Abstract
Systemic sclerosis is a multifactorial and heterogeneous disease. Genetic and environmental factors are known to interplay in the onset and progression of systemic sclerosis. Sex plays an important and determinant role in the development of such a disorder. Systemic sclerosis shows a significant female preponderance. However, the reason for this female preponderance is incompletely understood. Hormonal status, genetic and epigenetic differences, and lifestyle have been considered in order to explain female preponderance in systemic sclerosis. Sex chromosomes play a determinant role in contributing to systemic sclerosis onset and progression, as well as in its sex-biased prevalence. It is known, in fact, that X chromosome contains many sex- and immuno-related genes, thus contributing to immuno tolerance and sex hormone status. This review focuses mainly on the recent progress on epigenetic mechanisms—exclusively linked to the X chromosome—which would contribute to the development of systemic sclerosis. Furthermore, we report also some hypotheses (dealing with skewed X chromosome inactivation, X gene reactivation, acquired monosomy) that have been proposed in order to justify the female preponderance in autoimmune diseases. However, despite the intensive efforts in elucidating the mechanisms involved in the pathogenesis of systemic sclerosis, many questions remain still unanswered.
Similar content being viewed by others
Introduction
With a prevalence of 4 per 10,000 and a sex ratio (F/M) ranging from 5:1 to 12:1 [1,2], systemic sclerosis (SSc) is a rare and severe autoimmune disorder characterized by progressive fibrosis, vascular damage, and immune abnormalities [3–5]. SSc is a multifactorial disease, and genetic and environmental factors are known to interplay in the onset and progression of SSc [6]. Breaking of epigenetic homeostasis by environmental agents causes alterations in gene expression that would contribute to the development of SSc.
Sex plays an important and determinant role in the development of such a disease. SSc, as many other autoimmune diseases, shows a significant female preponderance. However, the reason for this female preponderance is incompletely understood.
Different factors may be involved in this sexual dimorphism, such as sex and reproductive hormones [7,8], fetal microchimerism [9,10], and different environmental exposure [11].
This review focuses mainly on the mechanisms—exclusively linked to the X chromosome—which contribute to the development of systemic sclerosis. For a detailed discussion on other mechanisms involved in the susceptibility to autoimmune diseases, including SSc, we suggest the reading of other excellent reviews [2,12,13].
The first part of this review deals with epigenetic regulation changes, such as X chromosome inactivation and loss of epigenetic control, which could contribute to the susceptibility of SSc. Then, a section is dedicated to the sex hormones, which could be potentially responsible for the sex-dependent discrepancy of disease. Sex hormones are known to influence genes on sex chromosome, thus exerting an effect on autoimmunity. Finally, a brief description on X-linked single nucleotide polymorphisms associated with the susceptibility to SSc is given.
The Female Preponderance in Systemic Sclerosis
SSc shows a significant female preponderance [14]. However, as shown in Table 1, sex ratio (F/M) for this disease ranges from 4 to 11. The female preponderance, such as other SSc clinical features, varies in different geographical areas and ethnicities [31], thus reflecting the possibility that genetic factors may underlie these differential phenotypic aspects. Another important factor to be considered is the population age. In fact, a female population in childbearing age is known to increase the F/M ratio [32]. Moreover, different criteria for definition and classification of SSc may make sex ratios imprecise and biased [23,33].
It is noteworthy that female preponderance reflects the different incidence of the disease between the sexes, and not severity. Male SSc patients have a more severe prognosis compared to female ones [15,34]. In conclusion, we cannot exclude that high rate of mortality in male population could influences the sex ratio of SSc.
X Chromosome and Dosage Compensation
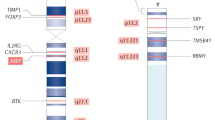
The X chromosome contains 1,098 genes, 70 % of which have been associated to various diseases, whereas Y chromosome encodes only a small number of genes, about 100 genes, most of which are different from the ones encoded by the X chromosome [35].
In order to achieve a dosage compensation in females, one copy of X chromosome is randomly inactivated by transcriptional silencing during early development [36,37]. More than 70 % of genes on the X chromosome are silenced. In females, the effect of dosage compensation is the occurrence of a mosaic expression of maternally or paternally derived X chromosome in different cell populations.
The mechanisms involved in this inactivation have not been fully clarified. DNA methylation, histone acetylation, methylation, and phosphorylation, as well as microRNAs, contribute to the initiation and maintenance of X chromosome inactivation [38–40].
Briefly, X chromosome inactivation begins at position Xq13.3, in the X Inactivation Center, a complex locus producing a large untranslated RNA, the X-inactive Specific transcript (XIST) and its antisense (TSIX) [37]. The XIST gene is expressed in the chosen inactivated X chromosome, and its RNA transcript binds to chromatin in order to inactivate it [41]. As proposed by Lyon [42], long interspersed repeat elements in the X chromosome may serve as specific signals for gene inactivation. However, this model is not sufficient to explain the mechanisms underlying gene inactivation, since these elements seem not to be essential for Xist localization [43]. Long terminal repeats and inverted repeat sequence represent additional regulatory sequence which may be involved in XCI [44,45].
Subsequently, several epigenetic modifications which contribute to the X chromosome inactivation and maintaining take place [46]. Following its recruitment by Xist, the polycomb complexes cause some histone changes on the X chromosome [47]. DNA methylation is required for stable maintenance of XCI only in embryo.
For the presence of one copy of X chromosome, males are more exposed to deleterious mutations on the X chromosome [48]. Therefore, having two copies of X chromosome, as in females, may be advantageous. Furthermore, a drastic skewing of X chromosome inactivation could facilitate, in females, the silencing of a harmful mutation. However, as discussed in the next section, the presence of an altered X chromosome inactivation in females may play an important role in the increased risk of autoimmunity. Furthermore, it has been shown, through transgenic mouse models, that XX complement confers greater susceptibility to autoimmune diseases, as compared with XY [49]. One explanation, discussed in detail in next sections, of this discrepancy between males and females is that female X-linked self-antigens may bypass their presentation in the thymus, thus avoiding the central tolerance induction.
