Abstract
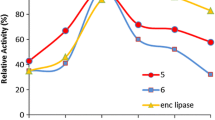
Candida rugosa lipase was immobilized with a sol–gel encapsulation procedure in the presence and absence of a calix[n]arene carboxylic acid derivative grafted onto magnetic nanoparticles or in the presence of the calix[n]arene carboxylic acid derivative with Fe3O4 magnetic nanoparticles as an additive. Through the enantioselective hydrolysis of racemic naproxen methyl ester and the hydrolysis of p-nitrophenylpalmitate, the relative enzyme activity was evaluated and tested. These results show that the encapsulated lipase without supports has lower conversion and enantioselectivity compared to the Calix[n]COOH-based encapsulated lipase. It has also been observed that the Calix[4]COOH-based encapsulated lipase has excellent enantioselectivity (enantiomeric ratio (E) > 400) as compared to encapsulated-free lipase enantioselectivity (E = 137), and it also has an enantiomeric excess value of ~98 % for S-naproxen.







Similar content being viewed by others
References
Houde, A., Kademi, A., & Leblanc, D. (2004). Applied Biochemistry and Biotechnology, 118, 155–170.
Svendsen, A. (2000). Biochimica et Biophysica Acta. (BBA), 1543, 223–238.
Lei, L., Bai, Y., Li, Y., Yi, L., Yang, Y., & Xia, C. (2009). Journal of Magnetism and Magnetic Materials, 321, 252–258.
Ranjbakhsh, E., Bordbara, A. K., Abbasi, M., Khosropour, A. R., & Shams, E. (2012). Chemical Engineering Journal, 179, 272–276.
Andrade, L. H., Rebelo, L. P., Netto, C. G. C. M., & Toma, H. E. (2010). Journal of Molecular Catalysis B: Enzymatic, 66, 55–62.
Aravindan, R., Anbumathi, P., & Viruthagiri, T. (2007). Indian Journal of Biotechnology, 6, 141–158.
Li, Y., Gaoa, F., Weia, W., Qua, J. B., Maa, G. H., & Zhoua, W. Q. (2010). Journal of Molecular Catalysis B: Enzymatic, 66, 182–189.
Yemul, O., & Imae, T. (2005). Biomacromolecules, 6, 2809–2814.
Chen, B., Hu, J., Miller, E. M., Xie, W., Cai, M., & Gross, R. A. (2008). Biomacromolecules, 9, 463–471.
Mateo, C., Abian, O., Fernandez-Lafuente, R., & Guisan, J. M. (2000). Enzyme and Microbial Technology, 26, 509–515.
Ge, J., Lu, D. N., Wang, J., & Liu, Z. (2009). Biomacromolecules, 10, 1612–1618.
Desai, P. D., Dave, A. M., & Devi, S. (2004). Journal of Molecular Catalysis B: Enzymatic, 31, 143–150.
Taqieddin, E., & Amiji, M. (2004). Biomaterials, 25, 1937–1945.
Chen, B., Miller, M. E., & Gross, R. A. (2007). Langmuir, 23, 6467–6474.
Secundo, F., Miehe-Brendle, J., Chelaru, C., Ferrandi, E. E., & Dumitriu, E. (2008). Microporous and Mesoporous Materials, 109, 350–361.
Chen, B., Miller, E. M., Miller, L., Maikner, J. J., & Gross, R. A. (2007). Langmuir, 23, 1381–1387.
Sorensen, M. H., Ng, J. B. S., Bergstrom, L., & Alberius, P. C. A. (2010). Journal of Colloid and Interface Science, 343, 359–365.
Fernandez-Lorente, G., Cabrera, Z., Godoy, C., Fernandez-Lafuente, R., Palomo, J. M., & Guisan, J. M. (2008). Process Biochemistry, 43, 1061–1067.
Palomo, J. M., Munoz, G., Fernandez-Lorente, G., Mateo, C., Fernandez-Lafuente, R., & Guisan, J. M. (2002). Journal of Molecular Catalysis B: Enzymatic, 19–20, 279–286.
