Abstract
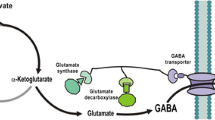
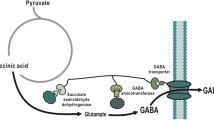
In general, gamma-aminobutyric acid (GABA) pathway involves the decarboxylation of glutamate, which is produced from sugar by Corynebacterium fermentation. GABA can be used for the production of pharmaceuticals and functional foods. Due to the increasing demand of GABA, it is essential to create an effective alternative pathway for the GABA production. In this study, Escherichia coli were engineered to produce GABA from glucose via GABA shunt, which consists of succinate dehydrogenase, succinate-semialdehyde dehydrogenase, and GABA aminotransferase. The three enzymes were physically attached to each other through a synthetic scaffold, and the Krebs cycle flux was redirected to the GABA pathway. By introduction of synthetic scaffold, 0.75 g/l of GABA was produced from 10 g/l of glucose at 30 °C and pH 6.5. The inactivation of competing metabolic pathways provided 15.4 % increase in the final GABA concentration.




Similar content being viewed by others
Abbreviations
- GABA:
-
gamma-aminobutyric acid
References
Datsenko, K. A., & Wanner, B. L. (2000). One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proceedings of the National Academy of Science of the United States of America, 97, 6640–6645.
Delebecque, C. J., Lindner, A. B., Silver, P. A., & Aldaye, F. A. (2011). Organization of intracellular reactions with rationally designed RNA assemblies. Science, 333, 470–474.
Dueber, J. E., Wu, G. C., Malmirchegini, G. R., Moon, T. S., Petzold, C. J., Ullal, A. V., Prather, K. L. J., & Keasling, J. D. (2009). Synthetic protein scaffolds provide modular control over metabolic flux. Nature Biotechnology, 27, 753–759.
Good, M. C., Zalatan, J. G. and Lim, W. A. (2011). Scaffold proteins: hubs for controlling the flow of cellular information. Science (New York, N.Y.) 332, 680–686.
Guzman, L. M., Belin, D., Carson, M. J., & Beckwith, J. (1995). Tight regulation, modulation, and high-level expression by vectors containing the arabinose PBAD promoter. Journal of Bacteriology, 177, 4121–4130.
Hirst, J., Sucheta, A., Ackrell, B. A. C., & Armstrong, F. A. (1996). Electrocatalytic voltammetry of succinate dehydrogenase: direct quantification of the catalytic properties of a complex electron-transport enzyme. Journal of the American Chemical Society, 118, 5031–5038.
Ignacio Tinoco Jr. KS, J. C. W., Joseph D. Puglisi, Gerard Harbison, David Rovnyak. (2002). Physical chemistry: principle and applications in biological sciences, 4th. Prentice Hall, Upper Saddle River, NJ.
Kook, M.-C., Seo, M.-J., Cheigh, C.-I., Lee, S.-J., Pyun, Y.-R., & Park, H. (2010). Enhancement of γ-amminobutyric acid production by Lactobacillus sakei B2–16 expressing glutamate decarboxylase from Lactobacillus plantarum ATCC 14917. Journal of the Korean Society Applied Biological Chemistry, 53, 816–820.
Le Vo, T., Ko, J.-s., Lee, S., Park, S. and Hong, S. (2013). Overexpression of Neurospora crassa OR74A glutamate decarboxylase in Escherichia coli for efficient GABA production. Biotechnology and Bioprocess Engineering, 18, 1062–1066.
Le Vo, T., Ko, J.-s., Park, S., Lee, S. and Hong, S. (2013). Efficient gamma-aminobutyric acid bioconversion by employing synthetic complex between glutamate decarboxylase and glutamate/GABA antiporter in engineered Escherichia coli. Journal of Industrial Microbiology and Biotechnology, 40, 927–933.
Lederer, T., Kintrup, M., Takahashi, M., Sum, P.-E., Ellestad, G. A., & Hillen, W. (1996). Tetracycline analogs affecting binding to Tn10-encoded Tet repressor trigger the same mechanism of induction. Biochemistry, 35, 7439–7446.
Maklashina, E., Berthold, D. A., & Cecchini, G. (1998). Anaerobic expression of Escherichia coli succinate dehydrogenase: functional replacement of fumarate reductase in the respiratory chain during anaerobic growth. Journal of Bacteriology, 180, 5989–5996.
Moon, T. S., Dueber, J. E., Shiue, E., & Prather, K. L. J. (2010). Use of modular, synthetic scaffolds for improved production of glucaric acid in engineered E. coli. Metabolic Engineering, 12, 298–305.
Oh CH, O. S. (2004). Effect of germinated brown rice extracts with enhanced levels of GABA on cancer cell proliferation and apoptosis. Journal of Medicinal Food, 7, 19.
Park, K.-B., & Oh, S.-H. (2006). Enhancement of γ-aminobutyric acid production in Chungkukjang by applying a Bacillus subtilis strain expressing glutamate decarboxylase from Lactobacillus brevis. Biotechnology Letters, 28, 1459–1463.
Park, S., Kim, E., Noh, W., Oh, Y., Kim, H., Song, B., Cho, K., Hong, S., Lee, S., & Jegal, J. (2013). Synthesis of nylon 4 from gamma-aminobutyrate (GABA) produced by recombinant Escherichia coli. Bioprocess and Biosystems Engineering, 36, 885–892.
Pham, V. D., Lee, S. H., Park, S. J., & Hong, S. H. (2015). Production of gamma-aminobutyric acid from glucose by introduction of synthetic scaffolds between isocitrate dehydrogenase, glutamate synthase and glutamate decarboxylase in recombinant Escherichia coli. Journal of Biotechnology, 207, 52–57.
Sambrook J, R. D. (2001) Molecular cloning: a laboratory manual.
Vo, T., Pham, V., Ko, J.-s., Lee, S., Park, S. and Hong, S. (2014). Improvement of gamma-amino butyric acid production by an overexpression of glutamate decarboxylase from Pyrococcus horikoshii in Escherichia coli. Biotechnology and Bioprocess Engineering, 19, 327–331.
Vo, T. D., & Hong, S. H. (2012). Optimization of gamma-Aminobutyric acid (GABA) bioconversion by recombinant Escherichia coli. Korean Society for Biotechnology and Bioengineering Journal, 27, 127–130.
Acknowledgments
This work was supported by a grant from the Next-Generation BioGreen 21 Program (SSAC, grant number: PJ01111601), Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pham, V.D., Somasundaram, S., Lee, S.H. et al. Redirection of Metabolic Flux into Novel Gamma-Aminobutyric Acid Production Pathway by Introduction of Synthetic Scaffolds Strategy in Escherichia Coli . Appl Biochem Biotechnol 178, 1315–1324 (2016). https://doi.org/10.1007/s12010-015-1948-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1948-9