X chromosome contains many important immune-related genes, such as interleukin 2 receptor γ (IL2RG), forkhead box P3 (FOXP3), CD40 ligand (CD40L, CD154), and interleukin-1 receptor-associated kinase 1 (IRAK1) [50–53]. As shown in Table 2, many of these X-linked and immuno-related genes have been associated with SSc.
Skewed X Inactivation
Skewed X chromosome inactivation (sXCI) is the preferential inactivation of one X chromosome in females, leading to a deviation from the theoretical 1:1 ratio [66]. Although skewed X inactivation is considered a normal process, it can cause hemizygosity, a possible condition which could give rise to some X-linked recessive disease [67]. Moreover, although this phenomenon is considered to be stable for all descendant cells, the frequency of sXCI, present in peripheral blood cells, increases with age [68].
In sXCI, some specific genes show different patterns of inactivation, which could contribute to the heterogeneity in gene expression among females [69], thus influencing immune regulation. Until now, there is no evidence for a genetic mechanism involved in human sXCI, and most of observations on X chromosome inactivation skewing suggest the occurrence of mechanisms acquired secondarily [70].
It has been hypothesized that in random X inactivation, the tolerance mechanisms are well performed because of the expression of self-antigens on both X chromosomes. However, under nonrandom X inactivation conditions, such as in sXCI, thymic dendritic cells which express one copy of X chromosome fail in recognizing thymic cells, thus causing some thymocytes to escape from negative central selection [71–73] (Fig. 1a).
Several hypotheses, linked to X chromosome abnormalities, have been proposed to explain the preponderance of autoimmune disorders in females. Such hypothesis may be applied also for SSc. a sXCI may induce a loss of mosaicism in females, leading ultimately to autoimmunity. According to this model [43–45], females with a normal (random) XCI have two subpopulations of thymic dendritic cells (DC) expressing maternal (m) or paternal (p) X-linked self-antigens. Under this condition, potential autoreactive thymocytes (thy) undergo a negative selection. On the contrary, in females with a severe sXCI, thymic dendritic cells expressing self antigens of one parent, will tolerize only the thymocytes containing the self-antigens from other parent. Such thymocytes will escape thymic negative selection. b Reactivation of genes from inactive X chromosome could cause an overexpression of X immunorelated genes [74,75]. c Acquired haploinsufficiency, caused by X monosomy, may induce autoantibodies production by peripheral B cells (BC) [76]. Each mechanism is not sufficient to explain SSc development, and one of them must be regarded as a possible cofactor, together with other genetic and epigenetic factors, in the onset of the disorder
sXCI could be involved in the pathogenesis of SSc. Ozbalkan et al., with the analysis of the methylation status in the androgen receptor gene, have shown an association between X inactivation and female predisposition to systemic sclerosis. In particular, they found a high rate of a skewed pattern of X chromosome inactivation in DNA of peripheral blood cells from patients with SSc compared to the control subjects [77]. Broen et al., showing a sXCI pattern in female patients affected by SSc, have found that such a skewed pattern was not restricted to a specific cell population, thus suggesting the occurrence of this inactivation in precursor cell populations. Moreover, they observed that the sXCI in SSc was associated with decreased expression of FOXP3, a member of the forkhead family of transcription factors and essential for regulatory CD4+ T cells [78].
What is the relationship between skewed X inactivation and SSc? According to Ozcelik, the failure of self-tolerance could arise from the skewing and not vice versa because of the extreme degree of skewing in SSc patients. Moreover, this author speculated that any mutation, which compromises cell survival, could induce skewed X inactivation [73].
In conclusion, it is noteworthy that there is a strong interplay between non-X-linked genes and X inactivation. In particular, non-X-linked mutations may influence X inactivation and expression of X-linked genes, through their ability in regulating transcriptional and/or translational events. Similarly, molecular abnormalities on the X chromosome may influence gene expressions from other chromosome [79].
Escaping Gene Dosage Compensation
It is known that about 15 % of genes escape X chromosome silencing in female humans. Furthermore, an additional 10 % of X-linked genes show a variable individual- and/or tissue-dependent profile of inactivation [69]. Thus, females may show substantial heterogeneity in X-linked gene expression. Although in normal conditions X chromosome inactivation is stably maintained, many environmental factors may induce to a loss of epigenetic control, which can reactivate silenced X-linked genes.
Gene escaping inactivation is a nonrandom event. X-linked gene escaping inactivation is known to be clustered and preferentially located in the distal portion of the X chromosome short arm [69]. The mechanisms involved in XCI escaping are unclear. In addition to the long interspersed repeat elements and the long terminal repeats, AT-rich motifs seem to be involved in escaping XCI with the control of Xist RNA recruitment [80]. Furthermore, other noncoding RNA may play an important role in gene escaping inactivation [81]. Finally, boundary elements, acting as insulator sequences, may arrest the spreading of heterochromatin into region escaping inactivation [82].
Several genes escaping from XCI have been identified, including tissue inhibitor of metalloproteinases 1 (TIMP1), Il-1R associated kinase (IRAK1), and inhibitor of nuclear factor-κB kinase-γ (IKBKB), which have been shown to be associated to the development and/or to the different clinical subsets of SSc [59,83,84].
These phenomena, resulting in sex-biased gene expression, could explain sex-specific phenotypes in complex diseases [85]. An altered gene dosage of X-linked genes may be implicated in the loss of immune tolerance and in the female preponderance to develop autoimmune disorders [48,86].
The reversal of X chromosome inactivation, X chromosome reactivation (XCR), is a critical step for diversifying X-linked gene pool in germ cell formation [87,88]. Mechanisms involved in X chromosome reactivation remain unclear. In some autoimmune diseases, it has been demonstrated that X-linked genes may be reactivated by DNA methyltransferase inhibitors [89].
