Abstract
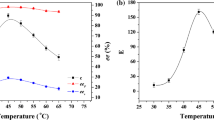
The effect of chain length on the enzymatic hydrolysis of various p-nitrophenyl esters was investigated. Specifically, the hydrolysis of various esters p-nitrophenyl butyrate (PNPB), p-nitrophenyl caprylate (PNPC), p-nitrophenyl laurate (PNPL), p-nitrophenyl myristate (PNPM) and p-nitrophenyl palmitate (PNPP) was studied in supercritical carbon dioxide (ScCO2) with lipase (Novozym 435). This indicates that the conversion of nitrophenyl esters decreases with increasing chain length. The effect of various parameters such as amount of water added, temperature, and enzyme loading was studied. The optimum temperature for the hydrolysis of PNPB and PNPC was 50°C but was 55°C for PNPL, PNPM, and PNPP in ScCO2. The reactions were also conducted in acetonitrile as the solvent, and it was found that the reactions reach equilibrium much faster in ScCO2 than in acetonitrile. The kinetics of the hydrolysis reactions were modeled using a Ping Pong Bi Bi model.





Similar content being viewed by others
References
Nini, L., Sarda, L., Comeau, L. C., Boitard, E., Dubes, J. P., & Chahinian, H. (2001). Biochimica et Biophysica Acta, 1534, 34–44.
Vaysse, L., Aboubakry, L., Moulin, G., & Dubreucq, E. (2002). Enzyme and Microbial Technology, 31, 648–655.
Pencreach, G., & Baratti, J. C. (1996). Enzyme and Microbial Technology, 18, 417–422.
Yadav, G. D., & Manjula Devi, K. (2004). Biochemical Engineering Journal, 17, 57–63.
Burdette, R. A., & Quinn, D. M. (1986). Journal of Biological Chemistry, 261, 12016–12021.
Charton, E., & Macrae, A. R. (1993). Enzyme and Microbial Technology, 15, 489–493.
Hui, Y., Wang, S., & Jiang, X. (1982). Journal of the American Chemical Society, 104, 347–349.
Gupta, M. N. (1992). European Journal of Biochemistry, 203, 25–32.
Harikrishna, S., Manohar, B., Divakar, S., Prapulla, S. G., & Karanth, N. G. (2000). Enzyme and Microbial Technology, 26, 131–136.
Chulalaksananukul, W., Condoret, J. S., & Combes, D. (1992). Enzyme and Microbial Technology, 14, 293–298.
Klibanov, A. M. (1989). Trends in Biochemical Sciences, 14, 141–144.
Turner, C., Persson, M., Mathiasson, L., Adlercreutz, P., & King, J. W. (2001). Enzyme and Microbial Technology, 29, 111–121.
Sovova, H., & Ucka, M. Z. (2003). Chemical Engineering Science, 58, 2339–2350.
Nagesha, G. K., Manohar, B., & Udaya Sankar, K. (2004). Journal of Supercritical Fluids, 32, 137–145.
Srivastava, S., & Madras, G. (2001). Journal of Chemical Technology and Biotechnology, 76, 890–892.
Matsuda, T., Harada, T., Nakamura, K., & Ikariya, T. (2005). Tetrahedron: Asymmetry, 16, 909–915.
Kumar, R., Modak, J., & Madras, G. (2005). Industrial & Engineering Chemistry Research, 43, 1568–1573.
Nakamura, K., Chi, Y. M., Yamada, Y., & Yano, T. (1985). Chemical Engineering Communications, 45, 207–212.
Srivastava, S., Modak, J., & Madras, G. (2002). Industrial & Engineering Chemistry Research, 41, 1940–1945.
Karel, B. E., & Klibanov, A. M. (1989). Biotechnology and Bioengineering, 34, 1178–1185.
Parvaresh, F., Robert, H., Thomas, D., & Legoy, M. D. (1992). Biotechnology and Bioengineering, 39, 467–473.
Morild, E. (1981). Advances in Protein Chemistry, 34, 93–166.
Lawin, L. R., Fife, W. K., & Tian, C. X. (2000). Langmuir, 16, 3583–3587.
Kodaka, M. (1983). Bulletin of the Chemical Society of Japan, 56, 191–2192.
Ishiwatari, T., & Fendler, H. (1994). Journal of the American Chemical Society, 106, 1908–1912.
Menger, F. M., & Portnoy, C. E. (1967). Journal of the American Chemical Society, 901, 1875–1878.
Tee, O. S., & Enos, J. A. (1988). Canadian Journal of Chemistry, 66, 3027–3030.
Yang, J., Koga, Y., Hideo, N., & Tsuneo, Y. (2002). Protein Engineering, 15, 147–152.
Hayashi, T., & Ikada, Y. (1990). Biotechnology and Bioengineering, 36, 593–600.
Macnaughton, S. J., & Foster, N. R. (1994). Industrial & Engineering Chemistry Research, 33, 2757–2763.
Chapoy, A., Mohammadi, A. H., Chareton, A., Tohidi, B., & Richon, D. (2004). Industrial & Engineering Chemistry Research, 43, 1794–1802.
Perrut, M. (1994). Chemical and Biochemical Engineering Quarterly, 8, 25–30.
Condoret, J.-S., Vankan, S., Joullia, X., & Marty, A. (1997). Chemical Engineering Science, 52, 213–220.
Srivastava, S., Modak, J., & Madras, G. (2003). Journal of Supercritical Fluids, 27, 55–64.
Almeida, M. C., Ruivo, R., Maia, C., Freire, L., Sampaio, T., & Barreiros, S. (2000). Enzyme and Microbial Technology, 22, 494–499.
