Abstract
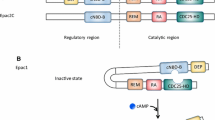
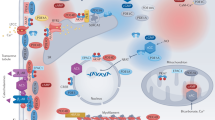
Cyclic adenosine 3′,5′-monophosphate (cAMP) mediates the biological effects of various hormones and neurotransmitters. Stimulation of cardiac β-adrenergic receptors (β-AR) via catecholamines leads to activation of adenylyl cyclases and increases cAMP production to enhance myocardial function. Because many other receptors signaling through cAMP generation exist in cardiac myocytes, a central question is how different hormones induce distinct cellular responses through the same second messenger. A large body of evidence suggests that the localization and compartmentalization of β-AR/cAMP signaling affects the net outcome of biological functions. Spatiotemporal dynamics of cAMP action is achieved by various proteins, including protein kinase A (PKA), phosphodiesterases, and scaffolding proteins such as A-kinase–anchoring proteins. In addition, the discovery of the cAMP target Epac (exchange proteins directly activated by cAMP), which functions in a PKA-independent manner, represents a novel mechanism for governing cAMP-signaling specificity. Aberrant cAMP signaling through dysregulation of β-AR/cAMP compartmentalization may contribute to cardiac remodeling and heart failure.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as:•• Of major importance
Bers DM. Calcium cycling and signaling in cardiac myocytes. Annu Rev Physiol. 2008;70:23–49.
Movsesian MA, Bristow MR. Alterations in cAMP-mediated signaling and their role in the pathophysiology of dilated cardiomyopathy. Curr Top Dev Biol. 2005;68:25–48.
Lohse MJ, Engelhardt S, Eschenhagen T. What is the role of beta-adrenergic signaling in heart failure? Circ Res. 2003;93:896–906.
Fischmeister R, Castro LR, Abi-Gerges A, et al. Compartmentalization of cyclic nucleotide signaling in the heart: the role of cyclic nucleotide phosphodiesterases. Circ Res. 2006;99:816–28.
Houslay MD. Underpinning compartmentalised cAMP signaling through targeted cAMP breakdown. Trends Biochem Sci. 2010;35:91–100.
Scott JD, Santana LF. A-kinase anchoring proteins: getting to the heart of the matter. Circulation. 2010;121:1264–71.
Gloerich M, Bos JL. Epac: defining a new mechanism for cAMP action. Annu Rev Pharmacol Toxicol. 2010;50:355–75.
Lompre AM, Hajjar RJ, Harding SE, et al. Ca2+ cycling and new therapeutic approaches for heart failure. Circulation. 2010;121:822–30.
Gauthier C, Leblais V, Kobzik L, et al. The negative inotropic effect of beta3-adrenoceptor stimulation is mediated by activation of a nitric oxide synthase pathway in human ventricle. J Clin Invest. 1998;102:1377–84.
Skeberdis VA, Gendviliene V, Zablockaite D, et al. beta3-adrenergic receptor activation increases human atrial tissue contractility and stimulates the L-type Ca2+ current. J Clin Invest. 2008;118:3219–27.
Kuschel M, Zhou YY, Spurgeon HA, et al. beta2-adrenergic cAMP signaling is uncoupled from phosphorylation of cytoplasmic proteins in canine heart. Circulation. 1999;99:2458–65.
Zhu WZ, Wang SQ, Chakir K, et al. Linkage of beta1-adrenergic stimulation to apoptotic heart cell death through protein kinase A-independent activation of Ca2+/calmodulin kinase II. J Clin Invest. 2003;111:617–25.
Engelhardt S, Hein L, Wiesmann F, Lohse MJ. Progressive hypertrophy and heart failure in beta1-adrenergic receptor transgenic mice. Proc Natl Acad Sci USA. 1999;96:7059–64.
Liggett SB, Tepe NM, Lorenz JN, et al. Early and delayed consequences of beta(2)-adrenergic receptor overexpression in mouse hearts: critical role for expression level. Circulation. 2000;101:1707–14.
Xiao RP, Zhang SJ, Chakir K, et al. Enhanced G(I) signaling selectively negates beta2-adrenergic receptor (AR)–but not beta1-AR-mediated positive inotropic effect in myocytes from failing rat hearts. Circulation. 2003;108:1633–9.
Talan MI, Ahmet I, Xiao RP, Lakatta EG. beta(2) AR agonists in treatment of chronic heart failure: long path to translation. J Mol Cell Cardiol. 2010, In press
Rybin VO, Xu X, Lisanti MP, Steinberg SF. Differential targeting of beta -adrenergic receptor subtypes and adenylyl cyclase to cardiomyocyte caveolae. A mechanism to functionally regulate the cAMP signaling pathway. J Biol Chem. 2000;275:41447–57.