Selmi et al., with a genomewide fine mapping of DNA methylation, found gene hyper- or hypomethylation exclusively located on X chromosome of SSc monozygotic twins [61].
The interaction between CD40 and its ligand (CD40L) plays a role in SSc by stimulating B cells and fibroblasts [90,91]. CD40 and CD40L expression has been shown to be increased in fibroblast from SSc patients [91,92]. Furthermore, CD40L expression was demonstrated to be elevated only in SSc female patients [75]. Such a gene reactivation on the inactive X chromosome would double the expression of related proteins in women [74], thus providing an explanation for the female predominance of SSc [75] (Fig. 1b).
Hypermethylation of the CpG islands and histone deacetylation in the promoter region FLI1, a collagen suppressor gene, were shown to occur in SSc fibroblasts and skin biopsy specimens by Wang et al. [93]. On the contrary, a reduced global DNA methylation was observed in CD4+ T cell DNA from SSc patients [60].
X Chromosome Monosomy
In sXCI, some specific genes show different patterns of inactivation, which could contribute to the heterogeneity in gene expression among females [69], thus influencing immune regulation. By using fluorescence in situ hybridization, a higher rate of monosomy was found in peripheral blood cells from SSc patients in comparison to healthy subjects [76]. Moreover, these increased monosomy rates have been mainly found in lymphocytes.
An unusual case of morphea, a localized form of SSc, has been found to be associated with a low-grade mosaic Turner’s syndrome (karyotype 45, X0) [94]. In conclusion, X chromosome haploinsufficiency, caused by X chromosome monosomy, may be involved in the development of SSc (Fig. 1c).
X-linked Noncoding RNA
Recently, noncoding RNAs (ncRNAs) have attracted attention for its involvement in the pathogenesis of various diseases, such as autoimmune disorders. Noncoding RNA, regulatory elements of gene expression, includes microRNA (miRNA), long noncoding RNA (lncRNA), circular RNA [95,96].
About 10 % of microRNA, a family of small double-stranded noncoding RNA, is localized on X chromosome, suggesting a possible role in regulating gene expression, as by transcriptional gene silencing [97,98]. X-linked miRNA may be involved in immune regulation. It is possible that X-linked miRNAs, which escape inactivation or are subject to skewed X inactivation, may influence immune response in females [99].
Recently, many miRNAs—some of which are involved in autoimmune and fibrotic processes, and located on X-chromosome—were observed to be dysregulated in skin tissue from SSc patients, and different regulation patterns were associated to the different subtypes of the disease [100–102].
ncRNAs, together with the above discussed epigenetic mechanisms, are known to be important players in the homeostasis and function of the immune system. In particular, ncRNAs are molecular regulators involved in many autoimmune diseases. Since many studies on these molecules rely on their expression profile in patient peripheral blood mononuclear cells and target tissues, it is hoped that further studies on various cell subpopulations, such as on lymphocyte subpopulations, may help to elucidate the mechanisms responsible of the pathogenesis of autoimmune diseases, including SSc.
Sex Hormones and X Chromosome
Sex chromosomes and sex hormones are profoundly interconnected. It is known that some X- and Y-linked genes are needed for the differentiation of gonads, which in turn are needed for the synthesis of sex hormones, as testosterone and estrogens. Sex hormone-induced signalling pathways may modulate genes on sex chromosomes [103].
It is known that females have stronger immune response compared with males. Such differences may be caused by sex hormones, through their modulation of Th1/Th2 response [11]. Generally speaking, androgens could promote a Th1 response and activation of CD8+ cells. On the contrary, estrogens could favor a Th2 response [103–105].
It was suggested that the deep partnership between sex chromosomes and sex hormones would reciprocally antagonize their effects in order to minimize the differences between male and female immune response [106]. Estrogens play an important role in lymphocyte maturation and activation, as well as in the synthesis of cytokines and antibodies [107,108]. Moreover, estrogen receptors participate in innate and adaptive immunity, regulating activity of antigen-presenting cells and dendritic cells [109].
Such a sex-biased difference in immune response could explain the predominance of autoimmune disorders in females. It is known, in fact, that estrogens, inducing an extramedullary hematopoiesis, could favor the autoreactive B cell escaping from negative selection. Moreover, estrogens are able to promote the survival of the autoreactive T cells [110].
Since SSc occurs in women in childbearing age more frequently than in men, it was suggested a pathogenic role of sex hormones in this autoimmune disease. It is well known that SSc patients show an altered hormonal status. In particular, 17β-estradiol would exert a profibrotic effect on normal and SSc fibroblasts [111].
Decreased levels of serum dehydroepiandrosterone in males, as well as in females in childbearing age, have been associated with SSc [112]. Similarly, decreased serum levels of testosterone and increased levels of prolactin were observed only in female SSc patients [113]. Serum levels of prolactin were found to be significantly elevated in SSc female patients than in control subjects. Such a hyperprolactinemia correlated positively with aggressiveness of skin involvement [114]. Afterwards, other authors showed that lymphocytes from SSc patients were additional active sources of prolactin. In these patients, lymphocyte-derived prolactin would stimulate the same lymphocytes to increase the synthesis of interleukin-2 receptor [115].
X-Linked Single Nucleotide Polymorphisms
It is well established that genetic predisposition is an important factor for SSc development. Single nucleotide polymorphisms of genes located on the sex chromosomes can directly influence the SSc susceptibility. These X-linked genes may have a predominant role in sex bias of SSc. Moreover, many of these SNPs may characterize distinct clinical SSc phenotypes.
Significant association was found between the rs638376 of interleukin-13 receptor subunit α2 (IL13RA2) and SSc in a female Caucasian population [58]. These authors have shown also an association between the SNP rs5946040 of IL13RA2 and dcSSc subtype. It is known that interleukin-13 plays an important role in normal tissue repair, as well as in fibrosis [116,117], and its serum level was found increased in SSc patients compared to control healthy subjects [118].
