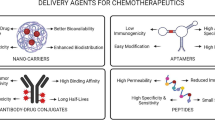
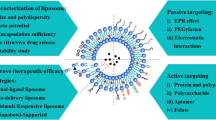
Abstract
Distribution of antineoplastic agents within tumors remains one of the major challenges in cancer chemotherapy because distribution is hampered by several factors related to the drug (its physicochemical characteristics) and to the neoplastic tissue (blood and lymphatic vasculature, cell density, extracellular matrix composition, and interstitium). The inhomogeneous distribution and structure of tumor vasculature lead to large avascular and hypoxic areas with low pH and high interstitial oncotic pressure. In these critical conditions, the gradient of drug concentrations from the vessels to the inner parts of the tumor is not sufficient to promote diffusion of pharmacologic agents. Again, cellular sequestration and binding to extracellular matrix represent further factors that limit drug distribution and reduce tumor sensitivity to chemotherapy. Several strategies have been investigated to circumvent drug resistance. The evaluation of liposomal and nanoparticle formulations and the characterization of newer bioreductive agents and drugs that should normalize tumor vasculature are in progress.
Similar content being viewed by others
References and Recommended Reading
Jain RK: Barriers to drug delivery in solid tumors. Sci Am 1994, 271:58–65.
Vaupel P, Kelleher DK, Hockel M: Oxygen status of malignant tumors: pathogenesis of hypoxia and significance for tumor therapy. Semin Oncol 2001, 28:29–35.
Vaupel P, Mayer A, Briest S, Hockel M: Hypoxia in breast cancer: role of blood flow, oxygen diffusion distances, and anemia in the development of oxygen depletion. Adv Exp Med Biol 2005, 566:333–342.
Jain RK: Determinants of tumor blood flow: a review. Cancer Res 1988, 48:2641–2658.
Maruyama K, Ishida O, Takizawa T, Moribe K: Possibility of active targeting to tumor tissues with liposomes. Adv Drug Deliv Rev 1999, 40:89–102.
Braun RD, Abbas A, Bukhari SO, Wilson W III: Hemodynamic parameters in blood vessels in choroidal melanoma xenografts and rat choroid. Invest Ophthalmol Vis Sci 2002, 43:3045–3052.
Al-Rawi MA, Mansel RE, Jiang WG: Lymphangiogenesis and its role in cancer. Histol Histopathol 2005, 20:283–298.
Grantab R, Sivananthan S, Tannock IF: The penetration of anticancer drugs through tumor tissue as a function of cellular adhesion and packing density of tumor cells. Cancer Res 2006, 66:1033–1039.
Jain RK: Transport of macromolecules in the tumor interstitium: a review. Cancer Res 1987, 47:3039–3051.
Toole BP: Hyaluronan: from extracellular glue to pericellular cue. Nat Rev Cancer 2004, 4:528–539.
Nederman T, Carlsson J: Penetration and binding of vinblastine and 5-fluorouracil in cellular spheroids. Cancer Chemother Pharmacol 1984, 13:131–135.
Erlanson M, Daniel-Szolgay E, Carlsson J: Relations between the penetration, binding and average concentration of cytostatic drugs in human tumour spheroids. Cancer Chemother Pharmacol 1992, 29:343–353.
Kuh H-J, Jang SH, Guillaume Wientjes M, et al.: Determinants of paclitaxel penetration and accumulation in human solid tumor. J Pharmacol Exp Ther 1999, 290:871–880.
Jang SH, Guillaume Wientjes M, Au JL-S: Enhancement of paclitaxel delivery to solid tumors by apoptosis-inducing pretreatment: effect of treatment schedule. J Pharmacol Exp Ther 2001, 296:1035–1042.
Semenza GL: Targeting HIF-1 for cancer therapy. Nat Rev Cancer 2003, 3:721–732.
Svastova E, Hulikova A, Rafajova M, et al.: Hypoxia activates the capacity of tumor-associated carbonic anhydrase IX to acidify extracellular pH. FEBS Lett 2004, 577:439–435.
Wike-Hooley JL, Haveman J, Reinhold HS: The relevance of tumour pH to the treatment of malignant disease. Radiother Oncol 1984, 2:343–366.
Madshus IH: Regulation of intracellular pH in eukaryotic cells. Biochem J 1988, 250:1–8.
Mahoney BP, Raghunand N, Baggett B, Gillies RJ: Tumor acidity, ion trapping and chemotherapeutics. I. Acid pH affects the distribution of chemotherapeutic agents in vitro. Biochem Pharmacol 2003, 66:1207–1218.
Vukovic V, Tannock IF: Influence of low pH on cytotoxicity of paclitaxel, mitoxantrone and topotecan. Br J Cancer 1997, 75:1167–1172.
Acker T, Plate KH: Hypoxia and hypoxia inducible factors (HIF) as important regulators of tumor physiology. Cancer Treat Res 2004, 117:219–248.
Hussein D, Estlin EJ, Dive C, Makin GW: Chronic hypoxia promotes hypoxia-inducible factor-1alpha-dependent resistance to etoposide and vincristine in neuroblastoma cells. Mol Cancer Ther 2006, 5:2241–2250.
Krishnamurthy P, Schuetz JD: The ABC transporter Abcg2/Bcrp: role in hypoxia mediated survival. Biometals 2005, 18:349–358.
Krishnamurthy P, Schuetz JD: Role of ABCG2/BCRP in biology and medicine. Annu Rev Pharmacol Toxicol. 2006, 46:381–410.