Jurevicius J, Fischmeister R. cAMP compartmentalization is responsible for a local activation of cardiac Ca2+ channels by beta-adrenergic agonists. Proc Natl Acad Sci USA. 1996;93:295–9.
DiPilato LM, Zhang J. Fluorescent protein-based biosensors: resolving spatiotemporal dynamics of signaling. Curr Opin Chem Biol. 2010;14:37–42.
Nikolaev VO, Bunemann M, Schmitteckert E, et al. Cyclic AMP imaging in adult cardiac myocytes reveals far-reaching beta1-adrenergic but locally confined beta2-adrenergic receptor-mediated signaling. Circ Res. 2006;99:1084–91.
•• Nikolaev VO, Moshkov A, Lyon AR, et al. Beta2-adrenergic receptor redistribution in heart failure changes cAMP compartmentalization. Science. 2010; 327:1653–7. This article describes a redistribution of the β 2 -ARs from the T-tubules to the cell crest in failing cardiomyocytes.
Zaccolo M. cAMP signal transduction in the heart: understanding spatial control for the development of novel therapeutic strategies. Br J Pharmacol. 2009;158:50–60.
Zaccolo M, Pozzan T. Discrete microdomains with high concentration of cAMP in stimulated rat neonatal cardiac myocytes. Science. 2002;295:1711–5.
Mongillo M, McSorley T, Evellin S, et al. Fluorescence resonance energy transfer-based analysis of cAMP dynamics in live neonatal rat cardiac myocytes reveals distinct functions of compartmentalized phosphodiesterases. Circ Res. 2004;95:67–75.
Rochais F, Abi-Gerges A, Horner K, et al. A specific pattern of phosphodiesterases controls the cAMP signals generated by different Gs-coupled receptors in adult rat ventricular myocytes. Circ Res. 2006;98:1081–8.
Richter W, Day P, Agrawal R, et al. Signaling from beta1- to beta2-adrenergic receptors is defined by differential interactions with PDE4. EMBO J. 2008;27:384–93.
Perry SJ, Baillie GS, Kohout TA, et al. Targeting of cyclic AMP degradation to beta 2-adrenergic receptors by beta-arrestins. Science. 2002;298:834–6.
Baillie GS, Sood A, McPhee I, et al. beta-Arrestin-mediated PDE4 cAMP phosphodiesterase recruitment regulates beta-adrenoceptor switching from Gs to Gi. Proc Natl Acad Sci USA. 2003;100:940–5.
•• Leroy J, Abi-Gerges A, Nikolaev VO, et al. Spatiotemporal dynamics of beta-adrenergic cAMP signals and L-type Ca2+ channel regulation in adult rat ventricular myocytes: role of phosphodiesterases. Circ Res. 2008; 102:1091–100. This is a study showing the spatiotemporal contribution of PDE isoforms 3 and 4 to the β-AR response of cardiac myocytes.
Abi-Gerges A, Richter W, Lefebvre F, et al. Decreased expression and activity of cAMP phosphodiesterases in cardiac hypertrophy and its impact on beta-adrenergic cAMP signals. Circ Res. 2009;105:784–92.
Ding B, Abe J, Wei H, et al. Functional role of phosphodiesterase 3 in cardiomyocyte apoptosis: implication in heart failure. Circulation. 2005;111:2469–76.
Lehnart SE, Wehrens XH, Reiken S, et al. Phosphodiesterase 4D deficiency in the ryanodine-receptor complex promotes heart failure and arrhythmias. Cell. 2005;123:25–35.
Sin YY, Edwards HV, Li X, et al. Disruption of the cyclic AMP phosphodiesterase-4 (PDE4)-HSP20 complex attenuates the beta-agonist induced hypertrophic response in cardiac myocytes. J Mol Cell Cardiol. 2011, In press
Dodge KL, Khouangsathiene S, Kapiloff MS, et al. mAKAP assembles a protein kinase A/PDE4 phosphodiesterase cAMP signaling module. EMBO J. 2001;20:1921–30.
Dodge-Kafka KL, Langeberg L, Scott JD. Compartmentalization of cyclic nucleotide signaling in the heart: the role of A-kinase anchoring proteins. Circ Res. 2006;98:993–1001.
Lygren B, Carlson CR, Santamaria K, et al. AKAP complex regulates Ca2+ re-uptake into heart sarcoplasmic reticulum. EMBO Rep. 2007;8:1061–7.
McConnell BK, Popovic Z, Mal N, et al. Disruption of protein kinase A interaction with A-kinase-anchoring proteins in the heart in vivo: effects on cardiac contractility, protein kinase A phosphorylation, and troponin I proteolysis. J Biol Chem. 2009;284:1583–92.
Kapiloff MS, Jackson N, Airhart N. mAKAP and the ryanodine receptor are part of a multi-component signaling complex on the cardiomyocyte nuclear envelope. J Cell Sci. 2001;114:3167–76.
