Abstract
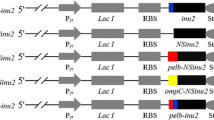
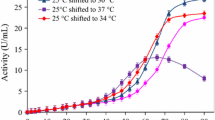
Escherichia coli is one of the most commonly used host strains for recombinant protein production. More and more research works on the production of recombinant protein indicate that extracellular production throughout a culture medium is more convenient and attractive compared to intracellular production. In present work, inducing temperature and isopropyl β-d-1-thiogalactopyranoside (IPTG) concentration were investigated to decrease the formation of inclusion body and increase the amount of soluble recombinant cutinase initially. Enzyme activity in the culture medium reached to 118.9 U/ml at 64 h of culture, and no inclusion body was detected in cytoplasm under the inducement condition of 0.2 mM IPTG and 30°C. In addition, it was found that a large amount of cutinase had been accumulated in periplasm since 16-h cultivation under the same inducement condition. Therefore, glycine and surfactant sodium taurodeoxycholate (TDOC) were further used to promote the leakage of recombinant cutinase from periplasm. Supplied with 100 mM glycine and 1 mM TDOC, the amount of cutinase in periplasm decreased remarkably, and the activity in the culture medium reached to 146.2 and 149.2 U/ml after 54 h of culturing, respectively.




Similar content being viewed by others
References
Egmond, M. R., & de Vlieg, J. (2000). Biochimie, 82, 1015–1021.
Vertommen, M. A., Nierstrasz, V. A., Veer, M., & Warmoeskerken, M. M. (2005). Journal of Biotechnology, 120, 376–386.
Alisch, M. M., Herrmann, A., & Zimmermann, W. (2006). Biotechnological Letters, 28, 681–685.
Degani, O., Gepstein, S., & Dosoretz, C. G. (2002). Applied Biochemistry and Biotechnology, 102–103, 277–289.
Sebastian, J., & Kolattukudy, P. E. (1988). Archives of Biochemistry and Biophysics, 263, 77–85.
Fett, W. F., Gerard, H. C., Moreau, R. A., Osman, S. F., & Jones, L. E. (1992). Applied and Environmental Microbiology, 58, 2123–2130.
Wang, G. Y., Michailides, T. J., Hammock, B. D., Lee, Y. M., & Bostock, R. M. (2000). Archives of Biochemistry and Biophysics, 382, 31–38.
Ferreira, B. S., Calado, C. R., van Keulen, F., Fonseca, L. P., Cabral, J. M., & da Fonseca, M. M. (2003). Applied Microbiology and Biotechnology, 61, 69–76.
Longhi, S., & Cambillau, C. (1999). Biochimica et Biophysica Acta, 1441, 185–196.
Wang, G. Y., Michailides, T. J., Hammock, B. D., Lee, Y. M., & Bostock, R. M. (2002). Fungal Genetics and Biology, 35, 261–276.
Chen, S., Tong, X., Woodard, R. W., Du, G., Wu, J., & Chen, J. (2008). The Journal of Biological Chemistry, 283, 25854–25862.
Badyakina, A. O., & Nesmeyanova, M. A. (2005). Process Biochemistry, 40, 509–518.
Jana, S., & Deb, J. K. (2005). Applied Microbiology and Biotechnology, 67, 289–298.
Mergulhao, F. J., Summers, D. K., & Monteiro, G. A. (2005). Biotechnology Advances, 23, 177–202.
Blight, M. A., Chervaux, C., & Holland, I. B. (1994). Current Opinion in Biotechnology, 5, 468–474.
Choi, J. H., & Lee, S. Y. (2004). Applied Microbiology and Biotechnology, 64, 625–635.
Jena, B. P. (2007). Current Opinion in Structural Biology, 17, 437–443.
Fett, W. F., Gerard, H. C., Moreau, R. A., Osman, S. F., & Jones, L. E. (1992). Current Microbiology, 25, 165–171.
Loh, B., Grant, C., & Hancock, R. E. (1984). Antimicrobial Agents and Chemotherapy, 26, 546–551.
Park, S. J., Georgiou, G., & Lee, S. Y. (1999). Biotechnology Progress, 15, 164–167.
Sorensen, H. P., & Mortensen, K. K. (2005). Journal of Biotechnology, 115, 113–128.
Shokri, A., Sanden, A. M., & Larsson, G. (2003). Applied Microbiology and Biotechnology, 60, 654–664.
Jin, W., & Takada, S. (2008). Journal of Molecular Biology, 377, 74–82.
Tang, J. B., Yang, H. M., Song, S. L., Zhu, P., & Ji, A. G. (2008). Food Chemistry, 108, 657–662.
Hammes, W., Schleifer, K. H., & Kandler, O. (1973). Journal of Bacteriology, 116, 1029–1053.
Lehrer, R. I., Barton, A., & Ganz, T. (1988). Immunol Methods, 108, 153–158.
Reese, E. T., & Maguire, A. (1969). Applied Microbiology, 17, 242–245.
Pugsley, A. P., Francetic, O., Possot, O. M., Sauvonnet, N., & Hardie, K. R. (1997). Gene, 192, 13–19.
Chen, S., Su, L., Billig, S., Zimmermann, W., Chen, J., & Wu, J. (2010). Journal of Molecular Catalysis. B, Enzymatic, 63, 121–127.
Yang, J., Moyana, T., MacKenzie, S., Xia, Q., & Xiang, J. (1998). Applied and Environmental Microbiology, 64, 2869–2874.
Hasenwinkle, D., Jervis, E., Kops, O., Liu, C., Lesnicki, G., Haynes, C. A., et al. (1997). Biotechnology and Bioengineering, 55, 854–863.
Acknowledgments
This work was supported by the National High-tech Research and Development Program of China (863 Program, No. 2009AA02Z204), the Major State Basic Research Development Program of China (973 Program; no. 2007CB714306), the National Natural Science Foundation of China (no. 30970057), and the Open Program of the Key Laboratory of Industrial Biotechnology, Ministry of Education (KLIB-KF200902).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, S., Liu, Z., Chen, J. et al. Study on Improvement of Extracellular Production of Recombinant Thermobifida fusca Cutinase by Escherichia coli . Appl Biochem Biotechnol 165, 666–675 (2011). https://doi.org/10.1007/s12010-011-9286-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-011-9286-z