Abstract
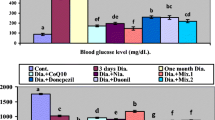
There is now substantial epidemiological evidence that diabetes is a risk factor for cerebrovascular disease. The protection by vanadium from focal cerebral ischemia in diabetic rats was studied in this paper. Rats with streptozotocin-induced diabetes were subjected to middle cerebral artery occlusion followed by 4 weeks of administration of 0.6 mg/ml sodium orthovanadate in drinking water. Vanadium significantly improved the outcome in diabetic rats after cerebral ischemia and reperfusion in terms of neurobehavioral function. Vanadium reduces brain damage in streptozotocin-induced diabetic rats by imitating action of insulin.


Similar content being viewed by others
References
Barceloux DG (1999) Vanadium. J Toxicol Clin Toxicol 37:265–278
Gil J, Miralpeix M, Carreras J, Bartrons R (1988) Insulin-like effects of vanadate on glucokinase activity and fructose 2,6-bisphosphate levels in the liver of diabetic rats. J Biol Chem 263:1868–1871
Shechter Y (1990) Insulin-mimetic effects of vanadate. Possible implications for future treatment of diabetes. Diabetes 39:1–5
Elberg G, Li J, Shechter Y (1994) Vanadium activates or inhibits receptor and non-receptor protein tyrosine kinases in cell-free experiments, depending on its oxidation state: possible role of endogenous vanadium in controlling cellular protein tyrosine kinase activity. J Biol Chem 269:9521–9527
Morinville A, Maysinger D, Shaver A (1998) From Vanadis to Atropos: vanadium compounds as pharmacological tools in cell death signalling. Trends Pharmacol Sci 19:452–460
Kawano T, Fukunaga K, Takeuchi Y, Morioka M, Yano S, Hamada J, Ushio Y, Miyamoto E (2001) Neuroprotective effect of sodium orthovanadate on delayed neuronal death after transient forebrain ischemia in gerbil hippocampus. J Cereb Blood Flow Metab 21:1268–1280
Berger L, Hakim AM (1986) The association of hyperglycemia with cerebral edemain stroke. Stroke 17:865–871
Lukovits TG, Mazzone TM, Gorelick TM (1999) Diabetes mellitus and cerebrovascular disease. Neuroepidemiology 18:1–14
Li PA, Siesjo BK (1997) Role of hyperglycaemia-related acidosis in ischaemic brain damage. Acta Physiol Scand 161:567–580
Li P-A, Gisselsson L, Keuker J, Vogel J, Smith M-L, Kuschinsky W, Siesjo BK (1998) Hyperglycemia-exaggerated ischemic brain damage following 30 min of middle cerebral artery occlusion is not due to capillary obstruction. Brain Res 804:36–44
Gisselsson L, Smith M-L, Siesjo B (1999) Hyperglycemia and focal brain ischemia. J Cereb Blood Flow Metab 19:288–297
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Preet A, Gupta BL, Yadava PK, Baquer NZ (2005) Efficacy of lower doses of vanadium in restoring altered glucose metabolism and antioxidant status in diabetic rat lenses. J Biosci 30(2):221–230
Lee EJ, Chen HY, Wu TS, Chen TY, Ayoub IA, Maynard KI (2002) Acute administration of Ginkgo biloba extract (EGb 761) affords neuroprotection against permanent and transient focal cerebral ischemia in Sprague–Dawley rats. J Neurosci Res 68:636–645
Ali A, Ahmad FJ, Pillai KK, Vohora D (2004) Evidence of the antiepileptic potential of amiloride with neuropharmacological benefits in rodent models of epilepsy and behavior. Epilepsy Behav 5:322–328
Lum G, Gambino SR (1974) A comparison of serum Vs heparinised plasma for routine chemistry tests. Am J Clin Pathol 61:108–113
Caliborne A (1985) Catalase activity. In: Wakd G (ed) CRC hand book of methods for oxygen radical research. CRC, Boca Raton, pp 283–294