Wu, Y., Wang, Y., Luo, G., & Dai, Y. (2009). Bioresource Technology, 100, 3459–3464.
Tudorache, M., Mahalu, D., Teodorescu, C., Stan, R., Bala, C., & Parvulescu, V. I. (2011). Journal of Molecular Catalysis B: Enzymatic, 69, 133–139.
Tudorache, M., Protesescu, L., Negoi, A., & Parvulescu, V. I. (2012). Applied Catalysis A: General, 437–438, 90–95.
Glad, M., Norrlow, O., Sellergren, B., Siegbahn, N., & Mosbach, K. (1985). Journal of Chromatography. A, 347, 11–23.
Venton, D. L., Cheesman, K. L., Chatterton, R. T., & Anderson, T. L. (1984). Biochimica et Biophysica Acta (BBA), 797, 343–347.
Avnir, D., Braun, S., Lev, O., & Ottolenghi, M. (1994). Chemistry of Materials, 6, 1605–1614.
Ellerby, L. M., Nishida, C. R., Nishida, F., Yamanaka, S. A., Dunn, B., Valentine, J. S., et al. (1992). Science, 255, 1113–1115.
Vidinha, P., Augusto, V., Almeida, M., Fonseca, I., Fidalgo, A., Ilharco, L., et al. (2006). Journal of Biotechnology, 121, 23–33.
Yilmaz, E., Sezgin, M., & Yilmaz, M. (2011). Journal of Molecular Catalysis B: Enzymatic, 69, 35–41.
Reetz, M. T., Tielmann, P., Wisenhofer, W., Konen, W., & Zonta, A. (2003). Advanced Synthesis and Catalysis, 345, 717–728.
Bohmer, V. (1995). Angewandte Chemie International Edition in English, 34, 713–745.
Asfari, Z., Böhmer, V., Harrowfield, J., & Vicens, J. (2001). Calixarenes 2001. Dordrecht: Kluwer.
Gokel, G. W. (1991). Crown ethers and cryptands. Cambridge: Royal Society of Chemistry.
Wenz, G. (1994). Angewandte Chemie International Edition in English, 33, 803–822.
Yilmaz, M., Memon, S., Tabakci, M., & Bartsch, R. A. (2006). In R. K. Bregg (Ed.), New frontiers in polymer research. New York: Nova Publishers.
Gutsche, C. D., Iqbal, M., & Stewart, D. (1986). The Journal of Organic Chemistry, 51, 742–745.
Sahin, O., & Yilmaz, M. (2012). Tetrahedron Letters, 53, 2319–2324.
Akceylan, E., & Yilmaz, M. (2011). Tetrahedron, 67, 6240–6245.
Sahin, O., Erdemir, S., Uyanik, A., & Yilmaz, M. (2009). Applied Catalysis A: General, 369, 36–41.
Sayin, S., Yilmaz, E., & Yilmaz, M. (2011). Organic and Biomolecular Chemistry, 9, 4021.
Wu, J. Y., & Liu, S. W. (2000). Enzyme and Microbial Technology, 26, 124–130.
Takac, S., & Bakkal, M. (2007). Process Biochemistry, 42, 1021–1027.
Gutsche, C. D., & Bauer, L. J. (1985). Journal of the American Chemical Society, 107, 6052–6059.
Arneud-Neu, F., Collins, E. M., Deasy, M., Ferguson, G., Harris, S. J., Kaitner, B., et al. (1989). Journal of the American Chemical Society, 111, 8681–8691.
Chang, S. K., & Cho, I. (1986). Journal of the Chemical Society Perkin Transactions, 1(1), 211–214.
Shinkai, S., Shiramama, Y., Satoh, H., Manabe, O., Arimura, T., Fujimato, K. & Matsuda, T. (1989). Journal of the Chemical Society, Perkin Transactions. II, 1167–1171
Ozcan, F., Ersoz, M., & Yilmaz, M. (2009). Materials Science and Engineering: C, 29, 2378–2383.
