Abstract
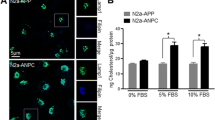
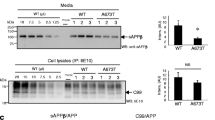
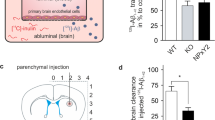
Previous studies have demonstrated that the low-density lipoprotein receptor-related protein-1 (LRP1) plays conflicting roles in Alzheimer’s disease (AD) pathogenesis, clearing β-amyloid (Aβ) from the brain while also enhancing APP endocytosis and resultant amyloidogenic processing. We have recently discovered that co-expression of mutant LRP1 C-terminal domain (LRP1-CT C4408R) with Swedish mutant amyloid precursor protein (APPswe) in Chinese hamster ovary (CHO) cells decreases Aβ production, while also increasing sAPPα and APP α-C-terminal fragment (α-CTF), compared with CHO cells expressing APPswe alone. Surprisingly, the location of this mutation on LRP1 corresponded with the α-secretase cleavage site of APP. Further experimentation confirmed that in CHO cells expressing APPswe or wild-type APP (APPwt), co-expression of LRP1-CT C4408R decreases Aβ and increases sAPPα and α-CTF compared with co-expression of wild-type LRP1-CT. In addition, LRP1-CT C4408R enhanced the unglycosylated form of LRP1-CT and reduced APP endocytosis as determined by flow cytometry. This finding identifies a point mutation in LRP1 which slows LRP1-CT-mediated APP endocytosis and amyloidogenic processing, while enhancing APP α-secretase cleavage, thus demonstrating a potential novel target for slowing AD pathogenesis.




Similar content being viewed by others
References
Bading, J. R., et al. (2002). Brain clearance of Alzheimer’s amyloid-beta40 in the squirrel monkey: A SPECT study in a primate model of cerebral amyloid angiopathy. Journal of Drug Targeting, 10(4), 359–368.
Bell, R. D., et al. (2009). SRF and myocardin regulate LRP-mediated amyloid-beta clearance in brain vascular cells. Nature Cell Biology, 11(2), 143–153.
Boucher, P., & Herz, J. (2011). Signaling through LRP1: Protection from atherosclerosis and beyond. Biochemical Pharmacology, 81(1), 1–5.
Bu, G., et al. (2006). LRP in amyloid-beta production and metabolism. Annals of the New York Academy of Sciences, 1086, 35–53.
Cam, J. A., et al. (2005). Rapid endocytosis of the low density lipoprotein receptor-related protein modulates cell surface distribution and processing of the beta-amyloid precursor protein. Journal of Biological Chemistry, 280(15), 15464–15470.
Cohen, T. J., et al. (2011). The acetylation of tau inhibits its function and promotes pathological tau aggregation. Nature Communications, 2, 252.
Deane, R., et al. (2004). LRP/amyloid beta-peptide interaction mediates differential brain efflux of Abeta isoforms. Neuron, 43(3), 333–344.
Deane, R., et al. (2008). apoE isoform-specific disruption of amyloid beta peptide clearance from mouse brain. The Journal of Clinical Investigation, 118(12), 4002–4013.
DeMattos, R. B., et al. (2004). ApoE and clusterin cooperatively suppress Abeta levels and deposition: Evidence that ApoE regulates extracellular Abeta metabolism in vivo. Neuron, 41(2), 193–202.
Deng, J., et al. (2015). Soluble amyloid precursor protein alpha inhibits tau phosphorylation through modulation of GSK3beta signaling pathway. Journal of Neurochemistry, 135(3), 630–637.
Dieckmann, M., et al. (2010). Lipoprotein receptors—An evolutionarily ancient multifunctional receptor family. Biological Chemistry, 391(11), 1341–1363.
Donahue, J. E., et al. (2006). RAGE, LRP-1, and amyloid-beta protein in Alzheimer’s disease. Acta Neuropathologica, 112(4), 405–415.
Hardy, J., & Selkoe, D. J. (2002). The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science, 297(5580), 353–356.
Herring, A., et al. (2008). Environmental enrichment counteracts Alzheimer’s neurovascular dysfunction in TgCRND8 mice. Brain Pathology, 18(1), 32–39.
Herz, J., & Strickland, D. K. (2001). LRP: A multifunctional scavenger and signaling receptor. The Journal of Clinical Investigation, 108(6), 779–784.
Hussain, M. M., et al. (1999). The mammalian low-density lipoprotein receptor family. Annual Review of Nutrition, 19, 141–172.
Kimberly, W. T., et al. (2003). Gamma-secretase is a membrane protein complex comprised of presenilin, nicastrin, Aph-1, and Pen-2. Proceedings of the National Academy of Sciences USA, 100(11), 6382–6387.
Koo, E. H., & Squazzo, S. L. (1994). Evidence that production and release of amyloid beta-protein involves the endocytic pathway. Journal of Biological Chemistry, 269(26), 17386–17389.
Kounnas, M. Z., et al. (1995). LDL receptor-related protein, a multifunctional ApoE receptor, binds secreted beta-amyloid precursor protein and mediates its degradation. Cell, 82(2), 331–340.
Lefort, R., et al. (2012). Cross-linking of cell surface amyloid precursor protein leads to increased beta-amyloid peptide production in hippocampal neurons: Implications for Alzheimer’s disease. Journal of Neuroscience, 32(31), 10674–10685.
