Abstract
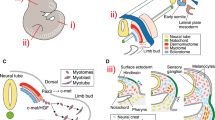
In the musculoskeletal system, muscle, tendon, and bone tissues develop in a spatially and temporally coordinated manner, and integrate into a cohesive functional unit by forming specific connections unique to each region of the musculoskeletal system. The mechanisms of these patterning and integration events are an area of great interest in musculoskeletal biology. Hox genes are a family of important developmental regulators and play critical roles in skeletal patterning throughout the axial and appendicular skeleton. Unexpectedly, Hox genes are not expressed in the differentiated cartilage or other skeletal cells, but rather are highly expressed in the tightly associated stromal connective tissues as well as regionally expressed in tendons and muscle connective tissue. Recent work has revealed a previously unappreciated role for Hox in patterning all the musculoskeletal tissues of the limb. These observations suggest that integration of the musculoskeletal system is regulated, at least in part, by Hox function in the stromal connective tissue. This review will outline our current understanding of Hox function in patterning and integrating the musculoskeletal tissues.




Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance•• Of major importance
Duboule D. Patterning in the vertebrate limb. Curr Opin Genet Dev. 1991;1(2):211–6. doi:10.1016/S0959-437X(05)80072-3.
Krumlauf R. Hox genes in vertebrate development. Cell. 1994;78(2):191–201.
Lewis EB. Genes and Developmental Pathways. Am Zool. 1963;3(1):33–56. doi:10.2307/3881152.
Lewis EB. A gene complex controlling segmentation in Drosophila. Nature. 1978;276(5688):565–70.
Lewis EB. Regulation of the genes of the bithorax complex in Drosophila. Cold Spring Harb Symp Quant Biol. 1985;50:155–64.
Wellik DM, Capecchi MR. Hox10 and Hox11 genes are required to globally pattern the mammalian skeleton. Science. 2003;301(5631):363–7. doi:10.1126/science.1085672.
Scott MP. Vertebrate homeobox gene nomenclature. Cell. 1992;71(4):551–3.
Pearson JC, Lemons D, McGinnis W. Modulating Hox gene functions during animal body patterning. Nature Rev Genet. 2005;6(12):893–904. doi:10.1038/nrg1726.
Condie BG, Capecchi MR. Mice with targeted disruptions in the paralogous genes hoxa-3 and hoxd-3 reveal synergistic interactions. Nature. 1994;370(6487):304–7. doi:10.1038/370304a0.
Davis AP, Witte DP, Hsieh-Li HM, Potter SS, Capecchi MR. Absence of radius and ulna in mice lacking hoxa-11 and hoxd-11. Nature. 1995;375(6534):791–5. doi:10.1038/375791a0.
Fromental-Ramain C, Warot X, Lakkaraju S, Favier B, Haack H, Birling C, et al. Specific and redundant functions of the paralogous Hoxa-9 and Hoxd-9 genes in forelimb and axial skeleton patterning. Development. 1996;122(2):461–72.
Fromental-Ramain C, Warot X, Messadecq N, LeMeur M, Dolle P, Chambon P. Hoxa-13 and Hoxd-13 play a crucial role in the patterning of the limb autopod. Development. 1996;122(10):2997–3011.
Horan GS, Ramirez-Solis R, Featherstone MS, Wolgemuth DJ, Bradley A, Behringer RR. Compound mutants for the paralogous hoxa-4, hoxb-4, and hoxd-4 genes show more complete homeotic transformations and a dose-dependent increase in the number of vertebrae transformed. Genes Dev. 1995;9(13):1667–77.
McIntyre DC, Rakshit S, Yallowitz AR, Loken L, Jeannotte L, Capecchi MR, et al. Hox patterning of the vertebrate rib cage. Development. 2007;134(16):2981–9. doi:10.1242/dev.007567.
van den Akker E, Fromental-Ramain C, de Graaff W, Le Mouellic H, Brulet P, Chambon P, et al. Axial skeletal patterning in mice lacking all paralogous group 8 Hox genes. Development. 2001;128(10):1911–21.
