Abstract
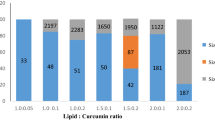
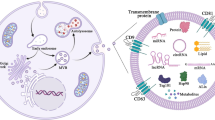
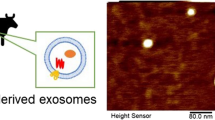
Exosomes, the extracellular secretary nano-vesicles, act as carriers of biomolecules to the target cells. They exhibit several attributes of an efficient drug delivery system. Curcumin, despite having numerous bioactive and therapeutic properties, has limited pharmaceutical use due to its poor water solubility, stability, and low systemic bioavailability. Hence, this study aims to enhance the therapeutic potential of curcumin, a model hydrophobic drug, by its encapsulation into milk exosomes. In the present study, we investigated the stability of free curcumin and exosomal curcumin in PBS and in vitro digestive processes. Additionally, their uptake and trans-epithelial transport were studied on Caco-2 cells. Curcumin in milk exosomes had higher stability in PBS, sustained harsh digestive processes, and crossed the intestinal barrier than free curcumin. In conclusion, the encapsulation of curcumin into the exosomes enhances its stability, solubility, and bioavailability. Therefore, the present study demonstrated that milk exosomes act as stable oral drug delivery vehicles.






Similar content being viewed by others
References
Mathivanan, S., Ji, H., & Simpson, R. J. (2010). Exosomes: extracellular organelles important in intercellular communication. Journal of Proteomics, 73, 1907–1920.
Valadi, H., Ekström, K., Bossios, A., Sjöstrand, M., Lee, J. J., & Lötvall, J. O. (2007). Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nature Cell Biology, 9, 654–659.
Wang, J., Yao, Y., Xiong, J., Wu, J., Tang, X., & Li, G. (2015). Evaluation of the inflammatory response in macrophages stimulated with exosomes secreted by Mycobacterium avium-infected macrophages. BioMed Research International, 2015, 1–9.
Sung, B. H., Ketova, T., Hoshino, D., Zijlstra, A., & Weaver, A. M. (2015). Directional cell movement through tissues is controlled by exosome secretion. Nature Communications, 6, 1–14.
Holder, B., Jones, T., Sancho, S. V., Rice, T. F., Donaldson, B., Bouqueau, M., Forbes, K., & Kampmann, B. (2016). Macrophage exosomes induce placental inflammatory cytokines: a novel mode of maternal-placental messaging. Traffic, 17, 168–178.
Wang, J., De, V. K., Faict, S., Frassanito, M. A., Ribatti, D., Vacca, A., & Menu, E. (2016). Multiple myeloma exosomes establish a favourable bone marrow microenvironment with enhanced angiogenesis and immunosuppression. The Journal of Pathology, 239, 162–173.
Karlsson, T., Lundholm, M., Widmark, A., & Persson, E. (2016). Tumor cell-derived exosomes from the prostate cancer cell line TRAMP-C1 impair osteoclast formation and differentiation. PloS One, 11, 1–12.
Harada, T., Yamamoto, H., Kishida, S., Kishida, M., Awada, C., Takao, T., & Kikuchi, A. (2016). Wnt5b-associated exosomes promote cancer cell migration and proliferation. Cancer Science, 108, 42–52.
Wu, L., Zhang, X., Zhang, B., Shi, H., Yuan, X., Sun, Y., Pan, Z., Qian, H., & Xu, W. (2016). Exosomes derived from gastric cancer cells activate NF-κB pathway in macrophages to promote cancer progression. Tumour Biology, 37, 12169–12180.
Munoz, J. L., Bliss, S. A., Greco, S. J., Ramkissoon, S. H., Ligon, K. L., & Rameshwar, P. (2013). Delivery of functional anti-miR-9 by mesenchymal stem cell-derived exosomes to glioblastoma multiforme cells conferred chemosensitivity. Molecular Therapy- Nucleic Acids, 2, 1–11.
Alvarez-Erviti, L., Seow, Y., Yin, H., Betts, C., Lakhal, S., & Wood, M. J. A. (2011). Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nature Biotechnology, 29, 341–345.
Haney, M. J., Klyachko, N. L., Zhao, Y., Gupta, R., Plotnikova, E. G., He, Z., Patel, T., Piroyan, A., Sokolsky, M., Kabanov, A. V., & Batrakova, E. V. (2015). Exosomes as drug delivery vehicles for Parkinson’s disease therapy. Journal of Controlled Release, 207, 18–30.
Tian, Y., Li, S., Song, J., Ji, T., Zhu, M., Anderson, G. J., et al. (2013). A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials, 35, 2383–2390.
Kim, M. S., Haney, M. J., Zhao, Y., Mahajan, V., Deygen, I., Klyachko, N. L., Inskoe, E., Piroyan, A., Sokolsky, M., Okolie, O., Hingtgen, S. D., Kabanov, A. V., & Batrakova, E. V. (2016). Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine, 12, 655–664.