Martins, J. F., Carvalho, I. B., Sampaio, T. C., & Barreiros, S. (1994). Enzyme and Microbial Technology, 16, 785–789.
Goddard, R., Bosley, J., & Al-Duri, B. (2000). Journal of Supercritical Fluids, 18, 121–130.
Dumont, T., Barth, D., Corbier, C., & Branlant, G. (1992). Biotechnology and Bioengineering, 40, 329–334.
Chrastill, J. (1982). Journal of Physical Chemistry, 86, 3016–3021.
Nag, A. (1988). Journal of Surfaces Science and Technology, 4, 91–96.
Shishikura, A., Fujimoto, K., Suzuki, T., & Arai, K. (1994). Journal of the American Oil Chemists’ Society, 71, 961–967.
Romero, M. D., Calvo, L., Alba, C., Habulin, M., Primožič, M., & Knez, Ž. (2005). The Journal of Supercritical Fluids, 33, 77–84.
Olsen, T., Kerton, F., Marriott, R., & Grogan, G. (2006). Enzyme and Microbial Technology, 39, 621.
Gubicza, L., Kabiri-Badr, A., Deoves, E., & Belafi-Bako, K. (2000). Journal of Biotechnology, 84, 193.
Güvenç, A., Kapucu, N., & Mehmetoğlu, Ü. (2002). Process Biochemistry, 10, 1–6.
Yadav, G. D., & Lathi, P. S. (2005). Journal of molecular catalysis. B, Enzymatic, 32, 107.
Chulalaksananukul, W., Condoret, J. S., Delorme, P., & Willemot, R. M. (1990). FEBS Letters, 276, 181–184.
Chulalaksananukul, S., Longo, M. A., Chulalaksananukul, W., Condoret, J.-S., & Combes, D. (1999). AFINIDAD LVI, 480, 121–125.
Valivety, R. H., Halling, P. J., & Macrae, A. R. (1993). Biotechnology Letters, 15, 1133–1138.
Lai, D. T., Hattori, N., & O’Connor, C. J. (1999). Journal of the American Oil Chemists’ Society, 76, 845–851.
Rizzi, M., Stylos, P., Riek, A., & Reuss, M. (1992). Enzyme and Microbial Technology, 14, 709–714.
Straathof, A. J. J., Rakels, J. L. L., Heijnen, J. J. (1992). In J. Tramper, M. H. Vermuë, H. H. Beeftink, U. von Stockar (Eds.), (pp. 137–144). Amsterdam: Elsevier.
Valivety, R. H., Halling, P. J., Peilow, A. D., & Macrae, A. R. (1992). Biochimica et Biophysica Acta, 1122, 143–146.
Martinelle, M., & Hult, K. (1995). Biochimica et Biophysica Acta, 125, 191–197.
Chulalaksananukul, W., Condoret, J. S., & Combes, D. (1993). Enzyme and Microbial Technology, 15, 691–698.
Goddard, R., Bosley, J., & Al-Duri, B. (2000). Journal of Chemical Technology and Biotechnology, 75, 715–721.
Goddard, R., Bosley, J., & Al-Duri, B. (2000). Journal of Supercritical Fluids, 18, 121–130.
Paiva, A. V., Rossum, D. V., & Malcasa, F. X. (2002). Biocatalysis and Biotransformation, 20, 43.
Goddard, R., Bosley, J., & Al-Duri, B. (2000). Journal of Supercritical Fluids, 18, 121.
Hernandez, F. J., De los Rios, A. P., Gomez, D., Rubio, M., & Villora, G. (2006). Applied Catalysis B, Environmental, 67, 121.
Marty, A., Chulalaksananukul, W., Willemot, R. M., & Condore, J. S. (1992). Biotechnology and Bioengineering, 39, 273–280.
Rizzi, M., Stylos, P., Riek, A., & Reuss, M. (1992). Enzyme and Microbial Technology, 14, 709–714.
Varma, M. N., & Madras, G. (2007). Industrial & Engineering Chemistry Research, 46, 1–6.
Yu, Z. R., Rizvi, S. S. H., & Zollweg, J. A. (1992). Biotechnology Progress, 8, 508.
Knez, Z., & Habulin, M. (1992). Progress in Biotechnology, 8, 401.
Knez, Z., & Habulin, M. (1994). Biocatalysis, 9, 115.
Tewari, Y. B., Hara, T., Phinney, K. W., & Mayhew, M. P. (2004). Journal of Molecular Catalysis. B, Enzymatic, 30, 131.
Kamat, S., Critchley, G., Beckman, E. J., & Russell, A. J. (1995). Biotechnology and Bioengineering, 46, 610.
Vermue, M. H., Tramper, J., Dejong, J. P. J., & Oostrom, W. H. M. (1992). Enzyme and Microbial Technology, 14, 649.
Vija, H., Telling, A., & Tougu, V. (1997). Bioorganic & Medicinal Chemistry Letters, 7, 259.
Paizs, C., Tosa, M., Bodai, V., Szakacs, G., Kmecz, I., Simandi, B., Majdik, C., Novak, L., Irimie, F. D., & Poppe, L. (2003). Tetrahedron: Asymmetry, 14, 1943.
Holmes, J. D., Steytler, D. C., Rees, G. D., & Robinson, B. H. (1998). Langmuir, 14, 6371.
Rantakyla, M., & Aaltonen, O. (1994). Biotechnology Letters, 16, 825.
Acknowledgements
The authors thank the Department of Biotechnology, India for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varma, M.N., Madras, G. Effect of Chain Length on Enzymatic Hydrolysis of p-Nitrophenyl Esters in Supercritical Carbon Dioxide. Appl Biochem Biotechnol 144, 213–223 (2008). https://doi.org/10.1007/s12010-007-8114-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-007-8114-y