Abstract
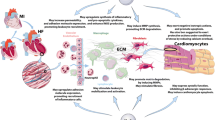
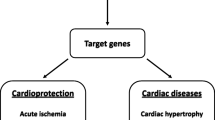
Recent studies have determined that expression of inflammatory mediators, such as cytokines and chemokines, is an important factor in the development and progression of heart failure (HF). These inflammatory mediators are expressed in response to various myocardial insults, including myocardial ischemia, viral infection, and toxins, and appear to have a detrimental effect on cardiac function and prognosis in HF patients. Our previous reports have shown activation of inflammatory cytokines, particularly tumor necrosis factor-α (TNF-α), in the myocardium and peripheral monocytes in patients with HF. Indeed, sustained increases in cytokines, including TNF-α and its receptor, lead to monocyte phenotype transition, myocytic apoptosis, and activation of matrix metalloproteinase. This in turn modifies the interstitial matrix, augmenting further ventricular remodeling. Thus, in view of the emerging importance of TNF-α in the pathogenesis of HF, we review the effects of TNF-α on the physiology of the heart and the development of clinical strategies to target the inflammatory cytokine cascade.
Similar content being viewed by others
References and Recommended Reading
Levine B, Kalman J, Mayer L, et al.: Elevated circulating levels of tumor necrosis factor in severe chronic heart failure. N Engl J Med 1990, 323:236–241.
Yokoyama T, Vaca L, Rossen RD, et al.: Cellular basis for the negative inotropic effects of tumor necrosis factoralpha in the adult mammalian heart. J Clin Invest 1993, 92:2303–2312.
Lo SK, Everitt J, Gu J, Malik AB: Tumor necrosis factor mediates experimental pulmonary edema by ICAM-1 and CD18-dependent mechanisms. J Clin Invest 1992, 89:981–988.
Hegewisch S, Weh HJ, Hossfeld DK: TNF-induced cardiomyopathy. Lancet 1990, 335:294–295.
Vaddi K, Nicolini FA, Mehta P, Mehta JL: Increased secretion of tumor necrosis factor-alpha and interferon-gamma by mononuclear leukocytes in patients with ischemic heart disease. Relevance in superoxide anion generation. Circulation 1994, 90:694–699.
Herskowitz A, Choi S, Ansari AA, Wesselingh S: Cytokine mRNA expression in postischemic/reperfused myocardium. Am J Pathol 1995, 146:419–428.
Irwin MW, Mak S, Mann DL, et al.: Tissue expression and immunolocalization of tumor necrosis factor-alpha in postinfarction dysfunctional myocardium. Circulation 1999, 99:1492–1498.
Satoh M, Nakamura M, Tamura G, et al.: Inducible nitric oxide synthase and tumor necrosis factor-alpha in myocardium in human dilated cardiomyopathy. J Am Coll Cardiol 1997, 29:716–724.
Satoh M, Tamura G, Segawa I, et al.: Expression of cytokine genes and presence of enteroviral genomic RNA in endomyocardial biopsy tissues of myocarditis and dilated cardiomyopathy. Virchows Arch 1996, 427:503–509.
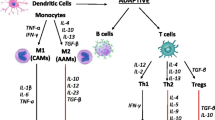
Rossi M, Young JW: Human dendritic cells: potent antigenpresenting cells at the crossroads of innate and adaptive immunity. J Immunol 2005, 175:1373–1381.
Medzhitov R, Janeway C Jr.: Innate immunity. N Engl J Med 2000, 343:338–344.
Anker SD, Coats AJ: Cardiac cachexia: a syndrome with impaired survival and immune and neuroendocrine activation. Chest 1999, 115:836–847.
Rock FL, Hardiman G, Timans JC, et al.: A family of human receptors structurally related to Drosophila Toll. Proc Natl Acad Sci U S A 1998, 95:588–593.
