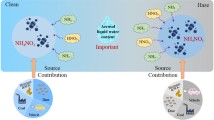
Abstract
Nitrate is an important component of atmospheric particulate matter and affects air quality, climate, human health, and the ecosystem. Nitrate was previously considered a permanent sink for nitrogen oxides (NOx). However, this viewpoint has been challenged in recent years because growing research evidence has shown the transformation of nitrate into NOx (i.e., renoxification). The photolysis of nitrate/HNO3, especially in the particulate phase or adsorbed on particles, can be a significant renoxification process in the atmosphere. The formation and photolysis of nitrate in aerosol not only change the diurnal variation of NOx, but also provide long-distance transport of NOx in the form of nitrate, which affects local and regional atmospheric chemistry and air quality. This review summarizes recent advances in the fundamental understanding of the photolysis of nitrate/HNO3 under various atmospheric conditions, with a focus on mechanisms and key factors affecting the process. The atmospheric implications are discussed and future research is recommended.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Acker K, Moller D, Wieprecht W, Meixner F X, Bohn B, Gilge S, Plass-Dulmer C, Berresheim H (2006). Strong daytime production of OH from HNO2 at a rural mountain site. Geophysical Research Letters, 33(2): L02809
Alif A, Boule P (1991). Photochemistry and environment.14. phototransformation of nitrophenols induces by excitation of nitrite and nitrate ions. Journal of Photochemistry and Photobiology A-Chemistry, 59(3): 357–367
Atkinson R, Baulch D L, Cox R A, Crowley J N, Hampson R F, Hynes R G, Jenkin M E, Rossi M J, Troe J (2004). Evaluated kinetic and photochemical data for atmospheric chemistry: Volume I: Gas phase reactions of Ox, HOx, NOx and SOx species. Atmospheric Chemistry and Physics, 4: 1461–1738
Baergen A M, Donaldson D J (2013). Photochemical renoxification of nitric acid on real urban grime. Environmental Science & Technology, 47(2): 815–820
Baergen A M, Donaldson D J (2016). Formation of reactive nitrogen oxides from urban grime photochemistry. Atmospheric Chemistry and Physics, 16(10): 6355–6363
Bao F, Jiang H, Zhang Y, Li M, Ye C, Wang W, Ge M, Chen C, Zhao J (2020). The key role of sulfate in the photochemical renoxification on real PM2.5. Environmental Science & Technology, 54(6): 3121–3128
Bao F, Li M, Zhang Y, Chen C, Zhao J (2018). Photochemical aging of Beijing urban PM2.5: HONO production. Environmental Science & Technology, 52(11): 6309–6316
Benedict K B, McFall A S, Anastasio C (2017). Quantum yield of nitrite from the photolysis of aqueous nitrate above 300 nm. Environmental Science & Technology, 51(8): 4387–4395
Blanchard C L, Tanenbaum S, Hidy G M (2007). Effects of sulfur dioxide and oxides of nitrogen emission reductions on fine particulate matter mass concentrations: regional comparisons. Journal of the Air & Waste Management Association, 57(11): 1337–1350
Blaszczak-Boxe C S, Saiz-Lopez A (2018). Nitrate photolysis in ice and snow: a critical review of its multiphase chemistry. Atmospheric Environment, 193: 224–241
Brezonik P L, Fulkerson-Brekken J (1998). Nitrate-induced photolysis in natural waters: Controls controls on concentrations of hydroxyl radical photo-intermediates by natural scavenging agents. Environmental Science & Technology, 32(19): 3004–3010
Burkholder J B, Talukdar R K, Ravishankara A R, Solomon S (1993). Temperature-dependence of the HNO3 UV absorption cross-sections. Journal of Geophysical Research, 98(D12): 22937–22948
