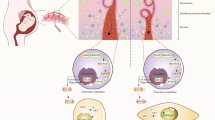
Abstract
Crohn’s disease (CD) with externally fistulizing openings indicates the aggressive and relapsing manifestation and results in undesirable long-term outcomes of patients. MSC-based approach combined with multidisciplinary strategy has mandated a redefinition of the administration and management of numerous recurrent and refractory diseases whereas the spatio-temporal evaluation of the metabolokinetics and efficacy of MSCs on intractable CD with enterocutaneous fistula (EF) are largely inaccessible and dauntingly complex. Herein, we primitively established dual-fluorescence expressing placenta-derived MSCs (DF-MSCs) and explored their multidimensional attributes, including cytomorphology, immunophenotying, multilineage differentiation and long-term proliferation, together with the recognition of bifluorescence intensity (BLI). Then, with the aid of in vivo living imaging, clinicopathological or inflammatory cytokine examinations and in vitro analyses, we systematically and meticulously dissected the metabolokinetics and curative effect of MSCs on mice with refractory Crohn’s-like EF (EF mice), together with revealing the underlying mechanism including reactive oxygen species (ROS) and neovascularization. Strikingly, the DF-MSCs exhibited stabilized BLI and biological properties. The spatio-temporal distribution and therapeutic process of MSCs in EF mice were intuitively delineated. Meanwhile, our data indicated the curative mechanisms of DF-MSCs by simultaneously downregulating ROS and accelerating neovascularization. Collectively, we systematically illuminated the spatio-temporal biofunction and mechanism of DF-MSCs on EF mice. Our findings have supplied new references for safety and effectiveness assessments as well as the establishment of guidelines for optimal administrations of MSC-based cytotherapy in preclinical studies, which collectively indicates the prospect of P-MSC administration in clinical trials during a wide spectrum of disease remodeling including the fistulizing CD.

Graphical abstract






Similar content being viewed by others
References
Aguirre, A., & Nugent, C. A. (2015). Images in clinical medicine: oral manifestation of Crohn’s disease. The New England Journal of Medicine, 373(13), 1250. https://doi.org/10.1056/NEJMicm1413715.
Alicka, M., Major, P., Wysocki, M., & Marycz, K. (2019). Adipose-derived mesenchymal stem cells isolated from patients with type 2 diabetes show reduced “stemness” through an altered secretome profile, impaired anti-oxidative protection, and mitochondrial dynamics deterioration. Journal of Clinical Medicine, 8(6). https://doi.org/10.3390/jcm8060765.
Baksh, D., Yao, R., & Tuan, R. S. (2007). Comparison of proliferative and multilineage differentiation potential of human mesenchymal stem cells derived from umbilical cord and bone marrow. Stem Cells, 25(6), 1384–1392. https://doi.org/10.1634/stemcells.2006-0709.
Cao, H., Yue, Z., Gao, H., Chen, C., Cui, K., Zhang, K., Cheng, Y., Shao, G., Kong, D., Li, Z., Ding, D., & Wang, Y. (2019). In vivo real-time imaging of extracellular vesicles in liver regeneration via aggregation-induced emission luminogens. ACS Nano, 13(3), 3522–3533. https://doi.org/10.1021/acsnano.8b09776.
Cho, Y. B., Park, K. J., Yoon, S. N., Song, K. H., Kim, D. S., Jung, S. H., Kim, M., Jeong, H. Y., & Yu, C. S. (2015). Long-term results of adipose-derived stem cell therapy for the treatment of Crohn’s fistula. Stem Cells Translational Medicine, 4(5), 532–537. https://doi.org/10.5966/sctm.2014-0199.
Ciccocioppo, R., Gallia, A., Sgarella, A., Kruzliak, P., Gobbi, P. G., & Corazza, G. R. (2015). Long-term follow-up of Crohn disease fistulas after local injections of bone marrow-derived mesenchymal stem cells. Mayo Clinic Proceedings, 90(6), 747–755. https://doi.org/10.1016/j.mayocp.2015.03.023.
