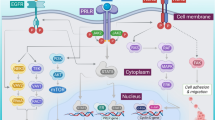
Abstract
Doxorubicin is an effective chemotherapeutic agent prescribed to treat solid tumors (e.g., ovary, breast, and gastrointestinal cancers). This anti-cancer drug has various side effects, such as allergic reactions, cardiac damage, hair loss, bone marrow suppression, vomiting, and bladder irritation. The most dangerous side effect of doxorubicin is cardiomyopathy, leading to congestive heart failure. The exact mechanisms of doxorubicin-induced cardiotoxicity remain incompletely understood. Alteration in myocardial structure and functional cardiac disorders is provoked by doxorubicin administration; subsequently, cardiomyopathy and congestive heart failure can occur. Congestive heart failure due to doxorubicin is associated with mortality and morbidity. Probably, doxorubicin-induced cardiotoxicity starts from myocardial cell injury and is followed by left ventricular dysfunction. Many factors and multiple pathways are responsible for the creation of doxorubicin-induced cardiotoxicity. Inflammatory cytokines, oxidative stress pathways, mitochondrial damage, intracellular Ca2+ overload, iron-free radical production, DNA, and myocyte membrane injuries have critical roles in the pathophysiology of doxorubicin-induced cardiotoxicity. Unfortunately, there are currently a few medications for the treatment of doxorubicin-induced cardiotoxicity in clinical settings. Extensive basic and clinical researches have been carried out to discover preventive treatments. This review briefly discusses the basic and experimental approaches for treating or preventing doxorubicin-mediated cardiotoxicity based on its pathophysiological mechanisms.



Similar content being viewed by others
Data Availability
Available upon request.
Code Availability
Not applicable.
References
Najafi, M., Shayesteh, M. R. H., Mortezaee, K., Farhood, B., & Haghi-Aminjan, H. (2020). The role of melatonin on doxorubicin-induced cardiotoxicity: A systematic review. Life Sciences, 241, 117173.
Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 71, 209–249.
Young, R. C., Ozols, R. F., & Myers, C. E. (1981). The anthracycline antineoplastic drugs. New England Journal of Medicine, 305, 139–153.
Narkiewicz, K., Ratcliffe, L. E., Hart, E. C., Briant, L. J., Chrostowska, M., Wolf, J., Szyndler, A., Hering, D., Abdala, A. P., & Manghat, N. (2016). Unilateral carotid body resection in resistant hypertension: A safety and feasibility trial. JACC: Basic to Translational Science, 1, 313–324.
Hardaway, B. W. (2019). Adriamycin-associated cardiomyopathy: Where are we now? Updates in pathophysiology, dose recommendations, prognosis, and outcomes. Current Opinion in Cardiology, 34, 289–295.
Arcamone, F., Cassinelli, G., Fantini, G., Grein, A., Orezzi, P., Pol, C., & Spalla, C. (1969). Adriamycin, 14-hydroxydaimomycin, a new antitumor antibiotic from S. Peucetius var. caesius. Biotechnology and Bioengineering, 11, 1101–1110.
Kalyanaraman, B. (2020). Teaching the basics of the mechanism of doxorubicin-induced cardiotoxicity: Have we been barking up the wrong tree? Redox Biology, 29, 101394.
Yu, J., Wang, C., Kong, Q., Wu, X., Lu, J.-J., & Chen, X. (2018). Recent progress in doxorubicin-induced cardiotoxicity and protective potential of natural products. Phytomedicine, 40, 125–139.
Wenningmann, N., Knapp, M., Ande, A., Vaidya, T. R., & Ait-Oudhia, S. (2019). Insights into doxorubicin-induced cardiotoxicity: Molecular mechanisms, preventive strategies, and early monitoring. Molecular Pharmacology, 96, 219–232.
Speth, P. A., van Hoesel, Q. G., & Haanen, C. (1988). Clinical pharmacokinetics of doxorubicin. Clinical Pharmacokinetics, 15, 15–31. https://doi.org/10.2165/00003088-198815010-00002
Von Hoff, D. D., Layard, M. W., Basa, P., Davis, H. L., Jr., Von Hoff, A. L., Rozencweig, M., & Muggia, F. M. (1979). Risk factors for doxorubicin-induced congestive heart failure. Annals of Internal Medicine, 91, 710–717. https://doi.org/10.7326/0003-4819-91-5-710
Steinherz, L. J., Steinherz, P. G., Tan, C. T. C., Heller, G., & Murphy, M. L. (1991). Cardiac toxicity 4 to 20 years after completing anthracycline therapy. JAMA, 266, 1672–1677. https://doi.org/10.1001/jama.1991.03470120074036
Ganz, P. A., Hussey, M. A., Moinpour, C. M., Unger, J. M., Hutchins, L. F., Dakhil, S. R., Giguere, J. K., Goodwin, J. W., Martino, S., & Albain, K. S. (2008). Late cardiac effects of adjuvant chemotherapy in breast cancer survivors treated on Southwest Oncology Group protocol s8897. Journal of Clinical Oncology, 26, 1223–1230. https://doi.org/10.1200/jco.2007.11.8877
Avagimyan, A., Mkrtchyan, L., Abrahomovich, O., Sheibani, M., Guevorkyan, A., Sarrafzadegan, N., Kozhukhov, S., Agati, L., Astengiano, R., Zaritska, V., & Jndoyan, Z. (2021). AC-mode of chemotherapy as a trigger of cardiac syndrome X: A case study. Current Problems in Cardiology. https://doi.org/10.1016/j.cpcardiol.2021.100994
Stark, C., Taimen, P., Savunen, T., & Koskenvuo, J. (2018). Pegylated and liposomal doxorubicin is associated with high mortality and causes limited cardiotoxicity in mice. BMC Research Notes, 11, 148. https://doi.org/10.1186/s13104-018-3260-6
Boucek, R. J., Kunkel, E. M., Graham, T. P., Brenner, D., & Olson, R. D. (1987). Doxorubicinol, the metabolite of doxorubucin, is more cardiotoxic than doxorubicin. Pediatric Research, 21, 187–187. https://doi.org/10.1203/00006450-198704010-00127
Olson, R. D., Mushlin, P. S., Brenner, D. E., Fleischer, S., Cusack, B. J., Chang, B. K., & Boucek, R. J., Jr. (1988). Doxorubicin cardiotoxicity may be caused by its metabolite, doxorubicinol. Proceedings of the National Academy of Sciences of the United States of America, 85, 3585–3589. https://doi.org/10.1073/pnas.85.10.3585
Lehenbauer Ludke, A. R., Al-Shudiefat, A.A.-R.S., Dhingra, S., Jassal, D. S., & Singal, P. K. (2009). A concise description of cardioprotective strategies in doxorubicin-induced cardiotoxicity. Canadian Journal of Physiology and Pharmacology, 87, 756–763.