Interleukin-1 receptor-associated kinase 1 (IRAK1), with its modulating activity on NF-KB, is known to be involved in TLR pathway [119]. An association between some IRAK1 SNPs and diffuse cutaneous SSc and anti topo-I positive SSc patients was found [59]. However, the frequencies of IRAK1 SNP rs1059702 have been found associated only to the susceptibility to SSc with lung fibrosis [120].
The X-linked methyl-CpG-binding protein 2 (MECP2) encodes a protein that binds specifically to methylated DNA, thus participating in the epigenetic mechanism, which may be responsible cofactors for the onset of autoimmune disorders [121,122]. The rs17345 of MEPC2 was found to be associated to diffuse cutaneous SSc subtype in females of Caucasian ancestry [120].
It is noteworthy that IRAK1 and MEPC2 activities are strictly interrelated; thus, a variation of one gene may influence the expression of the other one, and vice versa [123]. Furthermore, since these two genes are in high linkage disequilibrium, the real contribution of these genes to the susceptibility to SSc remains still controversial [59,123].
The transcription factor forkhead box P3 (FOXP3), a member of the forkhead/winged-helix family, is widely known for its role in the development and function of regulatory T cells, which play an important role in peripheral tolerance by suppressing immune response of self-reactive T cells [124]. Moreover, alterations in regulatory T cells number as well as FOXP3 expression have been shown in SSc [125–129] and its clinical subtypes [130].
Recently, we have shown that rs2280883 of FOXP3 gene could be associated to the susceptibility to SSc in a Caucasian female population, with a preferential association with ACA+ patients and with those suffering from lcSSc. Unfortunately, for the low number of recruited patients, we cannot rule out such a possibility for male patients [131].
Tissue inhibitor of metalloproteinases 1 (TIMP-1) belongs to the TIMP gene family. The main function of its encoded protein is to inhibit the matrix metalloproteinases, peptidases involved in degradation of the extracellular matrix [132]. Dysregulation of TIMP-1 has been found in serum and/or skin biopsies of SSc patients [62,133,134].
An association between the rs4898 of the TIMP1 gene with SSc as well as with SSc-associated digital ulcer formations has been shown in a female Italian Caucasian population [135]. As mentioned above, TIMP-1 is known to undergo a polymorphic inactivation in females, thus leading to a gene dosage skewing in females [83]. In agreement with polymorphic X chromosome inactivation, rs4898 of TIMP-1 has been shown to be accompanied with a lower TIMP-1 protein expression in males but not in females in inflammatory bowel disease [136].
Conclusions
SSc is a complex, heterogeneous, and multifactorial disease. Both genetic and epigenetic factors would contribute to the sex-specific difference in SSc incidence. As already mentioned, many sex- and immune-related genes, crucial for the maintenance of physiological levels of sex hormones, as well as of immune tolerance, are located on the X chromosome. Epigenetic X-linked abnormalities, such as skewing of X inactivation and/or gene reactivation, could play an important role in predisposing females to develop SSc (Fig. 2).
Interplay between genetic and epigenetic factors in the influence of susceptibility to SSc. Only X-linked factors are shown. Genetic backgrounds are source of predisposition to the disease. Epigenetic and environmental factors are needed for the trigger of SSc (SNPs single nucleotide polymorphisms; XCI X chromosome inactivation)
Thus, the advantage of having a stronger immunoresponse to infections in females reflects the possibility of having a higher susceptibility to SSc. Understanding the mechanisms which underlie X chromosome abnormalities would be very useful for developing therapeutic strategies for autoimmune disorders, including SSc. Moreover, because of reversible nature of epigenetic modifications, potential therapeutics treatments could be developed for reversing these modifications, which could be involved in the pathogenesis of SSc. For example, modulation of miRNAs could offer a new potential benefit for selective therapies.
Finally, in the development of therapeutic strategies differences in the immune response between the sexes must be considered because they may differently affect pharmacokinetics and -dynamics in a sex-dependent manner.
References
Agarwal SK, Tan FK, Arnett FC (2008) Genetics and genomic studies in scleroderma (systemic sclerosis). Rheum Dis Clin North Am 34:17–40
Amur S, Parekh A, Mummaneni P (2012) Sex differences and genomics in autoimmune diseases. J Autoimmun 38:J254–J265
Silver RM (1991) Clinical aspect of systemic sclerosis (scleroderma). Ann Rheum Dis 50:854–861
Hachulla E, Launay D (2011) Diagnosis and classification of systemic sclerosis. Clin Rev Allergy Immunol 40:78–83
Katsumoto TR, Whitfield ML, Connolly MK (2011) The pathogenesis of systemic sclerosis. Annu Rev Pathol Mech Dis 6:509–537
Geyer M, Müller-Ladner U (2011) The pathogenesis of systemic sclerosis revisited. Clin Rev Allergy Immunol 40:92–103
Kucharz EJ, Jarczyk R, Jonderko G, Rubisz-Brezezinska J, Brzezinska-Wcislo L (1996) High serum level of prolactin in patients with systemic sclerosis. Clin Rheumatol 15:314