•• Carnegie GK, Soughayer J, Smith FD, et al. AKAP-Lbc mobilizes a cardiac hypertrophy signaling pathway. Mol Cell. 2008; 32:169–79. This article demonstrates in molecular detail that AKAP-Lbc activates a prohypertrophic signaling involving protein kinase D activation and the nuclear export of the class II histone deacetylase HDAC.
Christian F, Szaszak M, Friedl S, et al. Small molecule AKAP/PKA interaction disruptors that activate PKA interfere with compartmentalized cAMP signaling in cardiac myocytes. J Biol Chem. 2011, In press
Enserink JM, Christensen AE, de Rooij J, et al. A novel Epac-specific cAMP analogue demonstrates independent regulation of Rap1 and ERK. Nat Cell Biol. 2002;4:901–6.
Breckler M, Berthouze M, Laurent AC, et al. Rap-linked cAMP signaling Epac proteins: compartmentalization, functioning and disease implications. Cell Signal. 2011, In press
•• Metrich M, Lucas A, Gastineau M, et al. Epac mediates beta-adrenergic receptor-induced cardiomyocyte hypertrophy. Circ Res. 2008;102:959–65. This article demonstrates that Epac prohypertrohic signaling pathway involves calcineurin and CaMKII in adult cardiac myocytes in response to β-AR activation.
Rehmann H, Wittinghofer A, Bos JL. Capturing cyclic nucleotides in action: snapshots from crystallographic studies. Nat Rev Mol Cell Biol. 2007;8:63–73.
Metrich M, Berthouze M, Morel E, et al. Role of the cAMP-binding protein Epac in cardiovascular physiology and pathophysiology. Pflugers Arch. 2010;459:535–46.
Morel E, Marcantoni A, Gastineau M, et al. cAMP-binding protein Epac induces cardiomyocyte hypertrophy. Circ Res. 2005;97:1296–304.
Ulucan C, Wang X, Baljinnyam E, et al. Developmental changes in gene expression of Epac and its upregulation in myocardial hypertrophy. Am J Physiol Heart Circ Physiol. 2007;293:H1662–72.
Metrich M, Laurent AC, Breckler M, et al. Epac activation induces histone deacetylase nuclear export via a Ras-dependent signaling pathway. Cell Signal. 2010; 22:1459–68.
Pereira L, Metrich M, Fernandez-Velasco M, et al. The cAMP binding protein Epac modulates Ca2+ sparks by a Ca2+/calmodulin kinase signaling pathway in rat cardiac myocytes. J Physiol. 2007;583:685–94.
Kwak HJ, Park KM, Choi HE, et al. PDE4 inhibitor, roflumilast protects cardiomyocytes against NO-induced apoptosis via activation of PKA and Epac dual pathways. Cell Signal. 2008;20:803–14.
Yokoyama U, Patel HH, Lai NC, et al. The cyclic AMP effector Epac integrates pro- and anti-fibrotic signals. Proc Natl Acad Sci USA. 2008;105:6386–91.
Somekawa S, Fukuhara S, Nakaoka Y, et al. Enhanced functional gap junction neoformation by protein kinase A-dependent and Epac-dependent signals downstream of cAMP in cardiac myocytes. Circ Res. 2005;97:655–62.
Cazorla O, Lucas A, Poirier F, et al. The cAMP binding protein Epac regulates cardiac myofilament function. Proc Natl Acad Sci USA. 2009;106:14144–9.
Dodge-Kafka KL, Soughayer J, Pare GC, et al. The protein kinase A anchoring protein mAKAP coordinates two integrated cAMP effector pathways. Nature. 2005;437:574–8.
Sehrawat S, Ernandez T, Cullere X, et al. AKAP9 regulation of microtubule dynamics promotes Epac1-induced endothelial barrier properties. Blood. 2011;117:708–18.
Gloerich M, Ponsioen B, Vliem MJ, et al. Spatial regulation of cyclic AMP-Epac1 signaling in cell adhesion by ERM proteins. Mol Cell Biol. 2010;30:5421–31.
Acknowledgements
We thank Drs. Jeanne Mialet-Perez and Bertrand Crozatier for their critical reading of the manuscript. The work of Dr. Frank Lezoualc’h mentioned herein was supported by grants from Agence Nationale de la Recherche («HyperEpac» Genopath09) to Région Midi-Pyrénées. Drs. Magali Breckler, Magali Berthouze, and Anne-Coline Laurent were recipients of grants from Région île-de-France (CODIM), Fondation Lefoulon Delalande, and Groupe de Réflexion sur la Recherche Cardiovasculaire (G.R.R.C)/Fédération Française de Cardiologie (F. F. C).
Disclosures
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berthouze, M., Laurent, AC., Breckler, M. et al. New Perspectives in cAMP-Signaling Modulation. Curr Heart Fail Rep 8, 159–167 (2011). https://doi.org/10.1007/s11897-011-0062-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11897-011-0062-8