Abstract
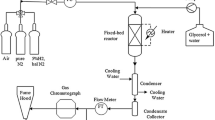
A kinetics model that takes the synergetic effect of carbon dioxide fraction on the methanol production rate into account is applied to the development of a mathematical model for the bench-scale reactor. A comparison between the simulation results and the experimental data corroborates the validity of the model. Several optimization strategies are suggested to maximize the methanol yield, among which the utilization of piecewise trajectories for wall temperature along the reactor axis as well as the optimal CO2 fraction at the inlet of the reactor is found to be the best strategy in the sense of methanol production per unit amount of the feed, in such a way that the optimization strategy considers the variation of the reaction temperature in the reactor and maximizes the synergetic effect on the production rate by the addition of carbon dioxide.
Similar content being viewed by others
References
G. A. Olah, H. Doggweiler, J. D. Felberg, S. Frohlich, M. J. Grdina, R. Karpeles, T. Keumi, S. Inaba, W. M. Ip, K. Lammerstsmak, G. Salem and D. C. Tabor, J. Am. Chem. Soc., 106, 2143 (1984).
G. A. Olah, Angew. Chem. Int. Ed., 44, 2636 (2005).
G. K. S. Prakash, M. C. Smart, Q.-J. Wang, A. Atti, V. Pleynet, B. Yang, K. McGrath, G. A. Olah, S. R. Narayanan, W. Chun, T. Valdez and S. Surampudi, J. Fluorine Chem., 125, 1217 (2004).
J. P. Lange, Catal. Today, 64, 3 (2001).
C. N. Satterfield, Heterogeneous catalysis in industrial practice, McGraw-Hill, New York (1991).
R. G. Herman, K. Klier, G. W. Simmons, B. P. Finn and J. B. Bulko, J. Catal., 56, 407 (1979).
G. C. Chinchen, P. S. Denny, D. G. Parger, M. S. Spenser and D. A. Whan, Appl. Catal., 30, 333 (1987).
B. Denise and R. P. A. Sneeden, J. Mol. Catal., 17, 359 (1982).
K. Klier, V. Chatikavanij, R. G. Herman and G. W. Simmons, J. Catal., 74, 343 (1982).
J. F. Edwards and G. L. Schrader, J. Phys. Chem., 88, 5624 (1984).
A. Coteron and A. N. Hayhurst, Chem. Eng. Sci., 49, 209 (1994).
M. A. McNeil, C. J. Schack and R. G. Rinker, Appl. Catal., 50, 265 (1989).
H.-W. Lim, M.-J. Park, S.-H. Kang, H.-J. Chae, J. W. Bae and K.-W. Jun, Ind. Eng. Chem. Res., 48, 10448 (2009).
I. Løvik, M. Hillestad and T. Hertzberg, Comput. Chem. Eng., 22, S707 (1998).
H. Kordabadi and A. Jahanmiri, Chem. Eng. J., 108, 249 (2005).
M. R. Rahimpour and M. Lotfinejad, Chem. Eng. Technol., 30, 1062 (2007).
M. R. Rahimpour and H. Elekaei Behjatia, Fuel Process. Technol., 90, 279 (2009).
G. H. Graaf, P. J. J. M. Sijtsema, E. J. Stamhuis and G. E. H. Joosten, Chem. Eng. Sci., 41, 2883 (1986).
P. Mizsey, E. Newson, T. Truong and P. Hottinger, Appl. Catal. A: Gen., 213, 233 (2001).
K. L. Ng, D. Chadwick and B. A. Toseland, Chem. Eng. Sci., 54, 3587 (1999).
H. Kordabadi and A. Jahanmiri, Chem. Eng. Process., 46, 1299 (2007).
H. J. Chae, S. T. Choo, H. I. Choi and S. Nam, Ind. Eng. Chem. Res., 39, 1159 (2000).
T. H. Chilton and A. P. Colburn, Ind. Eng. Chem., 26, 1183 (1934).
R. H. Perry and D. W. Green, Perry’s Chemical Engineers’ Handbook, McGraw-Hill, New York (1997).
B. J. Lommerts, G. H. Graaf and A. A. C. M. Beenackers, Chem. Eng. Sci., 55, 5589 (2000).
R. Suwanwarangkul, E. Croiset, M. W. Fowler, P. L. Douglas, E. Entchev and M. A. Douglas, J. Power Sources, 122, 9 (2003).
E. N. Fuller, P. D. Schettler and J. C. Gidding, Ind. Eng. Chem., 58, 19 (1966).
K. R. Westerterp, W. P. M. Van Swaaij and A. A. C. M. Beenackers, Chemical reactor design and operation, Wiley, Chichester (1987).
D. E. Goldberg, Genetic Algorithms in Search, Optimization, and Machine Learning, Addison-Wesley, Reading (1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lim, HW., Jun, H.J., Park, MJ. et al. Optimization of methanol synthesis reaction on Cu/ZnO/Al2O3/ZrO2 catalyst using genetic algorithm: Maximization of the synergetic effect by the optimal CO2 fraction. Korean J. Chem. Eng. 27, 1760–1767 (2010). https://doi.org/10.1007/s11814-010-0311-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-010-0311-7