Straub RH, Zeuner M, Lock G, Scholmerich J, Lang B (1997) High prolactin and low dehydroepiandrosterone sulphate serum levels in patients with severe systemic sclerosis. Br J Rheumatol 36:426–432
Nelson JL, Furst DE, Maloney S et al (1998) Microchimerism and HLA-compatible relationships of pregnancy in scleroderma. Lancet 351:559–562
Oliver JE, Silman AJ (2009) Why are women predisposed to autoimmune rheumatic diseases? Arthritis Res. Ther 11:252
Tiniakou E, Costenbader KH, Kriegel MA (2013) Sex-specific environmental influences on the development of autoimmune diseases. Clin Immunol. doi:10.1016/j.clim.2013.02.011
Ballestar E (2011) Epigenetic alterations in autoimmune rheumatic diseases. Nat Rev Rheumatol 7:263–271
Bianchi I, Lleo A, Gershwin ME, Invernizzi P (2102) The X chromosome and immune associated genes. J Autoimm 38:J187–J192
Chifflot H, Fautrel B, Sordet C, Chatelus E, Sibilia J (2008) Incidence and prevalence of systemic sclerosis: a systematic literature review. Semin Arthritis Rheum 37:223–235
Mayes MD, Lacey JV Jr, Beebe-Dimmer J et al (2003) Prevalence, incidence, survival, and disease characteristics of systemic sclerosis in a large US population. Arthritis Rheum 48:2246–2255
Le Guern V, Mahr A, Mouthon L, Jeanneret D, Carzon M, Guillevin L (2004) Prevalence of systemic sclerosis in a French multi-ethnic county. Rheumatology 43:1129–37
Alamanos Y, Tsifetaki N, Voulgari PV et al (2005) Epidemiology of systemic sclerosis in northwest Greece 1981 to 2002. Semin Arthritis Rheum 34:714–720
Roberts-Thomson PJ, Walker JG, Lu TY et al (2006) Scleroderma in South Australia: further epidemiological observations supporting a stochastic explanation. Intern Med J 36:489–497
Hunzelmann N, Genth E, Krieg T et al (2008) The registry of the German Network for Systemic Scleroderma: frequency of disease subsets and patterns of organ involvement. Rheumatology (Oxford) 47:1185–1192
Joven BE, Almodovar R, Carmona L, Carreira PE (2010) Survival, causes of death, and risk factors associated with mortality in Spanish systemic sclerosis patients: results from a single university hospital. Semin Arthritis Rheum 39:285–293
Lo Monaco A, Bruschi M, La Corte R, Volpinari S, Trotta F (2011) Epidemiology of systemic sclerosis in a district of northern Italy. Clin Exp Rheumatol 29:S10–14
Simeón-Aznar CP, Fonollosa-Plá V, Tolosa-Vilella C et al (2012) Registry of the Spanish network for systemic sclerosis: clinical pattern according to cutaneous subsets and immunological status. Semin Arthritis Rheum 41:789–800
Andréasson K, Saxne T, Bergknut C, Hesselstrand R, Englund M (2013) Prevalence and incidence of systemic sclerosis in southern Sweden: population-based data with case ascertainment using the 1980 ARA criteria and the proposed ACR-EULAR classification criteria. Ann Rheum Dis. doi:10.1136/annrheumdis-2013-203618
El Adssi H, Cirstea D, Virion JM, Guillemin F, de Korwin JD (2013) Estimating the prevalence of systemic sclerosis in the Lorraine region, France, by the capture–recapture method. Semin Arthritis Rheum 42:530–538
Gelber AC, Manno RL, Shah AA et al (2013) Race and association with disease manifestations and mortality in scleroderma: a 20-year experience at the Johns Hopkins Scleroderma Center and review of the literature. Medicine (Baltimore) 92:191–205
Hsiu Ling AL, Teng GG, Law WG (2013) Disease characteristics of the Singapore Systemic Sclerosis Cohort. Proc Singapore Healthcare 22:8–14
Razykov I, Levis B, Hudson M, Baron M, Thombs BD, on behalf of the Canadian Scleroderma Research Group (2013) Prevalence and clinical correlates of pruritus in patients with systemic sclerosis: an updated analysis of 959 patients. Rheumatology (Oxford). doi:10.1093/rheumatology/ket275
Subcommittee for Scleroderma Criteria of the American Rheumatism Association Diagnostic and Therapeutic Criteria Committee (1980) Preliminary criteria for the classification of systemic sclerosis (scleroderma). Arthritis Rheum 23:581–590
LeRoy EC, Black C, Fleischmajer R (1988) Scleroderma (systemic sclerosis): classification, subsets, and pathogenesis. J Rheumatol 15:202–205
LeRoy EC, Medsger TA Jr (2001) Criteria for the classification of early systemic sclerosis. J Rheumatol 28:1573–1576
Nietert PJ, Mitchell HC, Bolster MB et al (2006) Racial variation in clinical and immunological manifestations of systemic sclerosis. J Rheumatol 33:263–268
Steen VD, Oddis CV, Conte CG, Janoski J, Casterline GZ, Medsger TA Jr (1997) Incidence of systemic sclerosis in Allegheny County, Pennsylvania. A twenty-year study of hospital-diagnosed cases, 1963–1982. Arthritis Rheum 40:441–445
Barnes J, Mayes MD (2012) Epidemiology of systemic sclerosis: incidence, prevalence, survival, risk factors, malignancy, and environmental triggers. Curr Opin Rheumatol 24:165–170
Hoffmann-Vold AM, Molberg Ø, Midtvedt Ø, Garen T, Gran JT (2013) Survival and causes of death in an unselected and complete cohort of Norwegian patients with systemic sclerosis. J Rheumatol 40:1127–1133
Ross MT, Bentley DR, Tyler-Smith C (2006) The sequences of the human sex chromosomes. Curr Opin Genet Dev 16:213–218
Chow JC, Brown CJ (2003) Forming facultative heterochromatin: silencing of an X chromosome in mammalian females. Cell Mol Life Sci 60:2586–2603
