Abstract
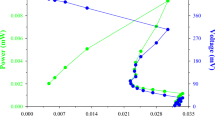
Rhamnolipid produced by Pseudomonas aeruginosa isolate Bs20 is viscous sticky oily yellowish brown liquid with a fruity odor. It showed solubility at aqueous pH > 4 with optimum solubility at pH 7–7.5 and freely soluble in ethyl acetate. This biosurfactant has a very high surface activity as it could lower the surface tension of water to 30 mN/m at about 13.4 mg/L, and it exhibited excellent stabilities at high temperatures (heating at 100°C for 1 h and autoclaving at 121°C for 10 min), salinities (up to 6% NaCl), and pH values (up to pH 13). The produced biosurfactant can be used in the crude form either as cell-free or cell-containing culture broth of the grown bacteria, since both preparations showed high emulsification indices ranged between 59% and 66% against kerosene, diesel, and motor oil. These characters make the test rhamnolipid a potential candidate for use in bioremediation of hydrocarbon-contaminated sites or in the petroleum industry. High-performance thin-layer chromatography densitometry revealed that the extracted rhamnolipid contained the two most active rhamnolipid homologues dirhamno dilipidic rhamnolipid and monorhamno dilipidic rhamnolipid at 44% and 56%, respectively, as compared to 51% and 29.5%, respectively, in a standard rhamnolipid preparation. The nature and ratio of these two rhamnolipid homologues showed to be strain dependent rather than medium-component dependent.









Similar content being viewed by others
References
Hardegger, M., Koch, T. A. K., Ochsner, U. A., Fiechter, A., & Reiser, J. (1994). Applied and Environmental Microbiology, 60, 3679–3687.
Mulligan, C. N., & Gibbs, B. F. (1993). In N. Kosaric (Ed.), Biosurfactants (pp. 339–345). New York: Marcel Dekker.
Reiser, J., Koch, A. K., Ochsner, U. A., & Fiechter, A. (1993). In N. Kosaric (Ed.), Biosurfactants: Production, properties, applications (pp. 231–249). New York, NY: Marcel Dekker.
Syldatk, C., & Wagner, F. (1987). In N. Kosaric, W. L. Cairns, & N. C. C. Gray (Eds.), Biosurfactants and biotechnology (pp. 89–120). New York: Marcel Dekker.
Mata-Sandoval, J. C., Karns, J., & Torrents, A. (1999). Journal of Chromatography. A, 864, 211–220.
Van Dyke, M. I., Couture, P., Brauer, M., Lee, H., & Trevors, J. T. (1993). Canadian Journal of Microbiology, 39, 1071–1078.
Schenk, T., Schuphan, I., & Schmidt, B. (1995). Journal of Chromatography. A, 693, 7–13.
Zhang, Y., & Miller, R. M. (1994). Applied and Environmental Microbiology, 60, 2101–2106.
Arino, S., Marchal, R., & Vandecasteele, J.-P. (1996). Applied Microbiology and Biotechnology, 45, 162–168.
Champion, J. T., Gilkey, J. C., Lamparsk, H., Retterer, J., & Miller, R. M. (1995). Journal of Colloid and Interface Science, 170, 569–574.
Zhang, Y., & Miller, R. M. (1992). Applied and Environmental Microbiology, 58, 3276–3282.
Maier, R. M., & Soberon-Chavez, G. (2000). Applied Microbiology and Biotechnology, 54, 625–633.
Abdel-Mawgoud, A. M., Aboulwafa, M. M., & Hassouna, N. A.-H. (2007). Egyptian Journal of Biotechnology, 27, 166–185.
Bodour, A. A., Drees, K. P., & Maier, R. M. (2003). Applied and Environmental Microbiology, 69, 3280–3287.
Ozdemir, G., Peker, S., & Helvaci, S. S. (2004). Colloids and Surfaces A: Physicochemical and Engineering Aspects, 234, 135–143.
Cooper, D. G., & Goldenberg, B. G. (1987). Applied and Environmental Microbiology, 53, 224–229.
Vater, J., Kablitz, B., Wilde, C., Franke, P., Mehta, N., & Cameotra, S. S. (2002). Applied and Environmental Microbiology, 68, 6210–6219.
Benincasa, M., Abalos, A., Oliveira, I., & Manresa, A. (2004). Antonie Van Leeuwenhoek, 85, 1–8.
Dubey, K., & Juwarkar, A. (2001). World Journal of Microbiology & Biotechnology, 17, 61–69.
Clifford, J. S., Ioannidis, M. A., & Legge, R. L. (2007). Journal of Colloid and Interface Science, 305, 361–365.
Fox, S. L., & Bala, G. A. (2000). Bioresource Technology, 75, 235–240.
Sen, R., & Swaminathan, T. (2005). Process Biochemistry, 40, 2953–2958.
Turkovskaya, O. V., Dmitrieva, T. V., & Muratova, A. Y. (2001). Applied Biochemistry and Microbiology, 37, 71–75.
Bonilla, M., Olivaro, C., Corona, M., Vazquez, A., & Soubes, M. (2005). Journal of Applied Microbiology, 98, 456–463.
Helvaci, S. S., Peker, S., & Ozdemir, G. (2004). Colloids and Surfaces. B, Biointerfaces, 35, 225–233.
Vogt Singer, M. E., & Finnerty, W. R. (1990). Canadian Journal of Microbiology, 36, 741–745.
Matsufuji, M., Nakata, K., & Yoshimoto, A. (1997). Biotechnology Letters, 19, 1213–1215.
Haba, E., Pinazo, A., Jauregui, O., Espuny, M. J., Infante, M. R., & Manresa, A. (2003). Biotechnology and Bioengineering, 81, 316–322.
Parkinson, M. (1985). Biotechnology Advances, 3, 65–83.
Nitschke, M., Costa, S. G. V. A. O., & Contiero, J. (2005). Biotechnology Progress, 21, 1593–1600.
Takeyama, H., Wada, M., & Matsunaga, T. (2002). Applied Biochemistry and Biotechnology, 98–100, 319–326.
Bognolo, G. (1999). Colloids and Surfaces A: Physicochemical and Engineering Aspects, 152, 41–52.
Desai, J. D., & Banat, I. M. (1997). Microbiology and Molecular Biology Reviews, 61, 47–64.
Acknowledgments
The authors would like to thank Dr. Salam M. Awada (AgSciTech, Logan, UT, USA) for kindly supplying the standard rhamnolipid and Dr. Anas M. Abdel-Mawgoud (ARCMP, NODCAR, Ministry of Health, Cairo, Egypt) for technical support and help in HPTLC experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Mawgoud, A.M., Aboulwafa, M.M. & Hassouna, N.AH. Characterization of Rhamnolipid Produced by Pseudomonas aeruginosa Isolate Bs20. Appl Biochem Biotechnol 157, 329–345 (2009). https://doi.org/10.1007/s12010-008-8285-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8285-1