Svoboda P, Mosinger B (1981) Catecholamines and the brain microsomal Na, K-adenosinetriphosphatase—I. Protection against lipoperoxidative damage. Biochem Pharmacol 30:427–432
Jollow DJ, Mitchell JR, Zampagline N, Gillette JR (1974) Bromobenzene induced liver necrosis: protective role of glutathione and evidence for 3,4-bromobenzene as the hepatic metabolite. Pharmacology 11:151–169
Wheeler CR, Salzman JA, Elsayed NM, Omaye ST, Korte DW Jr (1990) Automated assays for superoxide dismutase, catalase, glutathione peroxidase and glutathione reductase activity. Anal Biochem 184:193–199
Carlberg I, Mannerviek B (1975) Glutathione reductase levels in rat brain. J Biol Chem 250:5475–5480
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Nesto RW (2004) Correlation between cardiovascular disease and diabetes mellitus: current concepts. Am J Med 116:11–22
Zorzano A, Palacín M, Marti L, García-Vicente S (2009) Arylalkylamine vanadium salts as new anti-diabetic compounds. J Inorg Biochem 103(4):559–566
Esbak H, Enyedy EA, Kiss T, Yoshikawa Y, Sakurai H, Garribba E, Rehder D (2009) Amino acid-derivatised picolinato-oxidovanadium(IV) complexes: characterisation, speciation and ex vivo insulin-mimetic potential. J Inorg Biochem 103(4):590–600
Rehder D (2003) Biological and medical aspects of vanadium. Inorg Chem Commun 6(5):604–617
Crans DC, Smee JJ, Gaidamauskas E, Yang L (2004) The chemistry and biochemistry of vanadium and the biological activities exerted by vanadium compounds. Chem Rev 104(2):849–902
Rizk NN, Rafols JA, Dunbar JC (2006) Cerebral ischemia-induced apoptosis and necrosis in normal and diabetic rats: effects of insulin and C-peptide. Brain Res 1096:204–212
Hui L, Pei DS, Zhang QG, Guan QH, Zhang GY (2005) The neuroprotection of insulin on ischemic brain injury in rat hippocampus through negative regulation of JNK signaling pathway by PI3K/Akt activation. Brain Res 1052:1–9
Aragno M, Brignardello E, Tamagno E, Gatto V, Danni O, Boccuzzi G (1997) Dehydroepiandrosterone administration prevents the oxidative damage induced by acute hyperglycemia in rats. J Endocrinol 155:233–240
Kowluru RA, Engerman RL, Kern TS (1999) Abnormalities of retinal metabolism in diabetes or experimental galactosemia. VI. Comparison of retinal and cerebral cortex metabolism, and effects of antioxidant therapy. Free Radic Biol Med 26:371–378
Coyle JT, Puttfarcken PO (1993) Oxidative stress, glutamate and neurodegenerative disorders. Science 262:689–695
Bains JS, Shaw CA (1997) Neurodegenerative disorders in humans: the role of glutathione in oxidative stress-mediated neuronal death. Brain Res Rev 25:335–338
Shivakumar BR, Kolluri SV, Ravindranath V (1995) Glutathione and protein thiol homeostasis in brain during reperfusion after cerebral ischemia. J Pharmacol Exp Ther 274:1167–1173
Acknowledgment
This work was supported by the Key New Drugs Innovation project from Ministry of Science and Technology (2009ZX09502-017), Program for New Century Excellent Talents in University (NCET-10-0273), Beijing Nova Program (2010B035), the project from Key Laboratory of Mental Health, Chinese Academy of Sciences, Young Scientist project from IPCAS (08CX043004), NNSF grant (30800301), and the Knowledge Innovation Program of the Chinese Academy of Sciences (KSCX2-YW-R-254, KSCX2-EW-Q-18 and KSCX2-EW-J-8).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Z., Li, P., Zhao, D. et al. Protection by Vanadium, a Contemporary Treatment Approach to Both Diabetes and Focal Cerebral Ischemia in Rats. Biol Trace Elem Res 145, 66–70 (2012). https://doi.org/10.1007/s12011-011-9168-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-011-9168-9