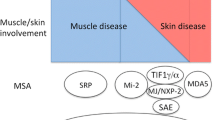
Abstract
Myositis autoantibodies continue to be the subject of substantial interest, with several significant recent developments. Recent studies have emphasized that anti-Jo-1 or other antisynthetases can be associated with interstitial lung disease, even in the absence of myositis. Anti-Jo-1 autoantibody levels were shown to correlate with disease activity over time. Immunization of mice with anti-Jo-1 led to muscle and lung inflammation, reminiscent of human disease. The complexity of the antibody picture in myositis continues to increase. Several new autoantibodies have been described or better characterized. Among these were a new antisynthetase reactive with phenylalanyl-transfer RNA synthetase (anti-Zo); an autoantibody that immunoprecipitates 155-and 140-kD proteins and was common in children but seemed to be associated with malignancy in adults; and an autoantibody to a small ubiquitin-like modifier-activating enzyme associated with dermatomyositis and interstitial lung disease. These findings have significant clinical implications and suggest promising areas of further research.
Similar content being viewed by others
References and Recommended Reading
Reichlin M, Mattioli M: Description of a serological reaction characteristic of polymyositis. Clin Immunol Immunopathol 1976, 5: 12–20.
Targoff IN: Myositis specific autoantibodies. Curr Rheumatol Rep 2006, 8: 196–203.
Targoff IN: Laboratory testing in the diagnosis and management of idiopathic inflammatory myopathies. Rheum Dis Clin North Am 2002, 28: 859–890.
Marie I, Hachulla E, Cherin P, et al.: Interstitial lung disease in polymyositis and dermatomyositis. Arthritis Rheum 2002, 47: 614–622.
Fathi M, Vikgren J, Boijsen M, et al.: Interstitial lung disease in polymyositis and dermatomyositis: longitudinal evaluation by pulmonary function and radiology. Arthritis Care Res 2008, 59: 677–685.
Yoshifuji H, Fujii T, Kobayashi S, et al.: Anti-aminoacyl-tRNA synthetase antibodies in clinical course prediction of interstitial lung disease complicated with idiopathic inflammatory myopathies. Autoimmun 2006, 39: 233–241.
Matsushita T, Hasegawa M, Fujimoto M, et al.: Clinical evaluation of anti-aminoacyl tRNA synthetase antibodies in Japanese patients with dermatomyositis. J Rheumatol 2007, 34: 1012–1018.
Friedman AW, Targoff IN, Arnett FC: Interstitial lung disease with autoantibodies against aminoacyl-tRNA synthetases in the absence of clinically apparent myositis. Semin Arthritis Rheum 1996, 26: 459–467.
Sato S, Kuwana M, Hirakata M: Clinical characteristics of Japanese patients with anti-OJ (anti-isoleucyl-tRNA synthetase) autoantibodies. Rheumatology (Oxford) 2007, 46: 842–845.
Hirakata M, Suwa A, Takada T, et al.: Clinical and immunogenetic features of patients with autoantibodies to asparaginyl-transfer RNA synthetase. Arthritis Rheum 2007, 56: 1295–1303.
Tillie-Leblond I, Wislez M, Valeyre D, et al.: Interstitial lung disease and anti-Jo-1 antibodies: difference between acute and gradual onset. Thorax 2008, 63: 53–59.
Schmidt WA, Wetzel W, Friedlander R, et al.: Clinical and serological aspects of patients with anti-Jo-1 antibodies-an evolving spectrum of disease manifestations. Clin Rheumatol 2000, 19: 371–377.
Douglas WW, Tazelaar HD, Hartman TE, et al.: Polymyositisdermatomyositis-associated interstitial lung disease. Am J Respir Crit Care Med 2001, 164: 1182–1185.