Davidson, S. M., Takov, K., & Yellon, D. M. (2016). Exosomes and cardiovascular protection. Cardiovascular Drugs and Therapy, 31, 77–86.
Kouwaki, T., Fukushima, Y., Daito, T., Sanada, T., Yamamoto, N., Mifsud, E. J., Leong, C. R., Tsukiyama-Kohara, K., Kohara, M., Matsumoto, M., Seya, T., & Oshiumi, H. (2016). Extracellular vesicles including exosomes regulate innate immune responses to hepatitis B virus infection. Frontiers in Immunology, 7, 1–13.
Ferreira, V. H., Nazli, A., Dizzell, S. E., Mueller, K., & Kaushic, C. (2015). The anti-inflammatory activity of curcumin protects the genital mucosal epithelial barrier from disruption and blocks replication of HIV-1 and HSV-2. PloS One, 10, 1–18.
Beevers, C. S., Chen, L., Liu, L., Luo, Y., Webster, N. J., & Huang, S. (2009). Curcumin disrupts the mammalian target of rapamycin-raptor complex. Cancer Research, 69, 1000–1008.
Gordon, O. N., Luis, P. B., Sintim, H. O., & Schneider, C. (2015). Unraveling curcumin degradation: autoxidation proceeds through spiroepoxide and vinylether intermediates en route to the main bicyclopentadione. The Journal of Biological Chemistry, 290, 4817–4828.
Sun, Q., Chen, X., Yu, J., Zen, K., Zhang, C. Y., & Li, L. (2013). Immune modulatory function of abundant immune-related microRNAs in microvesicles from bovine colostrum. Protein & Cell, 4, 197–210.
Athira, G., & Jyothi, A. (2014). Preparation and characterization of curcumin loaded cassava starch nanoparticles with improved cellular absorption. International Journal of Pharmacy and Pharmaceutical Sciences, 6, 171–176.
Kopf-Bolanz, K. A., Schwander, F., Gijs, M., Verge’res, G., Portmann, R., & Egger, L. (2011). Validation of an in vitro digestive system for studying macronutrient decomposition in humans. Journal of Nutrition, 142, 245–250.
Vij, R., Reddi, S., Kapila, S., & Kapila, R. (2016). Transepithelial transport of milk derived bioactive peptide VLPVPQK. Food Chemistry, 190, 681–688.
Colombo, M., Raposa, G., & Thery, C. (2014). Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annual Review of Cell and Developmental Biology, 30, 255–289.
Lasser, C., Alikhani, V. S., Ekstrom, K., Eldh, M., Paredes, P. T., & Bossios, A. (2011). Human saliva, plasma and breast milk exosomes contain RNA: uptake by macrophages. Journal of Translational Medicine, 9, 1–8.
Munagala, R., Aqil, F., Jeyabalan, J., & Gupta, R. (2016). Bovine milk-derived exosomes for drug delivery. Cancer Letters, 371, 48–61.
Madison, M. N., Roller, R. J., & Okeoma, C. M. (2014). Human semen contains exosomes with potent anti-HIV activity. Retrovirology, 11, 1–15.
Dear, J. W., Street, J. M., & Bailey, M. A. (2013). Urinary exosomes: a reservoir for biomarker discovery and potential mediators of intrarenal signaling. Proteomics, 13, 1572–1580.
Street, J. M., Barran, P. E., Mackay, C. L., Weidt, S., Balmforth, C., & Walsh, T. S. (2012). Identification and proteomic profiling of exosomes in human cerebrospinal fluid. Journal of Translational Medicine, 10, 1–7.
Thery, C., Ostrowski, M., & Segura, E. (2009). Membrane vesicles as conveyors of immune response. Nature Reviews Immunology, 9, 581–593.
Toro, J. D., Herschlik, L., Waldner, C., & Mongini, C. (2015). Emerging roles of exosomes in normal and pathological conditions: new insights for diagnosis and therapeutics applications. Frontiers in Immunology, 6, 1–12.
Ban, J., Lee, M., Im, W., & Kim, M. (2015). Low pH increases the yield of exosome isolation. Biochemical and Biophysical Research Communication, 461, 76–79.
Malik, Z. A., Kott, K. S., Poe, A. J., Kuo, T., Chen, L., Ferrara, K., & Knowlton, A. (2013). Cardiac myocyte exosomes: stability, HSP60 and proteomics. American Journal of Physiology Heart and Circulatory Physiology, 304, H954–H965.
Sun, D., Zhuang, X., Xiang, X., Liu, Y., Zhang, S., Liu, C., Barnes, S., Grizzle, W., Miller, D., & Zhang, H. (2010). A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Molecular Therapy, 9, 1606–1614.
Choudhury, S., Das, N., Ghosh, S., Ghosh, D., Chakroborty, S., & Ali, N. (2016). Vesicular (liposomal and nanoparticulated) delivery of curcumin: a comparative study on carbon tetrachloride mediated oxidative hepatocellular damage in rat model. International Journal of Nanomedicine, 11, 2179–2193.