Burger H, van Tol H, Brok M, et al.: Chronic imatinib mesylate exposure leads to reduced intracellular drug accumulation by induction of the ABCG2 (BCRP) and ABCB1 (MDR1) drug transport pumps. Cancer Biol Ther 2005, 4:747–752.
Calatozzolo C, Gelati M, Ciusani E, et al.: Expression of drug resistance proteins Pgp, MRP1, MRP3, MRP5 and GST-pi in human glioma. J Neurooncol 2005, 74:113–121.
Hofheinz RD, Gnad-Vogt SU, Beyer U, Hochhaus A: Liposomal encapsulated anti-cancer drugs. Anticancer Drugs 2005, 16:691–707.
Yih TC, Al-Fandi M: Engineered nanoparticles as precise drug delivery systems. J Cell Biochem 2006; 97:1184–1190.
Drummond DC, Meyer O, Hong K, et al.: Optimizing liposomes for delivery of chemotherapeutic agents to solid tumors. Pharmacol Rev 1999, 51:691–743.
Northfelt DW, Martin FJ, Working P, et al.: Doxorubicin encapsulated in liposomes containing surface-bound polyethylene glycol: pharmacokinetics, tumor localization, and safety in patients with AIDS-related Kaposi’s sarcoma. J Clin Pharmacol 1996, 36:55–63.
Kontermann RE: Immunoliposomes for cancer therapy. Curr Opin Mol Ther 2006, 8:39–45.
Gao Z, Fain HD, Rapoport N: Ultrasound-enhanced tumor targeting of polymeric micellar drug carriers. Mol Pharm 2004, 1:317–330.
Wu AM, Senter PD: Arming antibodies: prospects and challenges for immunoconjugates. Nat Biotechnol 2005, 23:1137–1146.
Ahmed M, Goldberg SN: Combination radiofrequency thermal ablation and adjuvant IV liposomal doxorubicin increases tissue coagulation and intratumoural drug accumulation. Int J Hyperthermia 2004, 20:781–802.
Zheng JH, Chen CT, Au JL, Wientjes MG: Time-and concentration-dependent penetration of doxorubicin in prostate tumors. AAPS PharmSci 2001, 3:E15.
Eikenes L, Tari M, Tufto I, et al.: Hyaluronidase induces a transcapillary pressure gradient and improves the distribution and uptake of liposomal doxorubicin (Caelyx) in human osteosarcoma xenografts. Br J Cancer 2005, 93:81–88.
Hurwitz, H. Fehrenbacher L, Novotny W, et al.: Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 2004, 350:2335–2342.
Tong RT, Boucher Y, Kozin SV et al.: Vascular normalization by vascular endothelial growth factor receptor 2 blockade induces a pressure gradient across the vasculature and improves drug penetration in tumors. Cancer Res 2004, 64:3731–3736.
Winkler, F. Kozin SV, Tong RT, et al.: Kinetics of vascular normalization by VEGFR2 blockade governs brain tumor response to radiation: role of oxygenation, angiopoietin-1, and matrix metalloproteinases. Cancer Cell 2004, 6:553–563.
Franco, M, Man S, Chen L, et al.: Targeted anti-vascular endothelial growth factor receptor-2 therapy leads to short-term and long-term impairment of vascular function and increase in tumor hypoxia. Cancer Res 2006, 66:3639–3648.
Hirpara JL, Clement MV, Pervaiz S: Intracellular acidification triggered by mitochondrial-derived hydrogen peroxide is an effector mechanism for drug-induced apoptosis in tumor cells. J Biol Chem 2001, 276:514–521.
Ahmad KA, Iskandar KB, Hirpara JL, et al.: Hydrogen peroxide-mediated cytosolic acidification is a signal for mitochondrial translocation of Bax during drug-induced apoptosis of tumor cells. Cancer Res 2004, 64:7867–7878.
De Milito A, Fais S: Tumor acidity, chemoresistance and proton pump inhibitors. Future Oncol 2005, 1:779–786.
Stratford IJ, Williams KJ, Cowen RL, Jaffar M: Combining bioreductive drugs and radiation for the treatment of solid tumors. Semin Radiat Oncol 2003, 13:42–52.
Boyle RG, Travers S: Hypoxia: targeting the tumour. Anticancer Agents Med Chem 2006, 6:281–286.
von Pawel J, von Roemeling R, Gatzemeier U, et al.: Tirazpamine plus cisplatin versus cisplatin in advanced non-small-cell lung cancer: A report of the international CATAPULT I study group. Cisplatin and Tirapazamine in Subjects with Advanced Previously Untreated Non-Small-Cell Lung Tumors. J Clin Oncol 2000, 18:1351–1359.
Rischin D, Peters L, Hicks R, et al.: Phase I trial of concurrent tirapazamine, cisplatin, and radiotherapy in patients with advanced head and neck cancer. J Clin Oncol 2001, 19:535–542.
Papadopoulou MV, Bloomer WD: NLCQ-1 (NSC 709257): exploiting hypoxia with a weak DNA-intercalating bioreductive drug. Clin Cancer Res 2003, 9:5714–5720.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Paolo, A., Bocci, G. Drug distribution in tumors: Mechanisms, role in drug resistance, and methods for modification. Curr Oncol Rep 9, 109–114 (2007). https://doi.org/10.1007/s11912-007-0006-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-007-0006-3