Dalal, J., Gandy, K., & Domen, J. (2012). Role of mesenchymal stem cell therapy in Crohn’s disease. Pediatric Research, 71(4 Pt 2), 445–451. https://doi.org/10.1038/pr.2011.56.
de la Guardia, R. D., Lopez-Millan, B., Roca-Ho, H., Bueno, C., Gutierrez-Aguera, F., Fuster, J. L., et al. (2019). Bone marrow mesenchymal stem/stromal cells from risk-stratified acute myeloid leukemia patients are anti-inflammatory in in vivo preclinical models of hematopoietic reconstitution and severe colitis. Haematologica, 104(2), e54–e58. https://doi.org/10.3324/haematol.2018.196568.
Du, W., Li, X., Chi, Y., Ma, F., Li, Z., Yang, S., et al. (2016). VCAM-1+ placenta chorionic villi-derived mesenchymal stem cells display potent pro-angiogenic activity. Stem Cell Research & Therapy, 7, 49. https://doi.org/10.1186/s13287-016-0297-0.
El Moshy, S., Radwan, I. A., Rady, D., Abbass, M. M. S., El-Rashidy, A. A., Sadek, K. M., et al. (2020). Dental stem cell-derived secretome/conditioned medium: the future for regenerative therapeutic applications. Stem Cells International, 2020, 7593402–7593429. https://doi.org/10.1155/2020/7593402.
Garrido-Trigo, A., & Salas, A. (2019). Molecular structure and function of Janus kinases: implications for the development of inhibitors. Journal of Crohn’s & Colitis, 14, S713–S724. https://doi.org/10.1093/ecco-jcc/jjz206.
Huo, J., Zhang, L., Ren, X., Li, C., Li, X., Dong, P., Zheng, X., Huang, J., Shao, Y., Ge, M., Zhang, J., Wang, M., Nie, N., Jin, P., & Zheng, Y. (2020). Multifaceted characterization of the signatures and efficacy of mesenchymal stem/stromal cells in acquired aplastic anemia. Stem Cell Research & Therapy, 11(1), 59. https://doi.org/10.1186/s13287-020-1577-2.
Kfoury, Y., & Scadden, D. T. (2015). Mesenchymal cell contributions to the stem cell niche. Cell Stem Cell, 16(3), 239–253. https://doi.org/10.1016/j.stem.2015.02.019.
Klionsky, D. J. (2009). Crohn’s disease, autophagy, and the Paneth cell. The New England Journal of Medicine, 360(17), 1785–1786. https://doi.org/10.1056/NEJMcibr0810347.
Lai, P., Chen, X., Guo, L., Wang, Y., Liu, X., Liu, Y., Zhou, T., Huang, T., Geng, S., Luo, C., Huang, X., Wu, S., Ling, W., du, X., He, C., & Weng, J. (2018). A potent immunomodulatory role of exosomes derived from mesenchymal stromal cells in preventing cGVHD. Journal of Hematology & Oncology, 11(1), 135. https://doi.org/10.1186/s13045-018-0680-7.
Lim, M., Wang, W., Liang, L., Han, Z. B., Li, Z., Geng, J., Zhao, M., Jia, H., Feng, J., Wei, Z., Song, B., Zhang, J., Li, J., Liu, T., Wang, F., Li, T., Li, J., Fang, Y., Gao, J., & Han, Z. (2018). Intravenous injection of allogeneic umbilical cord-derived multipotent mesenchymal stromal cells reduces the infarct area and ameliorates cardiac function in a porcine model of acute myocardial infarction. Stem Cell Research & Therapy, 9(1), 129. https://doi.org/10.1186/s13287-018-0888-z.
Murphy, M. B., Moncivais, K., & Caplan, A. I. (2013). Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Experimental & Molecular Medicine, 45, e54. https://doi.org/10.1038/emm.2013.94.
Neurath, M. F. (2017). Current and emerging therapeutic targets for IBD. Nature Reviews. Gastroenterology & Hepatology, 14(5), 269–278. https://doi.org/10.1038/nrgastro.2016.208.
Nombela-Arrieta, C., Ritz, J., & Silberstein, L. E. (2011). The elusive nature and function of mesenchymal stem cells. Nature Reviews. Molecular Cell Biology, 12(2), 126–131. https://doi.org/10.1038/nrm3049.
Panes, J., Garcia-Olmo, D., Van Assche, G., Colombel, J. F., Reinisch, W., Baumgart, D. C., et al. (2016). Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet, 388(10051), 1281–1290. https://doi.org/10.1016/S0140-6736(16)31203-X.