Selva-O’Callaghan A, Labrador-Horrillo M, Solans-Laque R, et al.: Myositis-specific and myositis-associated antibodies in a series of eighty-eight Mediterranean patients with idiopathic inflammatory myopathy. Arthritis Rheum 2006, 55: 791–798.
Okada S, Weatherhead E, Targoff IN, et al.: Global surface ultraviolet radiation intensity may modulate the clinical and immunologic expression of autoimmune muscle disease. Arthritis Rheum 2003, 48: 2285–2293.
Stone KB, Oddis CV, Fertig N, et al.: Anti-Jo-1 antibody levels correlate with disease activity in idiopathic inflammatory myopathy. Arthritis Rheum 2007, 56: 3125–3131.
Klein RQ, Teal V, Taylor L, et al.: Number, characteristics, and classification of patients with dermatomyositis seen by dermatology and rheumatology departments at a large tertiary medical center. J Am Acad Dermatol 2007, 57: 937–943.
Legault D, McDermott J, Crous-Tsanaclis AM, Boire G: Cancer-associated myositis in the presence of anti-Jo1 autoantibodies and the antisynthetase syndrome. J Rheumatol 2008, 35: 169–171.
Respicio G, Shwaiki W, Abeles M: A 58-year-old man with anti-Jo-1 syndrome and renal cell carcinoma: a case report and discussion. Conn Med 2007, 71: 151–153.
Buchbinder R, Forbes A, Hall S, et al.: Incidence of malignant disease in biopsy-proven inflammatory myopathy. A population-based cohort study. Ann Intern Med 2001, 134: 1087–1095.
Sigurgeirsson B, Lindelof B, Edhag O, Allander E: Risk of cancer in patients with dermatomyositis or polymyositis: A population-based study. N Engl J Med 1992, 326: 363–367.
Mogulkoc N, Kabasakal Y, Ekren PK, Bishop PW: An unusual presentation of anti-Jo-1 syndrome, mimicking lung metastases, with massive pleural and pericardial effusions. J Clin Rheumatol 2006, 12: 90–92.
Miller FW, Twitty SA, Biswas T, Plotz PH: Origin and regulation of a disease-specific autoantibody response: Antigenic epitopes, spectrotype stability, and isotype restriction of anti-Jo-1 antibodies. J Clin Invest 1990, 85: 468–475.
Katsumata Y, Ridgway WM, Oriss T, et al.: Species-specific immune responses generated by histidyl-tRNA synthetase immunization are associated with muscle and lung inflammation. J Autoimmun 2007, 29: 174–186.
Nagaraju K, Raben N, Loeffler L, et al.: Conditional up-regulation of MHC class I in skeletal muscle leads to self-sustaining autoimmune myositis and myositis-specific autoantibodies. Proc Natl Acad Sci U S A 2000, 97: 9209–9214.
Levine SM, Raben N, Xie D, et al.: Novel conformation of histidyl-transfer RNA synthetase in the lung: the target tissue in Jo-1 autoantibody-associated myositis. Arthritis Rheum 2007, 56: 2729–2739.
Casciola-Rosen L, Andrade F, Ulanet D, et al.: Cleavage by granzyme B is strongly predictive of autoantigen status: implications for initiation of autoimmunity. J Exp Med 1999, 190: 815–826.
Casciola-Rosen L, Nagaraju K, Plotz P, et al.: Enhanced autoantigen expression in regenerating muscle cells in idiopathic inflammatory myopathy. J Exp Med 2005, 201: 591–601.
Yamasaki Y, Yamada H, Nozaki T, et al.: Unusually high frequency of autoantibodies to PL-7 associated with milder muscle disease in Japanese patients with polymyositis/dermatomyositis. Arthritis Rheum 2006, 54: 2004–2009.
Targoff IN, Trieu EP, Miller FW: Reaction of anti-OJ autoantibodies with components of the multi-enzyme complex of aminoacyl-tRNA synthetases in addition to isoleucyl-tRNA synthetase. J Clin Invest 1993, 91: 2556–2564.