Wellik DM, Hawkes PJ, Capecchi MR. Hox11 paralogous genes are essential for metanephric kidney induction. Genes Dev. 2002;16(11):1423–32. doi:10.1101/gad.993302.
Chen F, Capecchi MR. Targeted mutations in hoxa-9 and hoxb-9 reveal synergistic interactions. Dev Biol. 1997;181(2):186–96. doi:10.1006/dbio.1996.8440.
Chen F, Capecchi MR. Paralogous mouse Hox genes, Hoxa9, Hoxb9, and Hoxd9, function together to control development of the mammary gland in response to pregnancy. Proc Natl Acad Sci U S A. 1999;96(2):541–6.
Chen F, Greer J, Capecchi MR. Analysis of Hoxa7/Hoxb7 mutants suggests periodicity in the generation of the different sets of vertebrae. Mech Dev. 1998;77(1):49–57.
Gavalas A, Trainor P, Ariza-McNaughton L, Krumlauf R. Synergy between Hoxa1 and Hoxb1: the relationship between arch patterning and the generation of cranial neural crest. Development. 2001;128(15):3017–27.
Manley NR, Capecchi MR. Hox group 3 paralogs regulate the development and migration of the thymus, thyroid, and parathyroid glands. Dev Biol. 1998;195(1):1–15. doi:10.1006/dbio.1997.8827.
Studer M, Gavalas A, Marshall H, Ariza-McNaughton L, Rijli FM, Chambon P, et al. Genetic interactions between Hoxa1 and Hoxb1 reveal new roles in regulation of early hindbrain patterning. Development. 1998;125(6):1025–36.
Wahba GM, Hostikka SL, Carpenter EM. The paralogous Hox genes Hoxa10 and Hoxd10 interact to pattern the mouse hindlimb peripheral nervous system and skeleton. Dev Biol. 2001;231(1):87–102. doi:10.1006/dbio.2000.0130.
Wellik DM. Hox patterning of the vertebrate axial skeleton. Dev Dynam. 2007;236(9):2454–63. doi:10.1002/dvdy.21286.
Wellik DM. Hox genes and vertebrate axial pattern. Curr Top Dev Biol. 2009;88:257–78. doi:10.1016/s0070-2153(09)88009-5.
Boulet AM, Capecchi MR. Multiple roles of Hoxa11 and Hoxd11 in the formation of the mammalian forelimb zeugopod. Development. 2004;131(2):299–309. doi:10.1242/dev.00936.
Hostikka SL, Capecchi MR. The mouse Hoxc11 gene: genomic structure and expression pattern. Mech Dev. 1998;70(1–2):133–45.
Kmita M, Tarchini B, Zakany J, Logan M, Tabin CJ, Duboule D. Early developmental arrest of mammalian limbs lacking HoxA/HoxD gene function. Nature. 2005;435(7045):1113–6. doi:10.1038/nature03648.
Xu B, Wellik DM. Axial Hox9 activity establishes the posterior field in the developing forelimb. Proc Natl Acad Sci U S A. 2011;108(12):4888–91. doi:10.1073/pnas.1018161108.
Xu B, Hrycaj SM, McIntyre DC, Baker NC, Takeuchi JK, Jeannotte L, et al. Hox5 interacts with Plzf to restrict Shh expression in the developing forelimb. Proc Natl Acad Sci U S A. 2013;110(48):19438–43. doi:10.1073/pnas.1315075110.
Karsenty G, Kronenberg HM, Settembre C. Genetic control of bone formation. Ann Rev Cell Dev Biol. 2009;25:629–48. doi:10.1146/annurev.cellbio.042308.113308.
Schweitzer R, Zelzer E, Volk T. Connecting muscles to tendons: tendons and musculoskeletal development in flies and vertebrates. Development. 2010;137(17):2807–17. doi:10.1242/dev.047498.
