Abstract
To understand the dynamics of oxidation of Nb, we examine the adsorption, absorption, and diffusion of an oxygen atom on, in, and into Nb(110) and Nb(100) surfaces, respectively, using density functional theory. Our calculations predict that the oxygen atom adsorbs on the threefold site on Nb(110) and the fourfold hollow site on Nb(100), and the adsorption energy is −5.08 eV and −5.18 eV, respectively. We find the long and short bridge sites to be transition states for O diffusion on Nb(110), while the on-top site is a rank-2 saddle point. In the subsurface region, the oxygen atom prefers the octahedral site, as in bulk niobium. Our results show also that the O atom is more stable on Nb(110) subsurface than on Nb(100) subsurface. The diffusion of oxygen atoms into niobium surfaces passes through transition states where the oxygen atom is coordinated to four niobium atoms. The diffusion barriers of the oxygen atom into Nb(110) and Nb(100) are 1.81 eV and 2.05 eV, respectively. An analysis of the electronic density of states reveals the emergence of well-localized electronic states below the lowest states of clean Nb surfaces due to d–p orbital hybridization.







Similar content being viewed by others
References
I.E. Wachs, L.E. Briand, J.-M. Jehng, L. Burcham, and X. Gao, Catal. Today 57, 323 (2000).
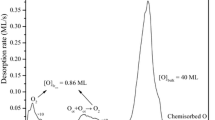
M. Grundner and J. Halbritter, J. Appl. Phys. 51, 397 (1980).
J.-I. Shirakashi, K. Matsumoto, N. Miura, and M. Konagai, J. Appl. Phys. 83, 5567 (1998).
B.P. Bewlay, M.R. Jackson, J.C. Zhao, and P.R. Subramanian, Metall. Mater. Trans. A 34A, 2043 (2003).
R. Pantel, M. Bujor, and J. Bardolle, Surf. Sci. 62, 589 (1977).
S. Usami, N. Tominaga, and T. Nakajima, Vacuum 27, 11 (1977).
H.H. Farrell and M. Strongin, Surf. Sci. 38, 18 (1973).
W.-S. Lo, H.-H. Chen, T.-S. Chien, C.-C. Tsan, and B.-S. Fang, Surf. Rev. Lett. 4, 651 (1997).
B. An, S. Fukuyama, K. Yokogawa, and M. Yoshimura, Phys. Rev. B 68, 115423 (2003).
R. Franchy, T.U. Bartke, and P. Gassmann, Surf. Sci. 366, 60 (1996).
C. Sürgers, M. Schöck, and H. von Löhneysen, Surf. Sci. 471, 209 (2001).
I. Arfaoui, J. Cousty, and H. Safa, Phys. Rev. B 65, 115413 (2002).
M. Wen, B. An, S. Fukuyama, and K. Yokogawa, Surf. Sci. 603, 216 (2009).
J.R. Sambrano, J. Andrés, A. Beltrán, F. Sensato, and E. Longo, Chem. Phys. Lett. 287, 620 (1998).
A. Fielicke, G. Meijer, and G. von Helden, J. Am. Chem. Soc. 125, 3659 (2003).
D.A. Kilimis and C.E. Lekka, Mater. Sci. Eng. B 144, 27 (2007).
G. Kresse and J. Furthmüller, Phys. Rev. B 54, 11169 (1996).
G. Kresse and J. Furthmüller, Comput. Mater. Sci. 6, 15 (1996).
G. Kresse and D. Joubert, Phys. Rev. B 59, 1758 (1999).
M. Methfessel and A.T. Paxton, Phys. Rev. B 40, 3616 (1989).
H.J. Monkhorst and J.D. Pack, Phys. Rev. B 13, 5188 (1976).
R. Roberge, J. Less Common Met. 40, 161 (1975).
C. Kittel, Introduction to Solid State Physics, 7th ed. (New York: Wiley, 1996).
M.R. Fellinger, H. Park, and J.W. Wilkins, Phys. Rev. B 81, 144119 (2010).
D. Nguyen-Manh, A.P. Horsfield, and S.L. Dudarev, Phys. Rev. B 73, 020101 (2006).
J. Neugebauer and M. Scheffler, Phys. Rev. B 46, 16067 (1992).
G. Henkelman, B.P. Uberuaga, and H. Jonsson, J. Chem. Phys. 113, 9901 (2000).
K.E. Yoon, D.N. Seidman, C. Antoine, and P. Bauer, Appl. Phys. Lett. 93, 132502 (2008).
C.E. Lekka, M.J. Mehl, N. Bernstein, and D.A. Papaconstantopoulos, Phys. Rev. B 68, 035422 (2003).
X. Pan, M.W. Ruckman, and M. Strongin, Phys. Rev. B 35, 3734 (1987).
J.K. Burdett and T. Hughbanks, J. Am. Chem. Soc. 106, 3101 (1984).
Acknowledgements
This technical effort was performed in support of the National Energy Technology Laboratory’s (NETL) ongoing research on Turbines Materials Development for Oxy-combustion Environments under the RES Contract DE-FE-0004000. This work used the computing facility at the Texas Advanced Computing Center (TACC) through Award# DMR120048 by the Extreme Science and Engineering Discovery Environment (XSEDE), which is supported by National Science Foundation grant number OCI-1053575. “This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.”
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tafen, D.N., Gao, M.C. Oxygen Atom Adsorption on and Diffusion into Nb(110) and Nb(100) from First Principles. JOM 65, 1473–1481 (2013). https://doi.org/10.1007/s11837-013-0735-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-013-0735-8