Chang W L, Bhave P V, Brown S S, Riemer N, Stutz J, Dabdub D (2011). Heterogeneous atmospheric chemistry, ambient measurements, and model calculations of N2O5: a review. Aerosol Science and Technology, 45(6): 665–695
Chang W L, Bhave P V, Brown S S, Riemer N, Stutz J, Dabdub D (2011). Heterogeneous atmospheric chemistry, ambient measurements, and model calculations of N2O5: a review. Aerosol Science and Technology, 45(6): 665–695
Cheng Y, Yu Q, Liu J, Sun Y, Liang L, Du Z, Geng G, Ma W, Qi H, Zhang Q, He K (2022). Formation of secondary inorganic aerosol in a frigid urban atmosphere. Frontiers of Environmental Science & Engineering, 16(2): 18
Du J, Zhu L (2011). Quantification of the absorption cross sections of surface-adsorbed nitric acid in the 335–365 nm region by Brewster angle cavity ring-down spectroscopy. Chemical Physics Letters, 511(4–6): 213–218
Dubowski Y, Colussi A J, Hoffmann M R (2001). Nitrogen dioxide release in the 302 nm band photolysis of spray-frozen aqueous nitrate solutions. Atmospheric implications. Journal of Physical Chemistry A, 105(20): 4928–4932
Dyson J E, Boustead G A, Fleming L T, Blitz M, Stone D, Arnold S R, Whalley L K, Heard D E (2021). Production of HONO from NO2 uptake on illuminated TiO2 aerosol particles and following the illumination of mixed TiO2/ammonium nitrate particles. Atmospheric Chemistry and Physics, 21(7): 5755–5775
Elena G A, Matthias S, Sasho G, Sabina B, Vincent B, Bruno C, Cornelius Z, Henri W (2014). Light-induced nitrous acid (HONO) production from NO2 heterogeneous reactions on household chemicals. Atmospheric Environment, 95: 391–399
Elshorbany Y F, Kleffmann J, Kurtenbach R, Lissi E, Rubio M, Villena G, Gramsch E, Rickard A R, Pilling M J, Wiesen P (2010). Seasonal dependence of the oxidation capacity of the city of Santiago de Chile. Atmospheric Environment, 44(40): 5383–5394
Fairbrother D H, Sullivan D J D, Johnston H S (1997). Global thermodynamic atmospheric modeling: search for new heterogeneous reactions. Journal of Physical Chemistry A, 101(40): 7350–7358
Finlayson-Pitts B J (2003). The tropospheric chemistry of sea salt: a molecular-level view of the chemistry of NaCl and NaBr. Chemical Reviews, 103(12): 4801–4822
Finlayson-Pitts B J, Pitts J N (2000). Chemistry of the Upper and Lower Atmosphere. San Diego: Academic Press
Finlayson-Pitts B J, Wingen L M, Sumner A L, Syomin D, Ramazan K A (2003). The heterogeneous hydrolysis of NO2 in laboratory systems and in outdoor and indoor atmospheres: an integrated mechanism. Physical Chemistry Chemical Physics, 5(2): 223–242
Frey M M, Roscoe H K, Kukui A, Savarino J, France J L, King M D, Legrand M, Preunkert S (2015). Atmospheric nitrogen oxides (NO and NO2) at Dome C, East Antarctica, during the OPALE campaign. Atmospheric Chemistry and Physics, 15(14): 7859–7875
Gankanda A, Grassian V H (2014). Nitrate photochemistry on laboratory proxies of mineral dust aerosol: wavelength dependence and action spectra. Journal of Physical Chemistry C, 118(50): 29117–29125
Gao R S, Fahey D W, Del Negro L A, Donnelly S G, Keim E R, Neuman J A, Teverovskaia E, Wennberg P O, Hanisco T F, Lanzendorf E J, et al. (1999). A comparison of observations and model simulations of NOx/NOy in the lower stratosphere. Geophysical Research Letters, 26(8): 1153–1156
Gen M S, Zhang R F, Huang D D, Li Y J, Chan C K (2019b). Heterogeneous SO2 oxidation in sulfate formation by photolysis of particulate nitrate. Environmental Science & Technology Letters, 6(2): 86–91