Pecoraro, M., Del Pizzo, M., Marzocco, S., Sorrentino, R., Ciccarelli, M., Iaccarino, G., Pinto, A., & Popolo, A. (2016). Inflammatory mediators in a short-time mouse model of doxorubicin-induced cardiotoxicity. Toxicology and applied pharmacology, 293, 44–52.
Osataphan, N., Phrommintikul, A., Chattipakorn, S. C., & Chattipakorn, N. (2020). Effects of doxorubicin-induced cardiotoxicity on cardiac mitochondrial dynamics and mitochondrial function: Insights for future interventions. Journal of Cellular and Molecular Medicine, 24, 6534–6557.
Singal, P., Li, T., Kumar, D., Danelisen, I., & Iliskovic, N. (2000). Adriamycin-induced heart failure: Mechanisms and modulation. Molecular and Cellular Biochemistry, 207, 77–86.
Minotti, G., Menna, P., Salvatorelli, E., Cairo, G., & Gianni, L. (2004). Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacological Reviews, 56, 185–229.
Tokarska-Schlattner, M., Zaugg, M., Zuppinger, C., Wallimann, T., & Schlattner, U. (2006). New insights into doxorubicin-induced cardiotoxicity: The critical role of cellular energetics. Journal of Molecular and Cellular Cardiology, 41, 389–405.
Aldieri, E., Bergandi, L., Riganti, C., Costamagna, C., Bosia, A., & Ghigo, D. (2002). Doxorubicin induces an increase of nitric oxide synthesis in rat cardiac cells that is inhibited by iron supplementation. Toxicology and Applied Pharmacology, 185, 85–90.
Bahadır, A., Kurucu, N., Kadıoğlu, M., & Yenilme, E. (2014). The role of nitric oxide in Doxorubicin-induced cardiotoxicity: Experimental study. Turkish Journal of Hematology, 31, 68.
Nozaki, N., Shishido, T., Takeishi, Y., & Kubota, I. (2004). Modulation of doxorubicin-induced cardiac dysfunction in toll-like receptor-2-knockout mice. Circulation, 110, 2869–2874.
Riad, A., Bien, S., Gratz, M., Escher, F., Heimesaat, M. M., Bereswill, S., Krieg, T., Felix, S. B., Schultheiss, H. P., & Kroemer, H. K. (2008). Toll-like receptor-4 deficiency attenuates doxorubicin-induced cardiomyopathy in mice. European Journal of Heart Failure, 10, 233–243.
Hu, C., Zhang, X., Zhang, N., Wei, W. Y., Li, L. L., Ma, Z. G., & Tang, Q. Z. (2020). Osteocrin attenuates inflammation, oxidative stress, apoptosis, and cardiac dysfunction in doxorubicin-induced cardiotoxicity. Clinical and Translational Medicine, 10, e124.
Pecoraro, M., Sorrentino, R., Franceschelli, S., Del Pizzo, M., Pinto, A., & Popolo, A. (2015). Doxorubicin-mediated cardiotoxicity: Role of mitochondrial connexin 43. Cardiovascular Toxicology, 15, 366–376.
Lyu, Y. L., Kerrigan, J. E., Lin, C.-P., Azarova, A. M., Tsai, Y.-C., Ban, Y., & Liu, L. F. (2007). Topoisomerase IIβ–mediated DNA double-strand breaks: Implications in doxorubicin cardiotoxicity and prevention by dexrazoxane. Cancer Research, 67, 8839–8846.
Horie, T., Ono, K., Nishi, H., Nagao, K., Kinoshita, M., Watanabe, S., Kuwabara, Y., Nakashima, Y., Takanabe-Mori, R., & Nishi, E. (2010). Acute doxorubicin cardiotoxicity is associated with miR-146a-induced inhibition of the neuregulin-ErbB pathway. Cardiovascular Research, 87, 656–664.
Rohrbach, S., Muller-Werdan, U., Werdan, K., Koch, S., Gellerich, N. F., & Holtz, J. (2005). Apoptosis-modulating interaction of the neuregulin/erbB pathway with antracyclines in regulating Bcl-xS and Bcl-xL in cardiomyocytes. Journal of Molecular and Cellular Cardiology, 38, 485–493.
Renu, K., Abilash, V., & Arunachalam, S. (2018). Molecular mechanism of doxorubicin-induced cardiomyopathy–An update. European Journal of Pharmacology, 818, 241–253.
Lebrecht, D., Setzer, B., Ketelsen, U.-P., Haberstroh, J. R., & Walker, U. A. (2003). Time-dependent and tissue-specific accumulation of mtDNA and respiratory chain defects in chronic doxorubicin cardiomyopathy. Circulation, 108, 2423–2429.
Lebrecht, D., Geist, A., Ketelsen, U. P., Haberstroh, J., Setzer, B., & Walker, U. (2007). Dexrazoxane prevents doxorubicin-induced long-term cardiotoxicity and protects myocardial mitochondria from genetic and functional lesions in rats. British Journal of Pharmacology, 151, 771–778.
Montaigne, D., Marechal, X., Baccouch, R., Modine, T., Preau, S., Zannis, K., Marchetti, P., Lancel, S., & Neviere, R. (2010). Stabilization of mitochondrial membrane potential prevents doxorubicin-induced cardiotoxicity in isolated rat heart. Toxicology and Applied Pharmacology, 244, 300–307.
Danz, E. D. B., Skramsted, J., Henry, N., Bennett, J. A., & Keller, R. S. (2009). Resveratrol prevents doxorubicin cardiotoxicity through mitochondrial stabilization and the Sirt1 pathway. Free Radical Biology and Medicine, 46, 1589–1597.