Ryan, P. L., Youngblood, R. C., Harvill, J., & Willard, S. T. (2005). Photonic monitoring in real time of vascular endothelial growth factor receptor 2 gene expression under relaxin-induced conditions in a novel murine wound model. Annals of the New York Academy of Sciences, 1041, 398–414. https://doi.org/10.1196/annals.1282.061.
Samsonraj, R. M., Raghunath, M., Nurcombe, V., Hui, J. H., van Wijnen, A. J., & Cool, S. M. (2017). Concise review: multifaceted characterization of human mesenchymal stem cells for use in regenerative medicine. Stem Cells Translational Medicine, 6(12), 2173–2185. https://doi.org/10.1002/sctm.17-0129.
Tao, H., Chen, X., Cao, H., Zheng, L., Li, Q., Zhang, K., Han, Z., Han, Z. C., Guo, Z., Li, Z., & Wang, L. (2019). Mesenchymal stem cell-derived extracellular vesicles for corneal wound repair. Stem Cells International, 2019, 5738510–5738519. https://doi.org/10.1155/2019/5738510.
Wang, L., Liu, T., Liang, R., Wang, G., Liu, Y., Zou, J., Liu, N., Zhang, B., Liu, Y., Ding, X., Cai, X., Wang, Z., Xu, X., Ricordi, C., Wang, S., & Shen, Z. (2020). Mesenchymal stem cells ameliorate beta cell dysfunction of human type 2 diabetic islets by reversing beta cell dedifferentiation. EBioMedicine, 51, 102615. https://doi.org/10.1016/j.ebiom.2019.102615.
Wei, Y., Hou, H., Zhang, L., Zhao, N., Li, C., Huo, J., Liu, Y., Zhang, W., Li, Z., Liu, D., Han, Z., Zhang, L., Song, B., Chi, Y., & Han, Z. (2019a). JNKi- and DAC-programmed mesenchymal stem/stromal cells from hESCs facilitate hematopoiesis and alleviate hind limb ischemia. Stem Cell Research & Therapy, 10(1), 186. https://doi.org/10.1186/s13287-019-1302-1.
Wei, Y., Wu, Y., Zhao, R., Zhang, K., Midgley, A. C., Kong, D., Li, Z., & Zhao, Q. (2019b). MSC-derived sEVs enhance patency and inhibit calcification of synthetic vascular grafts by immunomodulation in a rat model of hyperlipidemia. Biomaterials, 204, 13–24. https://doi.org/10.1016/j.biomaterials.2019.01.049.
Wei, Y., Zhang, L., Chi, Y., Ren, X., Gao, Y., Song, B., et al. (2020). High-efficient generation of VCAM-1(+) mesenchymal stem cells with multidimensional superiorities in signatures and efficacy on aplastic anaemia mice. Cell Proliferation, e12862. https://doi.org/10.1111/cpr.12862.
Wu, Q., Zhang, L., Su, P., Lei, X., Liu, X., Wang, H., Lu, L., Bai, Y., Xiong, T., Li, D., Zhu, Z., Duan, E., Jiang, E., Feng, S., Han, M., Xu, Y., Wang, F., & Zhou, J. (2015). MSX2 mediates entry of human pluripotent stem cells into mesendoderm by simultaneously suppressing SOX2 and activating NODAL signaling. Cell Research, 25(12), 1314–1332. https://doi.org/10.1038/cr.2015.118.
Yamamoto, N., Oyaizu, T., Enomoto, M., Horie, M., Yuasa, M., Okawa, A., & Yagishita, K. (2020). VEGF and bFGF induction by nitric oxide is associated with hyperbaric oxygen-induced angiogenesis and muscle regeneration. Scientific Reports, 10(1), 2744. https://doi.org/10.1038/s41598-020-59615-x.
Zhang, X., Yang, Y., Zhang, L., Lu, Y., Zhang, Q., Fan, D., Zhang, Y., Zhang, Y., Ye, Z., & Xiong, D. (2017). Mesenchymal stromal cells as vehicles of tetravalent bispecific Tandab (CD3/CD19) for the treatment of B cell lymphoma combined with IDO pathway inhibitor D-1-methyl-tryptophan. Journal of Hematology & Oncology, 10(1), 56. https://doi.org/10.1186/s13045-017-0397-z.