Medzhitov R, Preston-Hurlburt P, Janeway CA Jr: A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature 1997, 388:394–397.
Takeda K, Kaisho T, Akira S: Toll-like receptors. Annu Rev Immunol 2003, 21:335–376.
Satoh M, Tamura G, Segawa I, et al.: Enteroviral RNA in dilated cardiomyopathy. Eur Heart J 1994, 15:934–939.
Kurt-Jones E, Popova L, Kwinn L, et al.: Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nat Immunol 2000, 5:398–401.
Satoh M, Nakamura M, Akatsu T, et al.: Toll-like receptor 4 is expressed with enteroviral replication in myocardium from patients with dilated cardiomyopathy. Lab Invest 2004, 84:173–181.
Heil F, Hemmi H, Hochrein H, et al.: Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science 2004, 303:1526–1529.
Diebold SS, Kaisho T, Hemmi H, et al.: Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 2004, 303:1529–1531.
Triantafilou K, Orthopoulos G, Vakakis E, et al.: Human cardiac inflammatory responses triggered by Coxsackie B viruses are mainly Toll-like receptor (TLR) 8-dependent. Cell Microbiol 2005, 7:1117–1126.
Satoh M, Akatsu T, Ishikawa Y, et al.: Association between toll-like receptor 8 expression and adverse clinical outcomes in patients with enterovirus-associated dilated cardiomyopathy. Am Heart J 2007, 154:581–588.
Medzhitov R: Toll-like receptors and innate immunity. Nat Rev Immunol 2001, 1:135–145.
Napolitani G, Rinaldi A, Bertoni F, et al.: Selected Toll-like receptor agonist combinations synergistically trigger a T helper type 1-polarizing program in dendritic cells. Nat Immunol 2005, 6:679–776.
Maekawa Y, Anzai T, Yoshikawa T, et al.: Prognostic significance of peripheral monocytosis after reperfused acute myocardial infarction: a possible role for left ventricular remodeling. J Am Coll Cardiol 2002, 39:241–246.
Furman MI, Gore JM, Anderson FA, et al.: Elevated leukocyte count and adverse hospital events in patients with acute coronary syndromes: findings from the Global Registry of Acute Coronary Events (GRACE). Am Heart J 2004, 147:42–48.
Tashiro H, Shimokawa H, Yamamoto K, et al.: Monocyterelated cytokines in acute myocardial infarction. Am Heart J 1995, 130:446–452.
Marx N, Neumann FJ, Ott I, et al.: Induction of cytokine expression in leukocytes in acute myocardial infarction. J Am Coll Cardiol 1997, 30:165–170.
Oyama J, Blais C Jr, Liu X, et al.: Reduced myocardial ischemia-reperfusion injury in toll-like receptor 4-deficient mice. Circulation 2004, 109:784–789.
Satoh M, Shimoda Y, Maesawa C, et al.: Activated toll-like receptor 4 in monocytes is associated with heart failure after acute myocardial infarction. Int J Cardiol 2006, 109:226–234.
Bryant D, Becker L, Richardson J, et al.: Cardiac failure in transgenic mice with myocardial expression of tumor necrosis factor-alpha. Circulation 1998, 97:1375–1381.
Hirota H, Yoshida K, Kishimoto T, Taga T: Continuous activation of gp130, a signal-transducing receptor component for interleukin 6-related cytokines, causes myocardial hypertrophy in mice. Proc Natl Acad Sci U S A 1995, 92:4862–4866.
Vabulas RM, Ahmad-Nejad P, Ghose S, et al.: HSP70 as endogenous stimulus of the Toll/interleukin-1 receptor signal pathway. J Biol Chem 2002, 277:15107–15112.
Satoh M, Shimoda Y, Akatsu T, et al.: Elevated circulating levels of heat shock protein 70 are related to systemic inflammatory reaction through monocyte Toll signal in patients with heart failure after acute myocardial infarction. Eur J Heart Fail 2006, 8:810–815.