Gen M, Liang Z, Zhang R, Go Mabato B R, Chan C K (2022). Particulate nitrate photolysis in the atmosphere. Environmental Science: Atmospheres, 2(2): 111–127
Gen M, Zhang R, Huang D D, Li Y, Chan C K (2019a). Heterogeneous oxidation of SO2 in sulfate production during nitrate photolysis at 300 nm: effect of pH, relative humidity, irradiation intensity, and the presence of organic compounds. Environmental Science & Technology, 53(15): 8757–8766
George C, Ndour M, Balkanski Y, Ka O (2007). Photoenhanced uptake of NO2 on mineral dust. In: NATO Science Series IV: Earth and Environmental Sciences, Marrakech, Morocco. Dordrecht: Springer
George C, Strekowski R S, Kleffmann J, Stemmler K, Ammann M (2005). Photoenhanced uptake of gaseous NO2 on solid organic compounds: a photochemical source of HONO? Faraday Discussions, 130: 195–210, discussion 241–264, 519–524
Goldstein S, Rabani J (2007). Mechanism of nitrite formation by nitrate photolysis in aqueous solutions: the role of peroxynitrite, nitrogen dioxide, and hydroxyl radical. Journal of the American Chemical Society, 129(34): 10597–10601
Goodman A L, Miller T M, Grassian V H (1998). Heterogeneous reactions of NO2 on NaCl and Al2O3 particles. Journal of Vacuum Science & Technology A-Vacuum Surfaces and Films, 16(4): 2585–2590
Gu F T, Hu M, Zheng J, Guo S (2017). Research progress on particulate organonitrates. Progress in Chemistry, 29(9): 962–969 (in Chinese)
Gustafsson R J, Orlov A, Griffiths P T, Cox R A, Lambert R M (2006). Reduction of NO2 to nitrous acid on illuminated titanium dioxide aerosol surfaces: implications for photocatalysis and atmospheric chemistry. Chemical Communications (Cambridge, England), 37: 3936–3938
Han C, Yang W, Wu Q, Yang H, Xue X (2016). Heterogeneous photochemical conversion of NO2 to HONO on the humic acid surface under simulated sunlight. Environmental Science & Technology, 50(10): 5017–5023
Han M, Jafarikojour M, Mohseni M (2021). The impact of chloride and chlorine radical on nitrite formation during vacuum UV photolysis of water. Science of the Total Environment, 760: 143325
Han M, Mohseni M (2020). Impact of organic and inorganic carbon on the formation of nitrite during the VUV photolysis of nitrate containing water. Water Research, 168: 115169
Herrmann H, Ervens B, Nowacki P, Wolke R, Zellner R (1999). A chemical aqueous phase radical mechanism for tropospheric chemistry. Chemosphere, 38(6): 1223–1232
Honrath R E, Lu Y, Peterson M C, Dibb J E, Arsenault M A, Cullen N J, Steffen K (2002). Vertical fluxes of NOx, HONO, and HNO3 above the snowpack at Summit, Greenland. Atmospheric Environment, 36(15–16): 2629–2640
Honrath R E, Peterson M C, Guo S, Dibb J E, Shepson P B, Campbell B (1999). Evidence of NOx production within or upon ice particles in the Greenland snowpack. Geophysical Research Letters, 26(6): 695–698
Huang R J, Yang L, Cao J, Wang Q, Tie X, Ho K F, Shen Z, Zhang R, Li G, Zhu C, et al. (2017). Concentration and sources of atmospheric nitrous acid (HONO) at an urban site in Western China. Science of the Total Environment, 593–594: 165–172
Jiang N, Guo Y, Wang Q, Kang P R, Zhang R Q, Tang X Y (2017). Chemical composition characteristics of PM2.5 in three cities in Henan, Central China. Aerosol and Air Quality Research, 17(10): 2367–2380
Johnston H S, Chang S G, Whitten G (1974). Photolysis of nitric-acid vapor. Journal of Physical Chemistry, 78(1): 1–7