Liu, Y., Asnani, A., Zou, L., Bentley, V. L., Yu, M., Wang, Y., Dellaire, G., Sarkar, K. S., Dai, M., Chen, H. H., Sosnovik, D. E., Shin, J. T., Haber, D. A., Berman, J. N., Chao, W., & Peterson, R. T. (2014). Visnagin protects against doxorubicin-induced cardiomyopathy through modulation of mitochondrial malate dehydrogenase. Science Translational Medicine, 6, 266ra170. https://doi.org/10.1126/scitranslmed.3010189
Fisher, P. W., Salloum, F., Das, A., Hyder, H., & Kukreja, R. C. (2005). Phosphodiesterase-5 inhibition with sildenafil attenuates cardiomyocyte apoptosis and left ventricular dysfunction in a chronic model of doxorubicin cardiotoxicity. Circulation, 111, 1601–1610. https://doi.org/10.1161/01.cir.0000160359.49478.c2
Beak, J., Huang, W., Parker, J. S., Hicks, S. T., Patterson, C., Simpson, P. C., Ma, A., Jin, J., & Jensen, B. C. (2017). An oral selective alpha-1A adrenergic receptor agonist prevents doxorubicin cardiotoxicity. JACC. Basic to translational science, 2, 39–53. https://doi.org/10.1016/j.jacbts.2016.10.006
Yourtee, D. M., Elkins, L. L., Nalvarte, E. L., & Smith, R. E. (1992). Amplification of doxorubicin mutagenicity by cupric ion. Toxicology and Applied Pharmacology, 116, 57–65.
Colombo, R., Dalle Donne, I., & Milzani, A. (1990). Metal ions modulate the effect of doxorubicin on actin assembly. Cancer Biochemistry Biophysics, 11, 217–226.
Shi, Y., Moon, M., Dawood, S., McManus, B., & Liu, P. (2011). Mechanisms and management of doxorubicin cardiotoxicity. Herz, 36, 296–305.
Panjrath, G. S., Patel, V., Valdiviezo, C. I., Narula, N., Narula, J., & Jain, D. (2007). Potentiation of doxorubicin cardiotoxicity by iron loading in a rodent model. Journal of the American College of Cardiology, 49, 2457–2464.
Al-Shabanah, O. A., Aleisa, A. M., Hafez, M. M., Al-Rejaie, S. S., Al-Yahya, A. A., Bakheet, S. A., Al-Harbi, M. M., & Sayed-Ahmed, M. M. (2012). Desferrioxamine attenuates doxorubicin-induced acute cardiotoxicity through TFG-/Smad p53 pathway in rat model. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2012/619185
Al-Harbi, M., Al-Gharably, N. M., Al-Shabanah, O. A., Al-Bekairi, A. M., Osman, A. M. M., & Tawfik, H. N. (1992). Prevention of doxorubicin-induced myocardial and haematological toxicities in rats by the iron chelator desferrioxamine. Cancer Chemotherapy and Pharmacology, 31, 200–204.
Ammar, E.-S.M., Said, S. A., Suddek, G. M., & El-Damarawy, S. L. (2011). Amelioration of doxorubicin-induced cardiotoxicity by deferiprone in rats. Canadian Journal of Physiology and Pharmacology, 89, 269–276.
van Acker, S. A., van Balen, G. P., van den Berg, D. J., Bast, A., & van der Vijgh, W. J. (1998). Influence of iron chelation on the antioxidant activity of flavonoids. Biochemical Pharmacology, 56, 935–943.
Van Acker, S. A., Tromp, M. N., Griffioen, D. H., Van Bennekom, W. P., Van Der Vijgh, W. J., & Bast, A. (1996). Structural aspects of antioxidant activity of flavonoids. Free Radical Biology and Medicine, 20, 331–342.
Bast, A., Haenen, G. R., Bruynzeel, A. M., & Van der Vijgh, W. J. (2007). Protection by flavonoids against anthracycline cardiotoxicity: From chemistry to clinical trials. Cardiovascular Toxicology, 7, 154–159.
Willems, A. M., Bruynzeel, A. M., Kedde, M. A., Van Groeningen, C. J., Bast, A., & Van Der Vijgh, W. J. (2006). A phase I study of monohydroxyethylrutoside in healthy volunteers. Cancer Chemotherapy and Pharmacology, 57, 678–684.
Bast, A., Kaiserová, H., Den Hartog, G., Haenen, G., & Van Der Vijgh, W. (2007). Protectors against doxorubicin-induced cardiotoxicity: Flavonoids. Cell Biology and Toxicology, 23, 39–47.
Ehrke, M. J., Ryoyama, K., & Cohen, S. A. (1984). Cellular basis for adriamycin-induced augmentation of cell-mediated cytotoxicity in culture. Cancer Research, 44, 2497–2504.
Maccubbin, D. L., Wing, K. R., Mace, K. F., Ho, R. L., Ehrke, M. J., & Mihich, E. (1992). Adriamycin-induced modulation of host defenses in tumor-bearing mice. Cancer Research, 52, 3572–3576.
Nagai, K., Fukuno, S., Oda, A., & Konishi, H. (2016). Protective effects of taurine on doxorubicin-induced acute hepatotoxicity through suppression of oxidative stress and apoptotic responses. Anti-cancer Drugs, 27, 17–23.
Wang, S., Kotamraju, S., Konorev, E., Kalivendi, S., Joseph, J., & Kalyanaraman, B. (2002). Activation of nuclear factor-κB during doxorubicin-induced apoptosis in endothelial cells and myocytes is pro-apoptotic: The role of hydrogen peroxide. Biochemical Journal, 367, 729–740.
Sheibani, M., Nezamoleslami, S., Faghir-Ghanesefat, H., Hossein Emami, A., & Dehpour, A. R. (2020). Cardioprotective effects of dapsone against doxorubicin-induced cardiotoxicity in rats. Cancer Chemotherapy and Pharmacology, 85, 563–571.