Mullberg J, Althoff K, Jostock T, et al.: The importance of shedding of membrane proteins for cytokine biology. Eur Cytokine Netw 2000, 11:27–38.
Black RA, Rauch CT, Kozlosky CJ, et al.: A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 1997, 385:729–733.
Moss ML, Jin SL, Milla ME, et al.: Cloning of a disintegrin metalloproteinase that processes precursor tumour-necrosis factor-alpha. Nature 1997, 385:733–736.
Satoh M, Nakamura M, Satoh H, et al.: Expression of tumor necrosis factor-alpha-converting enzyme and tumor necrosis factor-alpha in human myocarditis. J Am Coll Cardiol 2000, 36:1288–1294.
Tsakadze NL, Sithu SD, Sen U, et al.: Tumor necrosis factor-alpha-converting enzyme (TACE/ADAM-17) mediates the ectodomain cleavage of intercellular adhesion molecule-1 (ICAM-1). J Biol Chem 2006, 281:3157–3164.
Satoh M, Nakamura M, Saitoh H, et al.: Tumor necrosis factor-alpha-converting enzyme and tumor necrosis factor-alpha in human dilated cardiomyopathy. Circulation 1999, 99:3260–3265.
Satoh M, Iwasaka J, Nakamura M, et al.: Increased expression of tumor necrosis factor-alpha converting enzyme and tumor necrosis factor-alpha in peripheral blood mononuclear cells in patients with advanced congestive heart failure. Eur J Heart Fail 2004, 6:869–875.
Diwan A, Dibbs Z, Nemoto S, et al.: Targeted overexpression of noncleavable and secreted forms of tumor necrosis factor provokes disparate cardiac phenotypes. Circulation 2004, 109:262–268.
Akatsu T, Nakamura M, Satoh M, Hiramori K: Increased mRNA expression of tumour necrosis factor-alpha and its converting enzyme in circulating leucocytes of patients with acute myocardial infarction. Clin Sci (Lond) 2003, 105:39–44.
Ghezzi P, Dinarello CA, Bianchi M, et al.: Hypoxia increases production of interleukin-1 and tumor necrosis factor by human mononuclear cells. Cytokine 1991, 3:189–194.
Spengler RN, Allen RM, Remick DG, et al.: Stimulation of alpha-adrenergic receptor augments the production of macrophage-derived tumor necrosis factor. J Immunol 1990, 145:1430–1434.
Jensen JC, Buresh C, Norton JA: Lactic acidosis increases tumor necrosis factor secretion and transcription in vitro. J Surg Res 1990, 49:350–353.
Grossman GB, Rohde LE, Clausell N: Evidence for increased peripheral production of tumor necrosis factor-alpha in advanced congestive heart failure. Am J Cardiol 2001, 88:578–581.
Boyle JJ, Bowyer DE, Weissberg PL, Bennett MR: Human blood-derived macrophages induce apoptosis in human plaque-derived vascular smooth muscle cells by Fas-ligand/Fas interactions. Arterioscler Thromb Vasc Biol 2001, 21:1402–1407.
Maier W, Altwegg LA, Corti R, et al.: Inflammatory markers at the site of ruptured plaque in acute myocardial infarction: locally increased interleukin-6 and serum amyloid A but decreased C-reactive protein. Circulation 2005, 111:1355–1361.
Satoh M, Ishikawa Y, Itoh T, et al.: The expression of TNF-alpha converting enzyme at the site of ruptured plaques in patients with acute myocardial infarction. Eur J Clin Invest 2008, 38:97–105.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Satoh, M., Minami, Y., Takahashi, Y. et al. Immune modulation: Role of the inflammatory cytokine cascade in the failing human heart. Curr Heart Fail Rep 5, 69–74 (2008). https://doi.org/10.1007/s11897-008-0012-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11897-008-0012-2