Murphy M, Kardon G. Origin of vertebrate limb muscle: the role of progenitor and myoblast populations. Curr Top Dev Biol. 2011;96:1–32. doi:10.1016/B978-0-12-385940-2.00001-2.
Kieny M, Chevallier A. Autonomy of tendon development in the embryonic chick wing. J Embryol Exp Morphol. 1979;49:153–65.
Shellswell G, Wolpert L. The pattern of muscle and tendon development in the chick wing. Vert Limb Somite Morphogen. 1977:71–86.
Chevallier A, Kieny M, Mauger A. Limb-somite relationship: origin of the limb musculature. J Embryol Exp Morphol. 1977;41(1):245–58.
Christ B, Jacob HJ, Jacob M. Experimental analysis of the origin of the wing musculature in avian embryos. Anatomy Embryol. 1977;150(2):171–86.
Ordahl CP, Le Douarin NM. Two myogenic lineages within the developing somite. Development. 1992;114(2):339–53.
Wachtler F, Christ B, Jacob HJ. On the determination of mesodermal tissues in the avian embryonic wing bud. Anatomy Embryol. 1981;161(3):283–9.
Thorogood PV, Hinchliffe JR. An analysis of the condensation process during chondrogenesis in the embryonic chick hind limb. J Embryol Exp Morphol. 1975;33(3):581–606.
Kardon G. Muscle and tendon morphogenesis in the avian hind limb. Development. 1998;125(20):4019–32.
Schweitzer R, Chyung JH, Murtaugh LC, Brent AE, Rosen V, Olson EN, et al. Analysis of the tendon cell fate using Scleraxis, a specific marker for tendons and ligaments. Development. 2001;128(19):3855–66.
Schroeter S, Tosney KW. Spatial and temporal patterns of muscle cleavage in the chick thigh and their value as criteria for homology. Am J Anat. 1991;191(4):325–50. doi:10.1002/aja.1001910402.
Schroeter S, Tosney KW. Ultrastructural and morphometric analysis of the separation of two thigh muscles in the chick. Am J Anat. 1991;191(4):351–68. doi:10.1002/aja.1001910403.
Kardon G, Campbell JK, Tabin CJ. Local extrinsic signals determine muscle and endothelial cell fate and patterning in the vertebrate limb. Dev Cell. 2002;3(4):533–45.
Brent AE, Braun T, Tabin CJ. Genetic analysis of interactions between the somitic muscle, cartilage and tendon cell lineages during mouse development. Development. 2005;132(3):515–28. doi:10.1242/dev.01605.
Edom-Vovard F, Schuler B, Bonnin M-A, Teillet M-A, Duprez D. Fgf4 positively regulates scleraxis and tenascin expression in chick limb tendons. Dev Biol. 2002;247(2):351–66. doi:10.1006/dbio.2002.0707.
Hasson P, DeLaurier A, Bennett M, Grigorieva E, Naiche LA, Papaioannou VE, et al. Tbx4 and tbx5 acting in connective tissue are required for limb muscle and tendon patterning. Dev Cell. 2010;18(1):148–56. doi:10.1016/j.devcel.2009.11.013.
Kardon G, Harfe BD, Tabin CJ. A Tcf4-positive mesodermal population provides a pre-pattern for vertebrate limb muscle patterning. Dev Cell. 2003;5(6):937–44.
Mathew SJ, Hansen JM, Merrell AJ, Murphy MM, Lawson JA, Hutcheson DA, et al. Connective tissue fibroblasts and Tcf4 regulate myogenesis. Development. 2011;138(2):371–84. doi:10.1242/dev.057463. The authors show that muscle connective tissue fibroblasts express Tcf4. Generation of Tcf4GFPCre mice provides the first genetic tool for manipulating these fibroblasts. This work presents definitive evidence that muscle connective tissue regulates muscle development in addition to muscle structure and function.
