Abstract
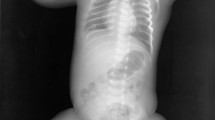
Osteogenesis imperfecta (OI) is a rare disorder of type 1 collagen with 13 currently identified types attributable to inherited abnormalities in type 1 collagen amount, structure, or processing. The disease is characterized by an increased susceptibility to bony fracture. In addition to the skeletal phenotype, common additional extraskeletal manifestations include blue sclerae, dentinogenesis imperfecta, vascular fragility, and hearing loss. Medical management is focused on minimizing the morbidity of fractures, pain, and bone deformities by maximizing bone health. Along with optimizing Vitamin D status and calcium intake and physical/occupational therapy, individualized surgical treatment may be indicated. Pharmacological therapy with bisphosphonate medications is now routinely utilized for moderate to severe forms and appears to have a good safety profile and bone health benefits. New therapies with other anti-resorptives as well as anabolic agents and transforming growth factor (TGF)β antibodies are in development. Other potential treatment modalities could include gene therapy or mesenchymal cell transplant. In the future, treatment choices will be further individualized in order to reduce disease morbidity and mortality.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Stevenson DA, Carey JC, Byrne JL, Srisukhumbowornchai S, Feldkamp ML. Analysis of skeletal dysplasias in the Utah population. Am J Med Genet A. 2012;158A(5):1046–54. doi:10.1002/ajmg.a.35327.
Sillence DO, Senn A, Danks DM. Genetic heterogeneity in osteogenesis imperfecta. J Med Genet. 1979;16(2):101–16.
Van Dijk FS, Sillence DO. Osteogenesis imperfecta: clinical diagnosis, nomenclature and severity assessment. Am J Med Genet A. 2014;164(6):1470–81. doi:10.1002/ajmg.a.36545. The Sillence classification labeled four different types of OI based on phenotype. As more types of OI have been identified, additional categories were created based on the genetic mutation. With this reclassification, the different OI syndromes will now be classified again by overarching categories which will compliment the genetic mutation and provide phenotype information.
Warman ML, Cormier-Daire V, Hall C, Krakow D, Lachman R, LeMerrer M, et al. Nosology and classification of genetic skeletal disorders: 2010 revision. Am J Med Genet A. 2011;155a(5):943–68. doi:10.1002/ajmg.a.33909.
Bodian DL, Chan T-F, Poon A, Schwarze U, Yang K, Byers PH, et al. Mutation and polymorphism spectrum in osteogenesis imperfecta type II: implications for genotype–phenotype relationships. Hum Mol Genet. 2009;18(3):463–71. doi:10.1093/hmg/ddn374.
Harrington J, Sochett E, Howard A. Update on the evaluation and treatment of osteogenesis imperfecta. Pediatr Clin North Am. 2014;61(6):1243–57. doi:10.1016/j.pcl.2014.08.010.
Shapiro JR, Lietman C, Grover M, Lu JT, Nagamani SCS, Dawson BC, et al. Phenotypic variability of osteogenesis imperfecta type V caused by an IFITM5 mutation. J Bone Miner Res. 2013;28(7):1523–30. doi:10.1002/jbmr.1891. Further information about the phenotype and its variability about this rare type of OI is presented.
Marini JC, Blissett AR. New genes in bone development: what’s new in osteogenesis imperfecta. J Clin Endocrinol Metab. 2013;98(8):3095–103. doi:10.1210/jc.2013-1505.
Semler O, Cheung MS, Glorieux FH, Rauch F. Wormian bones in osteogenesis imperfecta: correlation to clinical findings and genotype. Am J Med Genet A. 2010;152A(7):1681–7. doi:10.1002/ajmg.a.33448.
Ben Amor IM, Glorieux FH, Rauch F. Genotype-phenotype correlations in autosomal dominant osteogenesis imperfecta. J Osteoporos. 2011;2011:540178. doi:10.4061/2011/540178.
Evereklioglu C, Madenci E, Bayazıt YA, Yılmaz K, Balat A, Bekir NA. Central corneal thickness is lower in osteogenesis imperfecta and negatively correlates with the presence of blue sclera. Ophthalmic Physiol Opt. 2002;22(6):511–5. doi:10.1046/j.1475-1313.2002.00062.x.
Dimasi D, Chen J, Hewitt A, Klebe S, Davey R, Stirling J, et al. Novel quantitative trait loci for central corneal thickness identified by candidate gene analysis of osteogenesis imperfecta genes. Hum Genet. 2010;127(1):33–44. doi:10.1007/s00439-009-0729-3.
Chau FY, Wallace D, Vajaranant T, Herndon L, Lee P, Challa P, et al. Chapter 31—osteogenesis imperfecta and the eye. In: Sponseller JRSHBHGD, editor. Osteogenesis Imperfecta. San Diego: Academic; 2014. p. 289–303.