Wang, R., Bu, J., Liu, J., & Liao, S. (2008). Frontiers of Chemistry in China, 3(3), 348–352.
Chiou, S. H., & Wu, W. T. (2004). Biomaterials, 25, 197–204.
Wang, X., Dou, P., Zhao, P., Zhao, C., Ding, Y., & Xu, P. (2009). ChemSusChem, 2, 947–950.
Bradford, M. M. A. (1976). Analytical Biochemistry, 72, 248–254.
Chen, C. S., Fujimoto, Y., Girdaukas, G., & Sih, C. J. (1982). Journal of the American Chemical Society, 104, 7294–7299.
Gutsche, C. D. (1998). Calixarenes revisited. Cambridge: The Royal Society of Chemistry.
Vicens, J., & Böhmer, V. (1991). Calixarenes: a versatile class of macrocyclic compounds. Boston: Kluwer.
Yilmaz, E. (2012). Bioprocess and Biosystems Engineering, 35, 493–502.
Han, S. Y., & Kim, Y. A. (2004). Tetrahedron, 60, 2447–2467.
Reinhoudt, D.N., Eendebak, A.M., Nijenhuis, W.F., Verboom, W., Kloosterman, M., & Schoemaker, H.E.J. (1989). Journal of the Chemical Society, Chemical Communications, 399–400
Griebenow, K., Laureano, Y. D., Santos, A. M., Clemente, I. M., Rodriguez, L., Vidal, M. W., et al. (1999). Journal of the American Chemical Society, 121, 8157–8163.
Khmelnitsky, Y. L., Welch, S. H., Clark, D. S., & Dordick, J. S. (1994). Journal of the American Chemical Society, 116, 2647–2648.
Liu, Y. Y., Xu, J. H., & Hu, Y. (2000). Journal of Molecular Catalysis B: Enzymatic, 10, 523–529.
Itoh, T., Takagi, Y., Murakami, T., Hiyama, Y., & Tsukube, H. (1996). The Journal of Organic Chemistry, 61, 2158–2163.
Oshima, T., Sato, M., Shikaze, Y., Ohto, K., Inoue, K., & Baba, Y. (2007). Biochemical Engineering Journal, 35, 66–70.
Oshima, T., & Baba, Y. (2012). Journal of Inclusion Phenomena and Macrocyclic Chemistry, 73, 17–32.
Oshima, T., Suetsugu, A., Baba, Y., Shikaze, Y., Ohto, K., & Inoue, K. (2008). Journal of Membrane Science, 307, 284–291.
Uyanik, A., Sen, N., & Yilmaz, M. (2011). Bioresource Technology, 102, 4313–4318.
Barbosa, O., Ariza, C., Ortiz, C., & Torres, R. (2010). New Biotechnology, 27, 844–850.
Pereira, E. B., Castro, H. F., Moraes, F. F., & Zanin, G. M. (2001). Applied Biochemistry and Biotechnology, 91, 739–752.
Faber, K., Ottolina, G., & Riva, S. (1993). Biocatalysis, 8, 91–132.
Yilmaz, E., Sezgin, M., & Yilmaz, M. (2010). Journal of Molecular Catalysis B: Enzymatic, 62, 162–168.
Acknowledgments
We would like to thank the Scientific and Technological Research Council of Turkey (TUBITAK grant number 111T027 and CMST COST Action CM1005) and the Research Foundation of Selcuk University (BAP) for financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1003 kb)
Rights and permissions
About this article
Cite this article
Akoz, E., Akbulut, O.Y. & Yilmaz, M. Calix[n]arene Carboxylic Acid Derivatives as Regulators of Enzymatic Reactions: Enhanced Enantioselectivity in Lipase-Catalyzed Hydrolysis of (R/S)-Naproxen Methyl Ester. Appl Biochem Biotechnol 172, 509–523 (2014). https://doi.org/10.1007/s12010-013-0527-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0527-1