Zhang, K., Zhao, X., Chen, X., Wei, Y., Du, W., Wang, Y., et al. (2018a). Enhanced therapeutic effects of mesenchymal stem cell-derived exosomes with an injectable hydrogel for Hindlimb ischemia treatment. ACS Applied Materials & Interfaces, 10(36), 30081–30091. https://doi.org/10.1021/acsami.8b08449.
Zhang, L., Liu, C., Wang, H., Wu, D., Su, P., Wang, M., Guo, J., Zhao, S., Dong, S., Zhou, W., Arakaki, C., Zhang, X., & Zhou, J. (2018b). Thrombopoietin knock-in augments platelet generation from human embryonic stem cells. Stem Cell Research & Therapy, 9(1), 194. https://doi.org/10.1186/s13287-018-0926-x.
Zhang, L., Wang, H., Liu, C., Wu, Q., Su, P., Wu, D., Guo, J., Zhou, W., Xu, Y., Shi, L., & Zhou, J. (2018c). MSX2 initiates and accelerates mesenchymal stem/stromal cell specification of hPSCs by regulating TWIST1 and PRAME. Stem Cell Reports, 11(2), 497–513. https://doi.org/10.1016/j.stemcr.2018.06.019.
Zhang, S., Liu, Y., Zhang, X., Zhu, D., Qi, X., Cao, X., Fang, Y., Che, Y., Han, Z. C., He, Z. X., Han, Z., & Li, Z. (2018d). Prostaglandin E2 hydrogel improves cutaneous wound healing via M2 macrophages polarization. Theranostics, 8(19), 5348–5361. https://doi.org/10.7150/thno.27385.
Zhao, Q., Zhang, L., Wei, Y., Yu, H., Zou, L., Huo, J., Yang, H., Song, B., Wei, T., Wu, D., Zhang, W., Zhang, L., Liu, D., Li, Z., Chi, Y., Han, Z., & Han, Z. (2019). Systematic comparison of hUC-MSCs at various passages reveals the variations of signatures and therapeutic effect on acute graft-versus-host disease. Stem Cell Research & Therapy, 10(1), 354. https://doi.org/10.1186/s13287-019-1478-4.
Acknowledgements
We thank Ying Chi, Yimeng Wei, Yi Yu, Zhibo Han, Zhihai Han, Dengke Liu, Rucai Zhan and Baiqing Dong for technical support or constructive suggestions. Also, we appreciate Gang Liu and Yanyan Xu in Tianjin Medical University for their kind help and technical support during the construction of mice model. The authors would like to thank the enterprise postdoctoral working station of Tianjin Chase Sun Pharmaceutical Co., Ltd. and Precision Medicine Division of Health-Biotech (Tianjin) Stem Cell Research Institute Co., Ltd., and Postdoctoral workstation of Wuqing development Zone in Tianjin city for their kind support.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81770119), Project funded by China Postdoctoral Science Foundation (2019 M661033), Natural Science Foundation of Tianjin (19JCQNJC12500), Natural Science Foundation of Hebei (H2020206403), Natural Science Foundation of Shandong (2020, to LSZ), Science and Technology Project of Tianjin (17ZXSCSY00030), Nanyang Science and Technology Project of He-nan Province (JCQY012), Emergency project funded by Department of Science and Technology of Jiangxi Province (2020, to ZCH), Key project funded by Department of Science and Technology of Shangrao City (2020, to ZCH).
Author information
Authors and Affiliations
Contributions
H.H.: performed the experiments, collection and assembly of data; L.D., Y.L. and Z.H.: helped with collection and assembly of data; H.H., L.Z.: helped with the mice experiments; L.Z., Z.L. and X.C.: data analysis and interpretation, manuscript writing; L.Z. and X.C.: conception and design, final approval and revision of manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interests.
Ethics Approval
This experiment was approved by the Ethical Committee of Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Science and Peking Union Medical College (approval no. KT2019048-EC-1, KT2016011-EC-1) and in accordance with the Standard Operation Procedures (SOP) of the same facility.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Hou, H., Zhang, L., Duan, L. et al. Spatio-Temporal Metabolokinetics and Efficacy of Human Placenta-Derived Mesenchymal Stem/Stromal Cells on Mice with Refractory Crohn’s-like Enterocutaneous Fistula. Stem Cell Rev and Rep 16, 1292–1304 (2020). https://doi.org/10.1007/s12015-020-10053-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-020-10053-2