Betteridge Z, Gunawardena H, North J, et al.: Anti-synthetase syndrome: a new autoantibody to phenylalanyl transfer RNA synthetase (anti-Zo) associated with polymyositis and interstitial pneumonia. Rheumatology (Oxford) 2007, 46: 1005–1008.
Kao AH, Lacomis D, Lucas M, et al.: Anti-signal recognition particle autoantibody in patients with and patients without idiopathic inflammatory myopathy. Arthritis Rheum 2004, 50: 209–215.
Miller T, Al Lozi MT, Lopate G, Pestronk A: Myopathy with antibodies to the signal recognition particle: clinical and pathological features. J Neurol Neurosurg Psychiatry 2002, 73: 420–428.
Hengstman GJ, ter Laak HJ, Vree Egberts WT, et al.: Anti-signal recognition particle autoantibodies: marker of a necrotising myopathy. Ann Rheum Dis 2006, 65: 1635–1638.
Dimitri D, Andre C, Roucoules J, et al.: Myopathy associated with anti-signal recognition peptide antibodies: clinical heterogeneity contrasts with stereotyped histopathology. Muscle Nerve 2007, 35: 389–395.
Reichlin M, Arnett FC: Multiplicity of antibodies in myositis sera. Arthritis Rheum 1984, 27: 1150–1156.
Bohan A, Peter JB: Polymyositis and dermatomyositis. Parts 1 and 2. N Engl J Med 1975, 292: 344–347; 403–407.
Rutjes SA, Vree Egberts WT, Jongen P, et al.: Anti-Ro52 antibodies frequently co-occur with anti-Jo-1 antibodies in sera from patients with idiopathic inflammatory myopathy. Clin Exp Immunol 1997, 109: 32–40.
Frank MB, McCubbin VR, Trieu EP, et al.: The association of anti-Ro52 autoantibodies with myositis and scleroderma autoantibodies. J Autoimmun 1999, 12: 137–142.
Troyanov Y, Targoff IN, Tremblay JL, et al.: Novel classification of idiopathic inflammatory myopathies based on overlap syndrome features and autoantibodies: analysis of 100 French Canadian patients. Medicine 2005, 84: 231–249.
Koenig M, Fritzler MJ, Targoff IN, et al.: Heterogeneity of autoantibodies in 100 patients with autoimmune myositis: insights into clinical features and outcomes. Arthritis Res Ther 2007, 9: R78.
Steen VD: Autoantibodies in systemic sclerosis. Semin Arthritis Rheum 2005, 35: 35–42.
Kaji K, Fujimoto M, Hasegawa M, et al.: Identification of a novel autoantibody reactive with 155 and 140 kDa nuclear proteins in patients with dermatomyositis: an association with malignancy. Rheumatology 2007, 46: 25–28.
Targoff IN, Mamyrova G, Trieu EP, et al.: A novel autoantibody to a 155-kd protein is associated with dermatomyositis. Arthritis Rheum 2006, 54: 3682–3689.
Chinoy H, Fertig N, Oddis CV, et al.: The diagnostic utility of myositis autoantibody testing for predicting the risk of cancer-associated myositis. Ann Rheum Dis 2007, 66: 1345–1349.
Gunawardena H, Wedderburn LR, North J, et al.: Clinical associations of autoantibodies to a p155/140 kDa doublet protein in juvenile dermatomyositis. Rheumatology (Oxford) 2008, 47: 324–328.
Betteridge Z, Gunawardena H, North J, et al.: Identification of a novel autoantibody directed against small ubiquitin-like modifier activating enzyme in dermatomyositis. Arthritis Rheum 2007, 56: 3132–3137.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Targoff, I.N. Autoantibodies and their significance in myositis. Curr Rheumatol Rep 10, 333–340 (2008). https://doi.org/10.1007/s11926-008-0053-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11926-008-0053-2