O’Connell AC, Marini JC. Evaluation of oral problems in an osteogenesis imperfecta population. Oral Surg, Oral Med, Oral Pathol, Oral Radiol Endod. 1999;87(2):189–96. doi:10.1016/S1079-2104(99)70272-6.
Santos F, McCall AA, Chien W, Merchant S. Otopathology in osteogenesis imperfecta. Otol Neurotol. 2012;33(9):1562–6. doi:10.1097/MAO.0b013e31826bf19b.
Paterson CR, Monk EA, McAllion SJ. How common is hearing impairment in osteogenesis imperfecta? J Laryngol Otol. 2001;115(4):280–2.
Bonita RE, Cohen IS, Berko BA. Valvular heart disease in osteogenesis imperfecta: presentation of a case and review of the literature. Echocardiography. 2010;27(1):69–73. doi:10.1111/j.1540-8175.2009.00973.x.
Hansen B, Jemec GB. The mechanical properties of skin in osteogenesis imperfecta. Arch Dermatol. 2002;138(7):909–11.
Marini J. Osteogenesis imperfecta. In: De Groot LJ, Beck-Peccoz P, Chrousos G, Dungan K, Grossman A, Hershman JM, et al., editors. Endotext. South Dartmouth: MDText.com, Inc.; 2000.
LoMauro A, Pochintesta S, Romei M, D’Angelo MG, Pedotti A, Turconi AC, et al. Rib cage deformities alter respiratory muscle action and chest wall function in patients with severe osteogenesis imperfecta. PLoS ONE. 2012;7(4), e35965. doi:10.1371/journal.pone.0035965.
Sillence DO. Craniocervical abnormalities in osteogenesis imperfecta: genetic and molecular correlation. Pediatr Radiol. 1994;24(6):427–30.
Ibrahim AG, Crockard HA. Basilar impression and osteogenesis imperfecta: a 21-year retrospective review of outcomes in 20 patients. J Neurosurg Spine. 2007;7(6):594–600. doi:10.3171/spi-07/12/594.
Vetter U, Pontz B, Zauner E, Brenner RE, Spranger J. Osteogenesis imperfecta: a clinical study of the first ten years of life. Calcif Tissue Int. 1992;50:36–41.
Alanay Y, Avaygan H, Camacho N, Utine GE, Boduroglu K, Aktas D, et al. Mutations in the gene encoding the RER protein FKBP65 cause autosomal-recessive osteogenesis imperfecta. Am J Hum Genet. 2010;86(4):551–9. doi:10.1016/j.ajhg.2010.02.022.
Puig-Hervás MT, Temtamy S, Aglan M, Valencia M, Martínez-Glez V, Ballesta-Martínez MJ, et al. Mutations in PLOD2 cause autosomal-recessive connective tissue disorders within the Bruck syndrome—osteogenesis imperfecta phenotypic spectrum. Hum Mutat. 2012;33(10):1444–9. doi:10.1002/humu.22133.
Rauch F, Fahiminiya S, Majewski J, Carrot-Zhang J, Boudko S, Glorieux F, et al. Cole-Carpenter syndrome is caused by a heterozygous missense mutation in P4HB. Am J Hum Genet. 2015;96(3):425–31. doi:10.1016/j.ajhg.2014.12.027.
Garbes L, Kim K, Riess A, Hoyer-Kuhn H, Beleggia F, Bevot A, et al. Mutations in SEC24D, encoding a component of the COPII machinery, cause a syndromic form of osteogenesis imperfecta. Am J Hum Genet. 2015;96(3):432–9. doi:10.1016/j.ajhg.2015.01.002.
Bianchi ML. Hypophosphatasia: an overview of the disease and its treatment. Osteoporos Int: J Established Result Coop Between Eur Foundation Osteoporos Natl Osteoporos Foundation USA. 2015. doi:10.1007/s00198-015-3272-1.
Gong Y, Slee RB, Fukai N, Rawadi G, Roman-Roman S, Reginato AM, et al. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell. 2001;107(4):513–23.
Okajima T, Fukumoto S, Furukawa K, Urano T, Furukawa K. Molecular basis for the progeroid variant of Ehlers-Danlos Syndrome: identification and characterization of two mutations in galactosyltransferase I gene. J Biol Chem. 1999;274(41):28841–4. doi:10.1074/jbc.274.41.28841.
Greeley CS, Donaruma-Kwoh M, Vettimattam M, Lobo C, Williard C, Mazur L. Fractures at diagnosis in infants and children with osteogenesis imperfecta. J Pediatr Orthop. 2013;33(1):32–6. doi:10.1097/BPO.0b013e318279c55d.
Whyte MP, Obrecht SE, Finnegan PM, Jones JL, Podgornik MN, McAlister WH, et al. Osteoprotegerin deficiency and juvenile Paget’s disease. N Engl J Med. 2002;347(3):175–84. doi:10.1056/NEJMoa013096.