Dupont C, Gribnau J (2013) Different flavors of X-chromosome inactivation in mammals. Curr Opin Cell Biol 25:314–321
Jeppesen P, Turner BM (1993) The inactive X chromosome in female mammals is distinguished by a lack of histone H4 acetylation, a cytogenetic marker for gene expression. Cell 74:281–289
Chang SC, Tucker T, Thorogood NP, Brown CJ (2006) Mechanisms of X-chromosome inactivation. Front Biosci 11:852–866
Augui S, Elphege PN, Heard E (2011) Regulation of X-chromosome inactivation by the X-inactivation centre. Nat Rev Genet 12:429–442
Pontier DB, Gribnau J (2011) Xist regulation and function explored. Hum Genet 130:223–236
Lyon MF (1998) X-chromosome inactivation: a repeat hypothesis. Cytogenet Cell Genet 80:133–137
Wutz A (2011) Gene silencing in X-chromosome inactivation: advances in understanding facultative heterochromatin formation. Nat Rev Genet 12:542–553
Chow JC, Yen Z, Ziesche SM, Brown CJ (2005) Silencing of the mammalian X chromosome. Annu Rev Genomics Hum Genet 6:69–92
Vallot C, Huret C, Lesecque Y et al (2013) XACT, a long noncoding transcript coating the active X chromosome in human pluripotent cells. Nat Genet 45:239–241
Heard E, Disteche CM (2006) Dosage compensation in mammals: fine-tuning the expression of the X chromosome. Genes Dev 20:1848–1867
Plath K, Fang J, Mlynarczyk-Evans SK et al (2003) Role of histone H3 lysine 27 methylation in X inactivation. Science 300:131–135
Libert C, Dejager L, Pinheiro I (2010) The X chromosome in immune functions: when a chromosome makes the difference. Nat Rev Immunol 10:594–604
Smith-Bouvier DL, Divekar AA, Sasidhar M et al (2008) A role for sex chromosome complement in the female bias in autoimmune disease. J Exp Med 205:1099–1108
Noguchi M, Yi H, Rosenblatt HM et al (1993) Interleukin-2 receptor gamma chain mutation results in X-linked severe combined immunodeficiency in humans. Cell 73:147–157
Gottipati S, Rao NL, Fung-Leung WP (2008) IRAK1: a critical signaling mediator of innate immunity. Cell Signal 20:269–276
Ohkura N, Kitagawa Y, Sakaguchi S (2013) Development and maintenance of regulatory T cells. Immunity 38:414–423
Vadasz Z, Haj T, Kessel A, Toubim E (2013) B-regulatory cells in autoimmunity and immune mediated inflammation. FEBS Lett 587:2074–2078
Takahashi Y, Haga S, Ishizaka Y, Mimori A (2010) Autoantibodies to angiotensin-converting enzyme 2 in patients with connective tissue diseases. Arthritis Res Ther 12:R85
Rabquer BJ, Hou Y, Del Galdo F et al (2009) The proadhesive phenotype of systemic sclerosis skin promotes myeloid cell adhesion via ICAM-1 and VCAM-1. Rheumatology (Oxford) 48:734–740
Rabquer BJ, Tsou PS, Hou Y et al (2011) Dysregulated expression of MIG/CXCL9, IP-10/CXCL10 and CXCL16 and their receptors in systemic sclerosis. Arthritis Res Ther 13:R18
Liu X, Gao N, Li M et al (2013) Elevated levels of CD4+CD25+FoxP3+ T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction. PLoS One 8:e64531
Granel B, Allanore Y, Chevillard C et al (2006) IL13RA2 gene polymorphisms are associated with systemic sclerosis. J Rheumatol 33:2015–2019
Dieudé P, Bouaziz M, Guedj M et al (2011) Evidence of the contribution of the X chromosome to systemic sclerosis susceptibility: association with the functional IRAK1 196Phe/532Ser haplotype. Arthritis Rheum 63:3979–3987
Lei W, Luo Y, Lei W et al (2009) Abnormal DNA methylation in CD4+ T cells from patients with systemic lupus erythematosus, systemic sclerosis, and dermatomyositis. Scand J Rheumatol 38:369–374
Selmi C, Feghali-Bostwick CA, LLeo A et al (2012) X chromosome gene methylation in peripheral lymphocytes from monozygotic twins discordant for scleroderma. Clin Exp Immunol 169:253–262
Young-Min SA, Beeton C, Laughton R et al (2001) Serum TIMP-1, TIMP-2, and MMP-1 in patients with systemic sclerosis, primary Raynaud's phenomenon, and in normal controls. Ann Rheum Dis 60:846–851
van Bon L, Popa C, Huijbens R et al (2010) Distinct evolution of TLR-mediated dendritic cell cytokine secretion in patients with limited and diffuse cutaneous systemic sclerosis. Ann Rheum Dis 69:1539–1547
Ciechomska M, Huigens CA, Hügle T et al (2013) Toll-like receptor-mediated, enhanced production of profibrotic TIMP-1 in monocytes from patients with systemic sclerosis: role of serum factors. Ann Rheum Dis 72:1382–1389
Hong B, Reeves P, Panning B, Swanson MS, Yang TP (2001) Identification of an autoimmune serum containing antibodies against the Barr body. Proc Natl Acad Sci U S A 98:8703–8708
Payer B, Lee JT, Namekawa SH (2011) X-inactivation and X-reactivation: epigenetic hallmarks of mammalian reproduction and pluripotent stem cells. Hum Genet 130:265–280
Pessach IM, Notarangelo LD (2009) X-linked primary immunodeficiencies as a bridge to better understanding X-chromosome related autoimmunity. J Autoimmun 33:17–24
Lambert NC (2009) The price of silence. Arthritis Rheum 60:3164–3167
Carrel L, Willard HF (2005) X-inactivation profile reveals extensive variability in X-linked gene expression in females. Nature 434:400–404
Bolduc V, Chagnon P, Provost S et al (2008) No evidence that skewing of X chromosome inactivation patterns is transmitted to offspring in humans. J Clin Invest 118:333–341
Kast RE (1977) Predominance of autoimmune and rheumatic diseases in females. J Rheumatol 4:288–292