Chen, L., Bai, G., Yang, R., Zhao, G., Xu, C., & Leung, W. (2014). Encapsulation of curcumin in recombinant human H-chain ferritin increases its water-solubility and stability. Food Research International, 62, 1147–1153.
Boorn, J., Schlee, M., Coch, C., & Hartmann, G. (2011). siRNA delivery with exosome nanoparticles. Nature Biotechnology, 29, 325–326.
Anand, P., Kunnumakkara, A., Newman, R., & Aggarwal, B. B. (2007). Bioavailability of curcumin: problems and promises. Molecular Pharmaceutics, 4, 807–818.
Lee, W., Loo, C., Bebawy, M., Luk, F., Masen, R., & Rohanizadeh, R. (2013). Curcumin and its derivatives: their application in neuropharmacology and neuroscience in the 21st century. Current Neuropharmacology, 11, 338–378.
Sutradhar, B., Khatun, S., & Luna, I. (2013). Increasing possibilities of nanosuspension. Journal of Nanotechnology, 12, 204–217.
Priyadarsini, K. I. (2014). The chemistry of curcumin: from extraction to therapeutic. Molecules, 19, 20091–20112.
Priyadarsini, K. (2013). Chemical and structural features influencing the biological activity of curcumin. Current Pharmaceutical Design, 19, 2093–2100.
Wang, Y. J., Pan, M. H., Cheng, A. L., Lin, L. I., Ho, Y. S., Hsieh, C. Y., & Lin, J. K. (1997). Stability of curcumin in buffer solutions and characterization of its degradation products. Journal of Pharmaceutical and Biomedical Analysis, 15, 1867–1876.
Tonnesen, H. H., & Karlsen, J. (1985). Studies on curcumin and curcuminoids. VI. Kinetics of curcumin degradation in aqueous solution. Zeitschrift fur Lebensmittel- Untersuchung- Forschung, 180, 402–404.
Baddela, V. S., Nayan, V., Rani, P., Onteru, S. K., & Singh, D. (2015). Physicochemical biomolecular insights into buffalo milk-derived nanovesicles. Applied Biochemistry and Biotechnology, 178, 544–577.
Hidalgo, I. J., Raub, T. J., & Borchardt, R. T. (1989). Characterization of the human colon carcinoma cell line (Caco-2) as a model system for intestinal epithelial permeability. Gastroenterology, 96, 736–749.
Meunier, V., Bourrié, M., Berger, Y., & Fabre, G. (1995). The human intestinal epithelial cell line Caco-2; pharmacological and pharmacokinetic applications. Cell Biology and Toxicology, 11, 187–194.
Wolf, T., Baier, S. R., & Zempleni, J. (2015). The intestinal transport of bovine milk exosomes is mediated by endocytosis in human colon carcinoma Caco-2 cells and rat small intestinal IEC-6 cells. Journal of Nutrition, 145, 2201–2206.
Osterman, C. J., Lynch, J. C., Leaf, P., Gonda, A., Ferguson, B. H. R., Griffiths, D., & Wall, N. R. (2015). Curcumin modulates pancreatic adenocarcinoma cell-derived exosomal function. PloS One, 10, 1–17.
Taverna, S., Fontana, S., Monteleone, F., Pucci, M., Saieva, L., De Caro, V., Cardinale, V. G., Giallombardo, M., Vicario, E., Rolfo, C., Leo, G. D., & Alessandro, R. (2016). Curcumin modulates chronic myelogenous leukemia exosomes composition and affects angiogenicphenotype via exosomal miR-21. Oncotarget, 7, 30420–30439.
Artursson, P., Palm, K., & Luthman, K. (2001). Caco-2 monolayers in experimental and theoretical predictions of drug transport. Advanced Drug Delivery Reviews, 46, 27–43.
Chen, J., Xu, T., & Chen, C. (2015). The critical roles of miR-21 in anti-cancer effects of curcumin. Annals of Translational Medicine, 3, 330.
Mudduluru, G., George-William, J. N., Muppala, S., Asangani, I. A., Kumarswamy, R., Nelson, L. D., & Allgayer, H. (2015). Curcumin regulates miR-21 expression and inhibits invasion and metastasis in colorectal cancer. Bioscience Reports, 31, 185–197.
Acknowledgments
We are thankful to the Director, NDRI, Karnal, for providing the necessary facilities for this work. We are grateful to CRP on Nanotechnology-ICAR for the support. We acknowledge Dr. S. K. Tomar for providing the SEM facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Vashisht, M., Rani, P., Onteru, S.K. et al. Curcumin Encapsulated in Milk Exosomes Resists Human Digestion and Possesses Enhanced Intestinal Permeability in Vitro. Appl Biochem Biotechnol 183, 993–1007 (2017). https://doi.org/10.1007/s12010-017-2478-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-017-2478-4