Osteogenesis Imperfecta Foundation. OI Issues: Dental Care for Persons with OI. . http://www.oif.org/site/PageServer?pagename=Dental. Accessed 23 Sept 2015.
Dwan K, Phillipi Carrie A, Steiner Robert D, Basel D. Bisphosphonate therapy for osteogenesis imperfecta. Cochrane Database Syst Rev. 2014. doi:10.1002/14651858.CD005088.pub3.
Bishop N, Adami S, Ahmed SF, Anton J, Arundel P, Burren CP, et al. Risedronate in children with osteogenesis imperfecta: a randomised, double-blind, placebo-controlled trial. Lancet. 2013;382(9902):1424–32. doi:10.1016/S0140-6736(13)61091-0. This paper demonstrating benefits of oral risedronate therapy on bone mineral density and fracture rates re-opened the door to use of oral bisphosphonate for children with milder OI.
Munns CF, Rajab MH, Hong J, Briody J, Hogler W, McQuade M, et al. Acute phase response and mineral status following low dose intravenous zoledronic acid in children. Bone. 2007;41(3):366–70. doi:10.1016/j.bone.2007.05.002.
Munns CF, Rauch F, Mier RJ, Glorieux FH. Respiratory distress with pamidronate treatment in infants with severe osteogenesis imperfecta. Bone. 2004;35(1):231–4.
Munns CF, Rauch F, Zeitlin L, Fassier F, Glorieux FH. Delayed osteotomy but not fracture healing in pediatric osteogenesis imperfecta patients receiving pamidronate. J Bone Miner Res. 2004;19(11):1779–86.
Rauch F, Munns C, Land C, Glorieux FH. Pamidronate in children and adolescents with osteogenesis imperfecta: effect of treatment discontinuation. J Clin Endocrinol Metab. 2006;91(4):1268–74. doi:10.1210/jc.2005-2413.
Shapiro JR, Thompson CB, Wu Y, Nunes M, Gillen C. Bone mineral density and fracture rate in response to intravenous and oral bisphosphonates in adult osteogenesis imperfecta. Calcif Tissue Int. 2010;87(2):120–9. doi:10.1007/s00223-010-9383-y.
Bargman R, Posham R, Boskey AL, DiCarlo E, Raggio C, Pleshko N. Comparable outcomes in fracture reduction and bone properties with RANKL inhibition and alendronate treatment in a mouse model of osteogenesis imperfecta. Osteoporos Int. 2012;23(3):1141–50. doi:10.1007/s00198-011-1742-7.
Semler O, Netzer C, Hoyer-Kuhn H, Becker J, Eysel P, Schoenau E. First use of the RANKL antibody denosumab in osteogenesis imperfecta type VI. J Musculoskelet Neuronal Interact. 2012;12(3):183–8.
Antoniazzi F, Bertoldo F, Mottes M, Valli M, Sirpresi S, Zamboni G, et al. Growth hormone treatment in osteogenesis imperfecta with quantitative defect of type I collagen synthesis. J Pediatr. 1996;129(3):432–9.
Antoniazzi F, Monti E, Venturi G, Franceschi R, Doro F, Gatti D, et al. GH in combination with bisphosphonate treatment in osteogenesis imperfecta. Eur J Endocrinol. 2010;163(3):479–87. doi:10.1530/eje-10-0208.
Orwoll ES, Shapiro J, Veith S, Wang Y, Lapidus J, Vanek C, et al. Evaluation of teriparatide treatment in adults with osteogenesis imperfecta. J Clin Invest. 2014;124(2):491–8. doi:10.1172/jci71101. This paper demonstrating bone density and fracture benefits of oral risedronate re-opened the door to use of oral bisphosphonate for children with milder OI.
Grafe I, Yang T, Alexander S, Homan EP, Lietman C, Jiang MM, et al. Excessive transforming growth factor-beta signaling is a common mechanism in osteogenesis imperfecta. Nat Med. 2014;20(6):670–5. doi:10.1038/nm.3544. This paper offers a novel mechanistic explanation for how the collagen and collagen-related mutations in the OI types result in brittle bones and opens the door to a new, personalized therapeutic approach to therapy for the disease.
Otsuru S, Gordon PL, Shimono K, Jethva R, Marino R, Phillips CL, et al. Transplanted bone marrow mononuclear cells and MSCs impart clinical benefit to children with osteogenesis imperfecta through different mechanisms. Blood. 2012;120(9):1933–41. doi:10.1182/blood-2011-12-400085.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Inas H. Thomas declares no conflict of interest. Linda A. DiMeglio reports other payments from AMGEN, outside the submitted work.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Rare Bone Disease
iThenticate: 8 %
Rights and permissions
About this article
Cite this article
Thomas, I.H., DiMeglio, L.A. Advances in the Classification and Treatment of Osteogenesis Imperfecta. Curr Osteoporos Rep 14, 1–9 (2016). https://doi.org/10.1007/s11914-016-0299-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-016-0299-y