Sun, Z., Yan, B., Yu, W. Y., Yao, X., Ma, X., Sheng, G., & Ma, Q. (2016). Vitexin attenuates acute doxorubicin cardiotoxicity in rats via the suppression of oxidative stress, inflammation and apoptosis and the activation of FOXO3a. Experimental and Therapeutic Medicine, 12, 1879–1884.
Shaker, R. A., Abboud, S. H., Assad, H. C., & Hadi, N. (2018). Enoxaparin attenuates doxorubicin induced cardiotoxicity in rats via interfering with oxidative stress, inflammation and apoptosis. BMC Pharmacology and Toxicology, 19, 1–10.
Inchiosa, M. A., Jr., & Smith, C. M. (1990). Effects of ibuprofen on doxorubicin toxicity. Research Communications in Chemical Pathology and Pharmacology, 67, 63–78.
Guo, R., Wu, K., Chen, J., Mo, L., Hua, X., Zheng, D., Chen, P., Chen, G., Xu, W., & Feng, J. (2013). Exogenous hydrogen sulfide protects against doxorubicin-induced inflammation and cytotoxicity by inhibiting p38MAPK/NFκB pathway in H9c2 cardiac cells. Cellular Physiology and Biochemistry, 32, 1668–1680.
Yarmohammadi, F., Rezaee, R., & Karimi, G. (2021). Natural compounds against doxorubicin-induced cardiotoxicity: A review on the involvement of Nrf2/ARE signaling pathway. Phytotherapy Research, 35, 1163–1175.
Xiong, C., Wu, Y. Z., Zhang, Y., Wu, Z. X., Chen, X. Y., Jiang, P., Guo, H. C., Xie, K. R., Wang, K. X., & Su, S. W. (2018). Protective effect of berberine on acute cardiomyopathy associated with doxorubicin treatment. Oncology Letters, 15, 5721–5729.
Asensio-López, M. C., Soler, F., Pascual-Figal, D., Fernández-Belda, F., & Lax, A. (2017). Doxorubicin-induced oxidative stress: The protective effect of nicorandil on HL-1 cardiomyocytes. PLoS ONE, 12, e0172803.
Morgan, M. J., & Liu, Z. G. (2011). Crosstalk of reactive oxygen species and NF-κB signaling. Cell Research, 21, 103–115. https://doi.org/10.1038/cr.2010.178
Zhao, Y., Miriyala, S., Miao, L., Mitov, M., Schnell, D., Dhar, S. K., Cai, J., Klein, J. B., Sultana, R., Butterfield, D. A., Vore, M., Batinic-Haberle, I., Bondada, S., & St Clair, D. K. (2014). Redox proteomic identification of HNE-bound mitochondrial proteins in cardiac tissues reveals a systemic effect on energy metabolism after doxorubicin treatment. Free Radical Biology & Medicine, 72, 55–65. https://doi.org/10.1016/j.freeradbiomed.2014.03.001
Guo, C., Sun, L., Chen, X., & Zhang, D. (2013). Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regeneration Research, 8, 2003–2014. https://doi.org/10.3969/j.issn.1673-5374.2013.21.009
Sardão, V. A., Oliveira, P. J., Holy, J., Oliveira, C. R., & Wallace, K. B. (2009). Doxorubicin-induced mitochondrial dysfunction is secondary to nuclear p53 activation in H9c2 cardiomyoblasts. Cancer Chemotherapy and Pharmacology, 64, 811–827. https://doi.org/10.1007/s00280-009-0932-x
Ma, Q. (2013). Role of nrf2 in oxidative stress and toxicity. Annual Review of Pharmacology and Toxicology, 53, 401–426. https://doi.org/10.1146/annurev-pharmtox-011112-140320
Loboda, A., Damulewicz, M., Pyza, E., Jozkowicz, A., & Dulak, J. (2016). Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: An evolutionarily conserved mechanism. Cellular and Molecular Life Sciences: CMLS, 73, 3221–3247. https://doi.org/10.1007/s00018-016-2223-0
Singh, P., Sharma, R., McElhanon, K., Allen, C. D., Megyesi, J. K., Beneš, H., & Singh, S. P. (2015). Sulforaphane protects the heart from doxorubicin-induced toxicity. Free Radical Biology & Medicine, 86, 90–101. https://doi.org/10.1016/j.freeradbiomed.2015.05.028
Chen, M., Samuel, V. P., Wu, Y., Dang, M., Lin, Y., Sriramaneni, R., Sah, S. K., Chinnaboina, G. K., & Zhang, G. (2019). Nrf2/HO-1 mediated protective activity of genistein against doxorubicin-induced cardiac toxicity. Journal of Environmental Pathology, Toxicology and Oncology, 38, 143–152. https://doi.org/10.1615/JEnvironPatholToxicolOncol.2019029341
Cheng, X., Liu, D., Xing, R., Song, H., Tian, X., Yan, C., & Han, Y. (2020). Orosomucoid 1 attenuates doxorubicin-induced oxidative stress and apoptosis in cardiomyocytes via Nrf2 signaling. BioMed Research International, 2020, 5923572. https://doi.org/10.1155/2020/5923572
El-Agamy, D. S., El-Harbi, K. M., Khoshhal, S., Ahmed, N., Elkablawy, M. A., Shaaban, A. A., & Abo-Haded, H. M. (2019). Pristimerin protects against doxorubicin-induced cardiotoxicity and fibrosis through modulation of Nrf2 and MAPK/NF-kB signaling pathways. Cancer Management and Research, 11, 47–61. https://doi.org/10.2147/cmar.s186696
Fadillioglu, E., Oztas, E., Erdogan, H., Yagmurca, M., Sogut, S., Ucar, M., & Irmak, M. K. (2004). Protective effects of caffeic acid phenethyl ester on doxorubicin-induced cardiotoxicity in rats. Journal of Applied Toxicology: An International Journal, 24, 47–52.
Alkreathy, H., Damanhouri, Z. A., Ahmed, N., Slevin, M., Ali, S. S., & Osman, A.-M.M. (2010). Aged garlic extract protects against doxorubicin-induced cardiotoxicity in rats. Food and Chemical Toxicology, 48, 951–956.
