui, W. H., Glodzik, L., Li, Y., et al. (2009). FDG-PET changes in brain glucose metabolism from normal cognition to pathologically verified Alzheimer’s disease. European Journal of Nuclear Medicine and Molecular Imaging, 36(5), 811–822. doi:10.1007/s00259-008-1039-z.
Perani, D. (2014). FDG-PET and amyloid-PET imaging: the diverging paths. Current Opinion in Neurology, 27(4), 405–413. doi:10.1097/WCO.0000000000000109.
Perani, D., Schillaci, O., Padovani, A., Nobili, F. M., Iaccarino, L., Della Rosa, P. A., et al. (2014a). Erratum to “a survey of FDG- and amyloid-PET imaging in dementia and GRADE analysis”. BioMed Research International, 2014, 1–1. doi:10.1155/2014/246586.
Perani, D., Della Rosa, P. A., Cerami, C., Gallivanone, F., Fallanca, F., Vanoli, E. G., et al. (2014b). Validation of an optimized SPM procedure for FDG-PET in dementia diagnosis in a clinical setting. NeuroImage: Clinical, 6, 445–454. doi:10.1016/j.nicl.2014.10.009.
Perani, D., Cerami, C., Caminiti, S. P., Santangelo, R., Coppi, E., Ferrari, L., et al. (2015). Cross-validation of biomarkers for the early differential diagnosis and prognosis of dementia in a clinical setting. European Journal of Nuclear Medicine and Molecular Imaging. doi:10.1007/s00259-015-3170-y.
Rascovsky, K., Hodges, J. R., Knopman, D., Mendez, M. F., Kramer, J. H., Neuhaus, J., et al. (2011). Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain, 134(9), 2456–2477. doi:10.1093/brain/awr179.
Sibomana, M., Keller, S. H., Stute, S., & Comtat, C. (2012). Benefits of 3D scatter correction for the HRRT - a large axial FOV PET scanner. In 2012 I.E. Nuclear Science Symposium and Medical Imaging Conference Record (NSS/MIC) (pp. 2954–2957). New York: IEEE. doi:10.1109/NSSMIC.2012.6551675
Slomka, P. J., Pan, T., Berman, D. S., & Germano, G. (2015). Advances in SPECT and PET hardware. Progress in Cardiovascular Diseases, 57(6), 566–578. doi:10.1016/j.pcad.2015.02.002.
Smailagic, N., Vacante, M., Hyde, C., Martin, S., Ukoumunne, O., & Sachpekidis, C. (2015). 18 F-FDG PET for the early diagnosis of Alzheimer’s disease dementia and other dementias in people with mild cognitive impairment (MCI). In C. Sachpekidis (Ed.), Cochrane database of systematic reviews. Chichester, UK: John Wiley & Sons, Ltd.. doi:10.1002/14651858.CD010632.pub2.
Sperling, R. A., Aisen, P. S., Beckett, L. A., Bennett, D. A., Craft, S., Fagan, A. M., et al. (2011). Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement, 7(3), 280–292. doi:10.1016/j.jalz.2011.03.003.
Teras, M., Tolvanen, T., Johansson, J. J., Williams, J. J., & Knuuti, J. (2007). Performance of the new generation of whole-body PET/CT scanners: discovery STE and discovery VCT. European Journal of Nuclear Medicine and Molecular Imaging, 34(10), 1683–1692. doi:10.1007/s00259-007-0493-3.
Teune, L. K., Bartels, A. L., De Jong, B. M., Willemsen, A. T. M., Eshuis, S. A., De Vries, J. J., et al. (2010). Typical cerebral metabolic patterns in neurodegenerative brain diseases. Movement Disorders, 25(14), 2395–2404. doi:10.1002/mds.23291.
Townsend, D. W., Geissbuhler, A., Defrise, M., Hoffman, E. J., Spinks, T. J., Bailey, D. L., et al. (1991). Fully three-dimensional reconstruction for a PET camera with retractable septa. IEEE Transactions on Medical Imaging, 10(4), 505–512. doi:10.1109/42.108584.
Tzourio-Mazoyer, N., Landeau, B., Papathanassiou, D., Crivello, F., Etard, O., Delcroix, N., et al. (2002). Automated anatomical labeling of activations in SPM using a macroscopic anatomical Parcellation of the MNI MRI single-subject brain. NeuroImage, 15(1), 273–289. doi:10.1006/nimg.2001.0978.
Villemagne, V. L., & Chételat, G. (2016). Neuroimaging biomarkers in Alzheimer’s disease and other dementias. Ageing Research Reviews. doi:10.1016/j.arr.2016.01.004.
Zaidi, H. (2000). Comparative evaluation of scatter correction techniques in 3D positron emission tomography. European Journal of Nuclear Medicine, 27(12), 1813–1826. doi:10.1007/s002590000385.
Acknowledgments
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd. and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (www.fnih.org). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California. This research was funded by EU FP7 INMIND Project (FP7-HEALTH-2013, grant agreement no. 278850) and This work was supported by the Italian Ministry of Health (Ricerca Finalizzata Progetto Reti Nazionale AD NET-2011-02346784).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Data used in preparation of this article were obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu). As such, the investigators within the ADNI contributed to the design and implementation of ADNI and/or provided data but did not participate in analysis or writing of this report. A complete listing of ADNI investigators can be found at: http://adni.loni.usc.edu/wp-content/uploads/how_to_apply/ADNI_Acknowledgement_List.pdf
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Presotto, L., Ballarini, T., Caminiti, S.P. et al. Validation of 18F–FDG-PET Single-Subject Optimized SPM Procedure with Different PET Scanners. Neuroinform 15, 151–163 (2017). https://doi.org/10.1007/s12021-016-9322-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12021-016-9322-9