Stewart JJ (1998) The female X-inactivation mosaic in systemic lupus erythematosus. Immunol Today 19:352–357
Ozcelik T (2008) X chromosome inactivation and female predisposition to autoimmunity. Clin Rev Allergy Immunol 34:348–351
Lu Q, Wu A, Tesmer L, Ray D, Yousif N, Richardson B (2007) Demethylation of CD40LG on the inactive X in T cells from women with lupus. J Immunol 179:6352–6358
Lian X, Xiao R, Hu X et al (2012) DNA demethylation of CD40L in CD4+ T cells from women with systemic sclerosis: a possible explanation for female susceptibility. Arthritis Rheum 64:2338–2345
Invernizzi P, Miozzo M, Selmi C et al (2005) X chromosome monosomy: a common mechanism for autoimmune diseases. J Immunol 175:575–578
Ozbalkan Z, Bagislar S, Kiraz S (2005) Skewed X chromosome inactivation in blood cells of women with scleroderma. Arthritis Rheum 52:1564–1570
Broen JC, Wolvers-Tettero IL, Bon G-v et al (2010) Skewed X chromosomal inactivation impacts T regulatory cell function in systemic sclerosis. Ann Rheum Dis 69:2213–2216
Fish EN (2008) The X-files in immunity: sex-based differences predispose immune responses. Nat Rev Immunol 8:737–744
Agrelo R, Souabni A, Novatchkova M et al (2009) SATB1 defines the developmental context for gene silencing by Xist in lymphoma and embryonic cells. Dev Cell 16:507–516
Reinius B, Shi C, Hengshuo L et al (2010) Female-biased expression of long non-coding RNAs in domains that escape X-inactivation in mouse. BMC Genomics 11:614
Filippova GN (2008) Genetics and epigenetics of the multifunctional protein CTCF. Curr Top Dev Biol 80:337–360
Anderson CL, Brown CJ (1999) Polymorphic X-chromosome inactivation of the human TIMP1 gene. Am J Hum Genet 65:699–708
Artlett CM, Sassi-Gaha S, Rieger JL, Boesteanu AC, Feghali-Bostwick CA, Katsikis PD (2011) The inflammasome activating caspase 1 mediates fibrosis and myofibroblast differentiation in systemic sclerosis. Arthritis Rheum 63:3563–3574
Disteche CM (2012) Dosage compensation of the sex chromosomes. Annu Rev Genet 46:537–560
Invernizzi P, Pasini S, Selmi C, Gershwin ME, Podda M (2009) Female predominance and X chromosome defects in autoimmune diseases. J Autoimmun 33:12–16
Hajkova P, Erhardt S, Lane N et al (2002) Epigenetic reprogramming in mouse primordial germ cells. Mech Dev 117:15–23
Ohhata T, Wutz A (2013) Reactivation of the inactive X chromosome in development and reprogramming. Cell Mol Life Sci 70:2443–2461
Richardson B (2003) DNA methylation and autoimmune disease. Clin Immunol 109:72–79
Fukasawa C, Kawaguchi Y, Harigai M et al (2003) Increased CD40 expression in skin fibroblasts from patients with systemic sclerosis (SSc): role of CD40-CD154 in the phenotype of SSc fibroblasts. Eur J Immunol 33:2792–2800
Kawai M, Masuda A, Kuwana M (2008) A CD40–CD154 interaction in tissue fibrosis. Arthritis Rheum 58:3562–3573
Valentini G, Romano MF, Naclerio C et al (2000) Increased expression of CD40 ligand in activated CD4_ T lymphocytes of systemic sclerosis patients. J Autoimmun 15:61–66
Wang Y, Fan PS, Kahaleh B (2006) Association between enhanced type I collagen expression and epigenetic repression of the FLI1 gene in scleroderma fibroblasts. Arthritis Rheum 54:2271–2279
Karaca NE, Aksu G, Karaca E et al (2011) Progressive morphea of early childhood tracing Blaschko’s lines on the face: involvement of X chromosome monosomy in pathogenesis and clinical prognosis. Int J Dermatol 50:1406–1410
Wang KC, Chang HY (2011) Molecular mechanisms of long noncoding RNAs. Mol Cell 16:904–914
Kosik KS (2013) Circles reshape the RNA world. Nature 495:322–324
Guo XJ, Su B, Zhou ZM, Sha JH (2009) Rapid evolution of mammalian X-linked testis microRNAs. BMC Genomics 10:97
Barrett LW, Fletcher S, Wilton SD (2012) Regulation of eukaryotic gene expression by the untranslated gene regions and other non-coding elements. Cell Mol Life Sci 69:3613–3634
Pinheiro I, Dejager L, Libert C (2011) X-chromosome-located microRNAs in immunity: might they explain male/female differences? Bioessays 33:791–802
Zhu H, Li Y, Qu S (2012) MicroRNA expression abnormalities in limited cutaneous scleroderma and diffuse cutaneous scleroderma. J Clin Immunol 32:514–522
Honda N, Jinnin M, Kira-Etoh T et al (2013) miR-150 down-regulation contributes to the constitutive type I collagen overexpression in scleroderma dermal fibroblasts via the induction of integrin β3. Am J Pathol 182:206–216
Makino K, Jinnin M, Hirano A et al (2013) The downregulation of microRNA let-7a contributes to the excessive expression of type I collagen in systemic and localized scleroderma. J Immunol 190:3905–3915
Whitacre CC, Reingold SC, O’Looney PA (1999) A gender gap in autoimmunity. Science 283:1277–1278
Ober C, Loisel DA, Gilad Y (2008) Sex-specific genetic architecture of human disease. Nat Rev Genet 9:911–922
Gleicher N, Barad DH (2007) Gender as risk factor for autoimmune diseases. J Autoimmun 28:1–6
Palaszynski KM, Smith DL, Kamrava S, Burgoyne PS, Arnold AP, Voskuhl RR (2005) A yin-yang effect between sex chromosome complement and sex hormones on the immune response. Endocrinology 146:3280–3285
Medina KL, Garrett KP, Thompson LF, Rossi MI, Payne KJ, Kincade PW (2001) Identification of very early lymphoid precursors in bone marrow and their regulation by estrogen. Nat Immunol 2:718–724