Sheibani, M., Faghir-Ghanesefat, H., Dehpour, S., Keshavarz-Bahaghighat, H., Sepand, M. R., Ghahremani, M. H., Azizi, Y., Rahimi, N., & Dehpour, A. R. (2019). Sumatriptan protects against myocardial ischaemia-reperfusion injury by inhibition of inflammation in rat model. Inflammopharmacology, 27, 1071–1080. https://doi.org/10.1007/s10787-019-00586-5
Bazmandegan, G., Amirteimoury, M., Kaeidi, A., Shamsizadeh, A., Khademalhosseini, M., Nematollahi, M. H., Hassanipour, M., & Fatemi, I. (2019). Sumatriptan ameliorates renal injury induced by cisplatin in mice. Iranian Journal of Basic Medical Sciences, 22, 563–567. https://doi.org/10.22038/ijbms.2019.33620.8020
Eslami, F., Rahimi, N., Ostovaneh, A., Ghasemi, M., Dejban, P., Abbasi, A., & Dehpour, A. R. (2021). Sumatriptan reduces severity of status epilepticus induced by lithium-pilocarpine through nitrergic transmission and 5-HT(1B/D) receptors in rats: A pharmacological-based evidence. Fundamental & Clinical Pharmacology, 35, 131–140. https://doi.org/10.1111/fcp.12590
Mohammad, S., Hedyeh, F.-G., Yaser, A., Tahmineh, M., Hasan, Y. M., Roya Sattarzadeh, B., Amir Hossein, E., & Ahmad Reza, D. (2021). Anti-inflammatory and antioxidative effects of sumatriptan against doxorubicin-induced cardiotoxicity in rat. Acta Medica Iranica. https://doi.org/10.18502/acta.v59i7.7020
Iqbal, M., Dubey, K., Anwer, T., Ashish, A., & Pillai, K. K. (2008). Protective effects of telmisartan against acute doxorubicin-induced cardiotoxicity in rats. Pharmacological Reports, 60, 382.
Siveski-Iliskovic, N., Hill, M., Chow, D. A., & Singal, P. K. (1995). Probucol protects against adriamycin cardiomyopathy without interfering with its antitumor effect. Circulation, 91, 10–15. https://doi.org/10.1161/01.cir.91.1.10
Siveski-Iliskovic, N., Kaul, N., & Singal, P. K. (1994). Probucol promotes endogenous antioxidants and provides protection against adriamycin-induced cardiomyopathy in rats. Circulation, 89, 2829–2835. https://doi.org/10.1161/01.cir.89.6.2829
Nagi, M. N., & Mansour, M. A. (2000). Protective effect of thymoquinone against doxorubicin-induced cardiotoxicity in rats: A possible mechanism of protection. Pharmacological Research, 41, 283–289.
Sadzuka, Y., Sugiyama, T., Shimoi, K., Kinae, N., & Hirota, S. (1997). Protective effect of flavonoids on doxorubicin-induced cardiotoxicity. Toxicology letters, 92, 1–7.
Zhang, Y., Ma, C., Liu, C., & Wei, F. (2020). Luteolin attenuates doxorubicin-induced cardiotoxicity by modulating the PHLPP1/AKT/Bcl-2 signalling pathway. PeerJ, 8, e8845.
Chen, J.-Y., Hu, R.-Y., & Chou, H.-C. (2013). Quercetin-induced cardioprotection against doxorubicin cytotoxicity. Journal of Biomedical Science, 20, 1–11.
Kaiserová, H., Šimůnek, T., van der Vijgh, W. J., Bast, A., & Kvasničková, E. (2007). Flavonoids as protectors against doxorubicin cardiotoxicity: Role of iron chelation, antioxidant activity and inhibition of carbonyl reductase. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1772, 1065–1074.
Laughton, M. J., Halliwell, B., Evans, P. J., Robin, J., & Hoult, S. (1989). Antioxidant and pro-oxidant actions of the plant phenolics quercetin, gossypol and myricetin: Effects on lipid peroxidation, hydroxyl radical generation and bleomycin-dependent damage to DNA. Biochemical Pharmacology, 38, 2859–2865.
Chang, D., Li, H., Qian, C., & Wang, Y. (2019). Diohf protects against doxorubicin-induced cardiotoxicity through ERK1 signaling pathway. Frontiers in Pharmacology, 10, 1081.
Wang, H., Yu, P., Gou, H., Zhang, J., Zhu, M., Wang, Z. H., Tian, J. W., Jiang, Y. T., & Fu, F. H. (2012). Cardioprotective effects of 20(S)-ginsenoside Rh2 against doxorubicin-induced cardiotoxicity in vitro and in vivo. Evidence-Based Complementary and Alternative Medicine, 2012, 506214. https://doi.org/10.1155/2012/506214
Li, L., Pan, Q., Han, W., Liu, Z., Li, L., & Hu, X. (2007). Schisandrin B prevents doxorubicin-induced cardiotoxicity via enhancing glutathione redox cycling. Clinical Cancer Research, 13, 6753–6760. https://doi.org/10.1158/1078-0432.Ccr-07-1579
Zhu, W., Soonpaa, M. H., Chen, H., Shen, W., Payne, R. M., Liechty, E. A., Caldwell, R. L., Shou, W., & Field, L. J. (2009). Acute doxorubicin cardiotoxicity is associated with p53-induced inhibition of the mammalian target of rapamycin pathway. Circulation, 119, 99–106. https://doi.org/10.1161/CIRCULATIONAHA.108.799700
Timm, K. N., & Tyler, D. J. (2020). The role of AMPK activation for cardioprotection in doxorubicin-induced cardiotoxicity. Cardiovascular Drugs and Therapy, 34, 255–269. https://doi.org/10.1007/s10557-020-06941-x
Kobashigawa, L. C., Xu, Y. C., Padbury, J. F., Tseng, Y.-T., & Yano, N. (2014). Metformin protects cardiomyocyte from doxorubicin induced cytotoxicity through an AMP-activated protein kinase dependent signaling pathway: an in vitro study. PLoS ONE, 9, e104888.