Hall BK, Herring SW. Paralysis and growth of the musculoskeletal system in the embryonic chick. J Morphol. 1990;206(1):45–56. doi:10.1002/jmor.1052060105.
Hosseini A, Hogg DA. The effects of paralysis on skeletal development in the chick embryo. II. Effects on histogenesis of the tibia. J Anatomy. 1991;177:169–78.
Sharir A, Stern T, Rot C, Shahar R, Zelzer E. Muscle force regulates bone shaping for optimal load-bearing capacity during embryogenesis. Development. 2011;138(15):3247–59. doi:10.1242/dev.063768.
Murchison ND, Price BA, Conner DA, Keene DR, Olson EN, Tabin CJ, et al. Regulation of tendon differentiation by scleraxis distinguishes force-transmitting tendons from muscle-anchoring tendons. Development. 2007;134(14):2697–708. doi:10.1242/dev.001933.
Akiyama H, Kim JE, Nakashima K, Balmes G, Iwai N, Deng JM, et al. Osteo-chondroprogenitor cells are derived from Sox9 expressing precursors. Proc Natl Acad Sci U S A. 2005;102(41):14665–70. doi:10.1073/pnas.0504750102.
Blitz E, Sharir A, Akiyama H, Zelzer E. Tendon-bone attachment unit is formed modularly by a distinct pool of Scx- and Sox9-positive progenitors. Development. 2013;140(13):2680–90. doi:10.1242/dev.093906.
Sugimoto Y, Takimoto A, Akiyama H, Kist R, Scherer G, Nakamura T, et al. Scx+/Sox9+ progenitors contribute to the establishment of the junction between cartilage and tendon/ligament. Development. 2013;140(11):2280–8. doi:10.1242/dev.096354.
Blitz E, Viukov S, Sharir A, Shwartz Y, Galloway JL, Pryce BA, et al. Bone ridge patterning during musculoskeletal assembly is mediated through SCX regulation of Bmp4 at the tendon-skeleton junction. Dev Cell. 2009;17(6):861–73. doi:10.1016/j.devcel.2009.10.010. This work highlights the critical interaction between bone and tendon during the integration of these two tissues during development as well as key regulators of these processes.
Ralphs JR, Benjamin M, Waggett AD, Russell DC, Messner K, Gao J. Regional differences in cell shape and gap junction expression in rat Achilles tendon: relation to fibrocartilage differentiation. J Anatomy. 1998;193(Pt 2):215–22.
Thomopoulos S, Williams GR, Gimbel JA, Favata M, Soslowsky LJ. Variation of biomechanical, structural, and compositional properties along the tendon to bone insertion site. J Orthopaed Res. 2003;21(3):413–9. doi:10.1016/S0736-0266(03)00057-3.
Swinehart IT, Schlientz AJ, Quintanilla CA, Mortlock DP, Wellik DM. Hox11 genes are required for regional patterning and integration of muscle, tendon and bone. Development. 2013;140(22):4574–82. doi:10.1242/dev.096693. This paper shows that loss of Hox11 results in mis-patterning of the muscles and tendons of the zeugopod in addition to the well described skeletal phenotypes. Hoxa11eGFP is expressed in connective tissue stromal cells, suggesting that Hox function in these cells is required for proper patterning of the musculoskeletal system.
Stadler HS, Higgins KM, Capecchi MR. Loss of Eph-receptor expression correlates with loss of cell adhesion and chondrogenic capacity in Hoxa13 mutant limbs. Development. 2001;128(21):4177–88.
Acknowledgements
The authors would like to acknowledge Holly Fischer for creating the illustrations.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
K. M. Pineault and D. M. Wellik declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
All studies by K. M. Pineault and D. M. Wellik involving animal research were performed after approval by the appropriate institutional review boards.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pineault, K.M., Wellik, D.M. Hox Genes and Limb Musculoskeletal Development. Curr Osteoporos Rep 12, 420–427 (2014). https://doi.org/10.1007/s11914-014-0241-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-014-0241-0