Grimaldi CM, Cleary J, Dagtas AS, Moussai D, Diamond B (2002) Estrogen alters thresholds for B cell apoptosis and activation. J Clin Invest 109:1625–1633
Nalbandian G, Kovats S (2005) Understanding sex biases in immunity: effects of estrogen on the differentiation and function of antigen-presenting cells. Immunol Res 31:91–106
Pennell LM, Galligan CL, Fish EN (2011) Sex affects immunity. J Autoimmun 38:J282–J291
Soldano S, Montagna P, Villaggio B et al (2009) Endothelin and sex hormones modulate the fibronectin synthesis by cultured human skin scleroderma fibroblasts. Ann Rheum Dis 68:599–602
Hilty C, Brühlmann P, Sprott H et al (2000) Altered diurnal rhythm of prolactin in systemic sclerosis. J Rheumatol 27:2160–2165
Mirone L, Barini A, Barini A (2006) Androgen and prolactin (Prl) levels in systemic sclerosis (SSc): relationship to disease severity. Ann N Y Acad Sci 1069:257–262
Shahin AA, Abdoh S, Abdelrazik M (2002) Prolactin and thyroid hormones in patients with systemic sclerosis: correlations with disease manifestations and activity. Z Rheumatol 61:703–709
Czuwara-Ladykowska J, Sicinska J, Olszewska M, Uhrynowska-Tyszkiewicz I, Rudnicka L (2006) Prolactin synthesis by lymphocytes from patients with systemic sclerosis. Biomed Pharmacother 60:152–155
Kissin EY, Korn JH (2003) Fibrosis in scleroderma. Rheum Dis Clin North Am 29:351–369
Fuschiotti P (2011) Role of IL-13 in systemic sclerosis. Cytokine 56:544–549
Riccieri V, Rinaldi T, Spadaro A et al (2003) Interleukin-13 in systemic sclerosis: relationship to nailfold capillaroscopy abnormalities. Clin Rheumatol 22:102–106
Rao N, Nguyen S, Ngo K, Fung-Leung WP (2005) A novel splice variant of interleukin-1 receptor (IL-1R)-associated kinase 1 plays a negative regulatory role in Toll/IL-1R-induced inflammatory signaling. Mol Cell Biol 25:6521–6532
Carmona, F. D., Cénit, M. C., Diaz-Gallo, L. M. et al. (2013) New insight on the Xq28 association with systemic sclerosis. Ann. Rheum. Dis. 2013 Feb 26
Brooks WH, Le Dantec C, Pers JO, Youinou P, Renaudineau Y (2010) Epigenetics and autoimmunity. J Autoimmun 34:J207–J219
Adkins NL, Georgel PT (2011) MeCP2: structure and function. Biochem Cell Biol 89:1–11
Urdinguio RG, Fernandez AF, Lopez-Nieva P et al (2010) Disrupted microRNA expression caused by Mecp2 loss in a mouse model of Rett syndrome. Epigenetics 5:656–563
Sakaguchi S, Sakaguchi N, Shimizu J (2001) Immunologic tolerance maintained by CD25+ CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol Rev 182:18–32
Radstake TR, van Bon L, Broen J et al (2009) Increased frequency and compromised function of T regulatory cells in systemic sclerosis (SSc) is related to a diminished CD69 and TGFβ expression. PLoS ONE 4:e5981
Antiga E, Quaglino P, Bellandi S et al (2010) Regulatory T cells in the skin lesions and blood of patients with systemic sclerosis and morphoea. Br J Dermatol 162:1056–1063
Fenoglio D, Battaglia F, Parodi A et al (2011) Alteration of Th17 and Treg cell subpopulations coexist in patients affected with systemic sclerosis. Clin Immunol 139:249–257
Klein S, Kretz CC, Ruland V et al (2011) Reduction of regulatory T cells in skin lesions but not in peripheral blood of patients with systemic scleroderma. Ann Rheum Dis 70:1475–1481
Furuzawa-Carballeda J, Ortíz-Ávalos M, Lima G, Jurado-Santa Cruz F, Llorente L (2012) Subcutaneous administration of polymerized type I collagen downregulates interleukin (IL)-17A, IL-22 and transforming growth factor-β1 expression, and increases Foxp3-expressing cells in localized scleroderma. Clin Exp Dermatol 37:599–609
Slobodin G, Ahmad MS, Rosner I et al (2010) Regulatory T cells (CD4+CD25brighFoxP3+) expansion in systemic sclerosis correlates with disease activity and severity. Cell Immunol 261:77–80
D'Amico F, Skarmoutsou E, Marchini M et al (2013) Genetic polymorphisms of FOXP3 in Italian patients with systemic sclerosis. Immunol Lett 152:109–113
Murphy G (2011) Tissue inhibitors of metalloproteinases. Genome Biol 12:233
Toubi E, Kessel A, Grushko G, Sabo E, Rozenbaum M, Rosner I (2002) The association of serum matrix metalloproteinases and their tissue inhibitor levels with scleroderma disease severity. Clin Exp Rheumatol 20:221–224
Frost J, Ramsay M, Mia R, Moosa L, Musenge E, Tikly M (2012) Differential gene expression of MMP-1, TIMP-1 and HGF in clinically involved and uninvolved skin in South Africans with SSc. Rheumatology (Oxford) 51:1049–1052
Skarmoutsou E, D'Amico F, Marchini M, Malaponte G, Scorza R, Mazzarino MC (2012) Association of TIMP-1 +372 SNP with digital ulcer manifestation in female systemic sclerosis patients. Hum Immunol 73:950–953
Meijer MJ, Mieremet-Ooms MA, van Hogezand RA, Lamers CB, Hommes DW, Verspaget HW (2007) Role of matrix metalloproteinase, tissue inhibitor of metalloproteinase and tumor necrosis factor-α single nucleotide gene polymorphisms in inflammatory bowel disease. World J Gastroenterol 13:2960–2966
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
D’Amico, F., Skarmoutsou, E. & Mazzarino, M.C. The Sex Bias in Systemic Sclerosis: on the Possible Mechanisms Underlying the Female Disease Preponderance. Clinic Rev Allerg Immunol 47, 334–343 (2014). https://doi.org/10.1007/s12016-013-8392-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-013-8392-9