Zilinyi, R., Czompa, A., Czegledi, A., Gajtko, A., Pituk, D., Lekli, I., & Tosaki, A. (2018). The cardioprotective effect of metformin in doxorubicin-induced cardiotoxicity: The role of autophagy. Molecules (Basel, Switzerland). https://doi.org/10.3390/molecules23051184
Huelsenbeck, J., Henninger, C., Schad, A., Lackner, K. J., Kaina, B., & Fritz, G. (2011). Inhibition of Rac1 signaling by lovastatin protects against anthracycline-induced cardiac toxicity. Cell Death & Disease, 2, e190. https://doi.org/10.1038/cddis.2011.65
Rawat, P. S., Jaiswal, A., Khurana, A., Bhatti, J. S., & Navik, U. (2021). Doxorubicin-induced cardiotoxicity: An update on the molecular mechanism and novel therapeutic strategies for effective management. Biomedicine & Pharmacotherapy, 139, 111708. https://doi.org/10.1016/j.biopha.2021.111708
Vedam, K., Nishijima, Y., Druhan, L. J., Khan, M., Moldovan, N. I., Zweier, J. L., & Ilangovan, G. (2010). Role of heat shock factor-1 activation in the doxorubicin-induced heart failure in mice. American Journal of Physiology, Heart and Circulatory Physiology, 298, H1832–H1841. https://doi.org/10.1152/ajpheart.01047.2009
Liu, P., Bao, H. Y., Jin, C. C., Zhou, J. C., Hua, F., Li, K., Lv, X. X., Cui, B., Hu, Z. W., & Zhang, X. W. (2019). Targeting extracellular heat shock protein 70 ameliorates doxorubicin-induced heart failure through resolution of toll-like receptor 2-mediated myocardial inflammation. Journal of the American Heart Association, 8, e012338. https://doi.org/10.1161/jaha.119.012338
Shan, Y. X., Liu, T. J., Su, H. F., Samsamshariat, A., Mestril, R., & Wang, P. H. (2003). Hsp10 and Hsp60 modulate Bcl-2 family and mitochondria apoptosis signaling induced by doxorubicin in cardiac muscle cells. Journal of Molecular and Cellular Cardiology, 35, 1135–1143. https://doi.org/10.1016/s0022-2828(03)00229-3
Zhao, D., Xue, C., Li, J., Feng, K., Zeng, P., Chen, Y., Duan, Y., Zhang, S., Li, X., & Han, J. (2020). Adiponectin agonist ADP355 ameliorates doxorubicin-induced cardiotoxicity by decreasing cardiomyocyte apoptosis and oxidative stress. Biochemical and Biophysical Research Communications, 533, 304–312.
Ramond, A., Sartorius, E., Mousseau, M., Ribuot, C., & Joyeux-Faure, M. (2008). Erythropoietin pretreatment protects against acute chemotherapy toxicity in isolated rat hearts. Experimental Biology and Medicine, 233, 76–83. https://doi.org/10.3181/0706-RM-152
Man, S. M., Karki, R., Briard, B., Burton, A., Gingras, S., Pelletier, S., & Kanneganti, T.-D. (2017). Differential roles of caspase-1 and caspase-11 in infection and inflammation. Scientific Reports, 7, 45126. https://doi.org/10.1038/srep45126
Yu, W., Qin, X., Zhang, Y., Qiu, P., Wang, L., Zha, W., & Ren, J. (2020). Curcumin suppresses doxorubicin-induced cardiomyocyte pyroptosis via a PI3K/Akt/mTOR-dependent manner. Cardiovascular Diagnosis and Therapy, 10, 752–769. https://doi.org/10.21037/cdt-19-707
Chen, Y. L., Chung, S. Y., Chai, H. T., Chen, C. H., Liu, C. F., Chen, Y. L., Huang, T. H., Zhen, Y. Y., Sung, P. H., Sun, C. K., Chua, S., Lu, H. I., Lee, F. Y., Sheu, J. J., & Yip, H. K. (2015). Early administration of carvedilol protected against doxorubicin-induced cardiomyopathy. The Journal of Pharmacology and Experimental Therapeutics, 355, 516–527. https://doi.org/10.1124/jpet.115.225375
Dulhunty, A., & Casarotto, B. (2011). The ryanodine receptor: A pivotal Ca2+ regulatory protein and potential therapeutic drug target. Current Drug Targets, 12, 709–723.
Dewenter, M., von der Lieth, A., Katus, H. A., & Backs, J. (2017). Calcium signaling and transcriptional regulation in cardiomyocytes. Circulation Research, 121, 1000–1020.
Aziz, A. U. R., Geng, C., Li, W., Yu, X., Qin, K.-R., Wang, H., & Liu, B. (2019). Doxorubicin induces ER calcium release via Src in rat ovarian follicles. Toxicological Sciences, 168, 171–178.
Octavia, Y., Tocchetti, C. G., Gabrielson, K. L., Janssens, S., Crijns, H. J., & Moens, A. L. (2012). Doxorubicin-induced cardiomyopathy: From molecular mechanisms to therapeutic strategies. Journal of Molecular and Cellular Cardiology, 52, 1213–1225.
Yarmohmmadi, F., Rahimi, N., Faghir-Ghanesefat, H., Javadian, N., Abdollahi, A., Pasalar, P., Jazayeri, F., Ejtemaeemehr, S., & Dehpour, A. R. (2017). Protective effects of agmatine on doxorubicin-induced chronic cardiotoxicity in rat. European Journal of Pharmacology, 796, 39–44.
Khalilzadeh, M., Abdollahi, A., Abdolahi, F., Abdolghaffari, A. H., Dehpour, A. R., & Jazaeri, F. (2018). Protective effects of magnesium sulfate against doxorubicin induced cardiotoxicity in rats. Life Sciences, 207, 436–441.
Gross, R. A., Moises, H. C., Uhler, M. D., & Macdonald, R. L. (1990). Dynorphin A and cAMP-dependent protein kinase independently regulate neuronal calcium currents. Proceedings of the National Academy of Sciences, 87, 7025–7029.
North, R. A., Williams, J. T., Surprenant, A., & Christie, M. J. (1987). Mu and delta receptors belong to a family of receptors that are coupled to potassium channels. Proceedings of the National Academy of Sciences, 84, 5487–5491.