Li, S., et al. (2015). Swedish mutant APP-based BACE1 binding site peptide reduces APP β-cleavage and cerebral Aβ levels in Alzheimer’s mice. Scientific Reports, 5, 11322.
Lillis, A. P., et al. (2008). LDL receptor-related protein 1: Unique tissue-specific functions revealed by selective gene knockout studies. Physiological Reviews, 88(3), 887–918.
Mawuenyega, K. G., et al. (2010). Decreased clearance of CNS beta-amyloid in Alzheimer’s disease. Science, 330(6012), 1774.
Moestrup, S. K., et al. (1992). Distribution of the alpha 2-macroglobulin receptor/low density lipoprotein receptor-related protein in human tissues. Cell and Tissue Research, 269(3), 375–382.
Narita, M., et al. (1997). Alpha2-macroglobulin complexes with and mediates the endocytosis of beta-amyloid peptide via cell surface low-density lipoprotein receptor-related protein. Journal of Neurochemistry, 69(5), 1904–1911.
Neels, J. G., et al. (1999). The second and fourth cluster of class A cysteine-rich repeats of the low density lipoprotein receptor-related protein share ligand-binding properties. Journal of Biological Chemistry, 274(44), 31305–31311.
Parvathy, S., et al. (1999). Cleavage of Alzheimer’s amyloid precursor protein by alpha-secretase occurs at the surface of neuronal cells. Biochemistry, 38(30), 9728–9734.
Pietrzik, C. U., et al. (2002). The cytoplasmic domain of the LDL receptor-related protein regulates multiple steps in APP processing. EMBO Journal, 21(21), 5691–5700.
Pietrzik, C. U., et al. (2004). FE65 constitutes the functional link between the low-density lipoprotein receptor-related protein and the amyloid precursor protein. Journal of Neuroscience, 24(17), 4259–4265.
Qiu, Z., et al. (1999). Alpha2-macroglobulin enhances the clearance of endogenous soluble beta-amyloid peptide via low-density lipoprotein receptor-related protein in cortical neurons. Journal of Neurochemistry, 73(4), 1393–1398.
Rezai-Zadeh, K., et al. (2005). Green tea epigallocatechin-3-gallate (EGCG) modulates amyloid precursor protein cleavage and reduces cerebral amyloidosis in Alzheimer transgenic mice. Journal of Neuroscience, 25(38), 8807–8814.
Sagare, A., et al. (2007). Clearance of amyloid-beta by circulating lipoprotein receptors. Nature Medicine, 13(9), 1029–1031.
Sagare, A. P., et al. (2012). Low-density lipoprotein receptor-related protein 1: A physiological Abeta homeostatic mechanism with multiple therapeutic opportunities. Pharmacology & Therapeutics, 136(1), 94–105.
Shibata, M., et al. (2000). Clearance of Alzheimer’s amyloid-ss(1-40) peptide from brain by LDL receptor-related protein-1 at the blood-brain barrier. The Journal of Clinical Investigation, 106(12), 1489–1499.
Silverberg, G. D., et al. (2010). Amyloid efflux transporter expression at the blood–brain barrier declines in normal aging. Journal of Neuropathology and Experimental Neurology, 69(10), 1034–1043.
Ulery, P. G., et al. (2000). Modulation of beta-amyloid precursor protein processing by the low density lipoprotein receptor-related protein (LRP). Evidence that LRP contributes to the pathogenesis of Alzheimer’s disease. Journal of Biological Chemistry, 275(10), 7410–7415.
Vassar, R., et al. (1999). Beta-secretase cleavage of Alzheimer’s amyloid precursor protein by the transmembrane aspartic protease BACE. Science, 286(5440), 735–741.
Waldron, E., et al. (2008). LRP1 modulates APP trafficking along early compartments of the secretory pathway. Neurobiology of Diseases, 31(2), 188–197.
Zerbinatti, C. V., & Bu, G. (2005). LRP and Alzheimer’s disease. Reviews in the Neurosciences, 16(2), 123–135.
Zerbinatti, C. V., et al. (2004). Increased soluble amyloid-beta peptide and memory deficits in amyloid model mice overexpressing the low-density lipoprotein receptor-related protein. Proceedings of the National Academy of Sciences USA, 101(4), 1075–1080.
Zhang, X., & Song, W. (2013). The role of APP and BACE1 trafficking in APP processing and amyloid-beta generation. Alzheimer’s Research & Therapy, 5(5), 46.
Zlokovic, B. V., et al. (2010). Low-density lipoprotein receptor-related protein-1: A serial clearance homeostatic mechanism controlling Alzheimer’s amyloid beta-peptide elimination from the brain. Journal of Neurochemistry, 115(5), 1077–1089.
Acknowledgements
This work was supported by the NIH/NIA (R01AG050253, R01AG032432) and the Silver Endowment to J Tan. We would like to thank Dr. David Kang, Dr. Demian Obregon and Dr. Doug Shytle for their helpful discussion and Mr. Yang Gao for his technical support in confocal image analysis. We also appreciated Dr. Stefanie Hahn and Dr. Sascha Weggen for kindly providing us with CHO cells overexpressing human APP bearing the Swedish mutation (APPswe) and human LRP-CT.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Hou, H., Habib, A., Zi, D. et al. Low-Density Lipoprotein Receptor-Related Protein-1 (LRP1) C4408R Mutant Promotes Amyloid Precursor Protein (APP) α-Cleavage in Vitro. Neuromol Med 19, 300–308 (2017). https://doi.org/10.1007/s12017-017-8446-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-017-8446-x