Lashgari, N. A., Roudsari, N. M., Zandi, N., Pazoki, B., Rezaei, A., Hashemi, M., Momtaz, S., Rahimi, R., Shayan, M., Dehpour, A. R., & Abdolghaffari, A. H. (2021). Current overview of opioids in progression of inflammatory bowel disease; pharmacological and clinical considerations. Molecular Biology Reports, 48, 855–874. https://doi.org/10.1007/s11033-020-06095-x
Zamanian, G., Shayan, M., Rahimi, N., Bahremand, T., Shafaroodi, H., Ejtemaei-Mehr, S., Aghaei, I., & Dehpour, A. R. (2020). Interaction of morphine tolerance with pentylenetetrazole-induced seizure threshold in mice: The role of NMDA-receptor/NO pathway. Epilepsy & Behavior, 112, 107343. https://doi.org/10.1016/j.yebeh.2020.107343
Kelishomi, R. B., Ejtemaeemehr, S., Tavangar, S. M., Rahimian, R., Mobarakeh, J. I., & Dehpour, A. R. (2008). Morphine is protective against doxorubicin-induced cardiotoxicity in rat. Toxicology, 243, 96–104.
Goodenough, D. A., & Paul, D. L. (2009). Gap junctions. Cold Spring Harbor Perspectives in Biology, 1, a002576.
Severs, N. J. (1994). Pathophysiology of gap junctions in heart disease. Journal of Cardiovascular Electrophysiology, 5, 462–475.
Siti, H. N., Jalil, J., Asmadi, A. Y., & Kamisah, Y. (2020). Effects of quercetin on cardiac function in pressure overload and postischemic cardiac injury in rodents: A systematic review and meta-analysis. Cardiovascular Drugs and Therapy, 16, 1–15.
Srisakuldee, W., Makazan, Z., Nickel, B. E., Zhang, F., Thliveris, J. A., Pasumarthi, K. B., & Kardami, E. (2014). The FGF-2-triggered protection of cardiac subsarcolemmal mitochondria from calcium overload is mitochondrial connexin 43-dependent. Cardiovascular Research, 103, 72–80.
Pecoraro, M., Ciccarelli, M., Fiordelisi, A., Iaccarino, G., Pinto, A., & Popolo, A. (2018). Diazoxide improves mitochondrial connexin 43 expression in a mouse model of doxorubicin-induced cardiotoxicity. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms19030757
Elhadidy, M. G., Elmasry, A., Rabei, M. R., & Eladel, A. E. (2020). Effect of ghrelin on VEGF-B and connexin-43 in a rat model of doxorubicin-induced cardiomyopathy. Journal of Basic and Clinical Physiology and Pharmacology. https://doi.org/10.1515/jbcpp-2018-0212
Zhang, H., Zhang, A., Guo, C., Shi, C., Zhang, Y., Liu, Q., Sparatore, A., & Wang, C. (2011). S-diclofenac protects against doxorubicin-induced cardiomyopathy in mice via ameliorating cardiac gap junction remodeling. PLoS ONE, 6, e26441.
Michan, S., & Sinclair, D. (2007). Sirtuins in mammals: Insights into their biological function. Biochemical Journal, 404, 1–13.
Tomaselli, D., Steegborn, C., Mai, A., & Rotili, D. (2020). Sirt4: A multifaceted enzyme at the crossroads of mitochondrial metabolism and cancer. Frontiers in Oncology, 10, 474.
Dolinsky, V. W. (2017). The role of sirtuins in mitochondrial function and doxorubicin-induced cardiac dysfunction. Biological Chemistry, 398, 955–974.
Cheung, K. G., Cole, L. K., Xiang, B., Chen, K., Ma, X., Myal, Y., Hatch, G. M., Tong, Q., & Dolinsky, V. W. (2015). Sirtuin-3 (SIRT3) protein attenuates doxorubicin-induced oxidative stress and improves mitochondrial respiration in H9c2 cardiomyocytes. Journal of Biological Chemistry, 290, 10981–10993.
Liu, M. H., Shan, J., Li, J., Zhang, Y., & Lin, X. L. (2016). Resveratrol inhibits doxorubicin-induced cardiotoxicity via sirtuin 1 activation in H9c2 cardiomyocytes. Experimental and Therapeutic Medicine, 12, 1113–1118.
Ruan, Y., Dong, C., Patel, J., Duan, C., Wang, X., Wu, X., Cao, Y., Pu, L., Lu, D., & Shen, T. (2015). SIRT1 suppresses doxorubicin-induced cardiotoxicity by regulating the oxidative stress and p38MAPK pathways. Cellular Physiology and Biochemistry, 35, 1116–1124.
Pillai, V. B., Kanwal, A., Fang, Y. H., Sharp, W. W., Samant, S., Arbiser, J., & Gupta, M. P. (2017). Honokiol, an activator of Sirtuin-3 (SIRT3) preserves mitochondria and protects the heart from doxorubicin-induced cardiomyopathy in mice. Oncotarget, 8, 34082.
Needham, D. M., Shufelt, K. A., Tomlinson, G., Scholey, J. W., & Newton, G. E. (2004). Troponin I and T levels in renal failure patients without acute coronary syndrome: A systematic review of the literature. The Canadian Journal of Cardiology, 20, 1212–1218.
Jin, J.-P. (2016). Evolution, regulation, and function of N-terminal variable region of troponin T: Modulation of muscle contractility and beyond. International Review of Cell and Molecular Biology, 321, 1–28.
Bleuel, H., Deschl, U., Bertsch, T., Bölz, G., & Rebel, W. (1995). Diagnostic efficiency of troponin T measurements in rats with experimental myocardial cell damage. Experimental and Toxicologic Pathology, 47, 121–127.
Mair, J., & Apple, F. (1997). Progress in myocardial damage detection: New biochemical markers for clinicians. Critical Reviews in Clinical Laboratory Sciences, 34, 1–66.
Rahimi Balaei, M., Momeny, M., Babaeikelishomi, R., Ejtemaei Mehr, S., Tavangar, S. M., & Dehpour, A. R. (2010). The modulatory effect of lithium on doxorubicin-induced cardiotoxicity in rat. European Journal of Pharmacology, 641, 193–198. https://doi.org/10.1016/j.ejphar.2010.05.046
Chang, E. A., Jin, S. W., Nam, M. H., & Kim, S. D. (2019). Human induced pluripotent stem cells: Clinical significance and applications in neurologic diseases. Journal of Korean Neurosurgical Society, 62, 493–501. https://doi.org/10.3340/jkns.2018.0222
Matsui, T., Miyamoto, N., Saito, F., & Shinozawa, T. (2020). Molecular profiling of human induced pluripotent stem cell-derived cells and their application for drug safety study. Current Pharmaceutical Biotechnology, 21, 807–828. https://doi.org/10.2174/1389201021666200422090952
Devalla, H. D., & Passier, R. (2018). Cardiac differentiation of pluripotent stem cells and implications for modeling the heart in health and disease. Science Translational Medicine. https://doi.org/10.1126/scitranslmed.aah5457
Blinova, K., Dang, Q., Millard, D., Smith, G., Pierson, J., Guo, L., Brock, M., Lu, H. R., Kraushaar, U., Zeng, H., Shi, H., Zhang, X., Sawada, K., Osada, T., Kanda, Y., Sekino, Y., Pang, L., Feaster, T. K., Kettenhofen, R., … Gintant, G. (2018). International multisite study of human-induced pluripotent stem cell-derived cardiomyocytes for drug proarrhythmic potential assessment. Cell Reports, 24, 3582–3592. https://doi.org/10.1016/j.celrep.2018.08.079
Schwach, V., Slaats, R. H., & Passier, R. (2020). Human pluripotent stem cell-derived cardiomyocytes for assessment of anticancer drug-induced cardiotoxicity. Frontiers in Cardiovascular Medicine. https://doi.org/10.3389/fcvm.2020.00050
Maillet, A., Tan, K., Chai, X., Sadananda, S. N., Mehta, A., Ooi, J., Hayden, M. R., Pouladi, M. A., Ghosh, S., Shim, W., & Brunham, L. R. (2016). Modeling doxorubicin-induced cardiotoxicity in human pluripotent stem cell derived-cardiomyocytes. Science and Reports, 6, 25333. https://doi.org/10.1038/srep25333
Burridge, P. W., Li, Y. F., Matsa, E., Wu, H., Ong, S. G., Sharma, A., Holmström, A., Chang, A. C., Coronado, M. J., Ebert, A. D., Knowles, J. W., Telli, M. L., Witteles, R. M., Blau, H. M., Bernstein, D., Altman, R. B., & Wu, J. C. (2016). Human induced pluripotent stem cell-derived cardiomyocytes recapitulate the predilection of breast cancer patients to doxorubicin-induced cardiotoxicity. Nature Medicine, 22, 547–556. https://doi.org/10.1038/nm.4087
Cui, N., Wu, F., Lu, W. J., Bai, R., Ke, B., Liu, T., Li, L., Lan, F., & Cui, M. (2019). Doxorubicin-induced cardiotoxicity is maturation dependent due to the shift from topoisomerase IIα to IIβ in human stem cell derived cardiomyocytes. Journal of Cellular and Molecular Medicine, 23, 4627–4639. https://doi.org/10.1111/jcmm.14346
Zhao, L., & Zhang, B. (2017). Doxorubicin induces cardiotoxicity through upregulation of death receptors mediated apoptosis in cardiomyocytes. Science and Reports, 7, 44735. https://doi.org/10.1038/srep44735
McSweeney, K. M., Bozza, W. P., Alterovitz, W.-L., & Zhang, B. (2019). Transcriptomic profiling reveals p53 as a key regulator of doxorubicin-induced cardiotoxicity. Cell Death Discovery, 5, 102. https://doi.org/10.1038/s41420-019-0182-6
Li, J., Wang, P. Y., Long, N. A., Zhuang, J., Springer, D. A., Zou, J., Lin, Y., Bleck, C. K. E., Park, J. H., Kang, J. G., & Hwang, P. M. (2019). p53 prevents doxorubicin cardiotoxicity independently of its prototypical tumor suppressor activities. Proceedings of the National Academy of Sciences of the United States of America, 116, 19626–19634. https://doi.org/10.1073/pnas.1904979116
Deng, S., Yan, T., Jendrny, C., Nemecek, A., Vincetic, M., Gödtel-Armbrust, U., & Wojnowski, L. (2014). Dexrazoxane may prevent doxorubicin-induced DNA damage via depleting both Topoisomerase II isoforms. BMC Cancer, 14, 842. https://doi.org/10.1186/1471-2407-14-842
Ahmed, R. E., Anzai, T., Chanthra, N., & Uosaki, H. (2020). A brief review of current maturation methods for human induced pluripotent stem cells-derived cardiomyocytes. Frontiers in Cell and Developmental Biology. https://doi.org/10.3389/fcell.2020.00178
Pang, L. (2020). Toxicity testing in the era of induced pluripotent stem cells: A perspective regarding the use of patient-specific induced pluripotent stem cell–derived cardiomyocytes for cardiac safety evaluation. Current Opinion in Toxicology, 23–24, 50–55. https://doi.org/10.1016/j.cotox.2020.04.001
Soma, Y., Morita, Y., Kishino, Y., Kanazawa, H., Fukuda, K., & Tohyama, S. (2021). The present state and future perspectives of cardiac regenerative therapy using human pluripotent stem cells. Frontiers in Cardiovascular Medicine. https://doi.org/10.3389/fcvm.2021.774389
Funding
This study was supported by Iran National Sciences Foundation (INSF).
Author information
Authors and Affiliations
Contributions
ARD and MS designed the study. SN, MS, and YA performed the literature search and data analysis. MS and YA drafted the manuscript and edited it. MHF prepared all figures. MS prepared Table 1. FE and SN made critical revision and edited the manuscript. ARD, MS, and MS made final editions to the paper prior to the submission. All authors have read and agreed to the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical Approval
The protocol of this study was approved by the Ethics Committee of Tehran University of Medical Sciences.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Handling Editor: Y. James Kang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sheibani, M., Azizi, Y., Shayan, M. et al. Doxorubicin-Induced Cardiotoxicity: An Overview on Pre-clinical Therapeutic Approaches. Cardiovasc Toxicol 22, 292–310 (2022). https://doi.org/10.1007/s12012-022-09721-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-022-09721-1