Kenner R D, Rohrer F, Papenbrock T, Stuhl F (1986). Excitation mechanism for OH(A) in the ARF excimer laser photolysis of nitric-acid. Journal of Physical Chemistry, 90(7): 1294–1299
Kiendler-Scharr A, Mensah A A, Friese E, Topping D, Nemitz E, Prevot A S H, Aijala M, Allan J, Canonaco F, Canagaratna M, Carbone S, et al. (2016). Ubiquity of organic nitrates from nighttime chemistry in the European submicron aerosol. Geophysical Research Letters, 43(14): 7735–7744
Kleffmann J (2007). Daytime sources of nitrous acid (HONO) in the atmospheric boundary layer. ChemPhysChem, 8(8): 1137–1144
Kleffmann J, Benter T, Wiesen P (2004). Heterogeneous reaction of nitric acid with nitric oxide on glass surfaces under simulated atmospheric conditions. Journal of Physical Chemistry A, 108(27): 5793–5799
Lary D J, Shallcross D E (2000). Potential importance of the reaction CO+HNO3. Journal of Geophysical Research, 105(D9): 11617–11623
Lee B H, Mohr C, Lopez-Hilfiker F D, Lutz A, Hallquist M, Lee L, Romer P, Cohen R C, Iyer S, Kurtén T, et al. (2016). Highly functionalized organic nitrates in the southeast United States: contribution to secondary organic aerosol and reactive nitrogen budgets. Proceedings of the National Academy of Sciences of the United States of America, 113(6): 1516–1521
Lee T, Yu X Y, Ayres B, Kreidenweis S M, Malm W C, Collett J L Jr (2008). Observations of fine and coarse particle nitrate at several rural locations in the United States. Atmospheric Environment, 42(11): 2720–2732
Leriche M, Voisin D, Chaumerliac N, Monod A, Aumont B (2000). A model for tropospheric multiphase chemistry: application to one cloudy event during the CIME experiment. Atmospheric Environment, 34(29–30): 5015–5036
Li H Y, Zhang Q, Zheng B, Chen C R, Wu N N, Guo H Y, Zhang Y X, Zheng Y X, Li X, He K B (2018). Nitrate-driven urban haze pollution during summertime over the North China Plain. Atmospheric Chemistry and Physics, 18(8): 5293–5306
Li S, Matthews J, Sinha A (2008). Atmospheric hydroxyl radical production from electronically excited NO2 and H2O. Science, 319(5870): 1657–1660
Li X, Rohrer F, Hofzumahaus A, Brauers T, Häseler R, Bohn B, Broch S, Fuchs H, Gomm S, Holland F, et al. (2014). Missing gas-phase source of HONO inferred from Zeppelin measurements in the troposphere. Science, 344(6181): 292–296
Liang Z, Zhang R, Gen M, Chu Y, Chan C K (2021). Nitrate photolysis in mixed sucrose-nitrate-sulfate particles at different relative humidities. Journal of Physical Chemistry A, 125(17): 3739–3747
Liu J Y, Liu Z R, Ma Z Q, Yang S H, Yao D, Zhao S M, Hu B, Tang G Q, Sun J, Cheng M T, et al. (2021). Detailed budget analysis of HONO in Beijing, China: implication on atmosphere oxidation capacity in polluted megacity. Atmospheric Environment, 244: 117957
Logager T, Sehested K (1993). Formation and decay of peroxynitric acid: a pulse-radiolysis study. Journal of Physical Chemistry, 97(39): 10047–10052
Logan J A, Prather M J, Wofsy S C, Mcelroy M B (1981). Tropospheric chemistry: a global perspective. Journal of Geophysical Research, 86(NC8): 7210–7254
Ma Q X, Zhong C, Ma J Z, Ye C X, Zhao Y Q, Liu Y, Zhang P, Chen T Z, Liu C, Chu B W, He H (2021). Comprehensive study about the photolysis of nitrates on mineral oxides. Environmental Science & Technology, 55(13): 8604–8612
Mack J, Bolton J R (1999). Photochemistry of nitrite and nitrate in aqueous solution: a review. Journal of Photochemistry and Photobiology a-Chemistry, 128(1–3): 1–13
Maria H J, Mcdonald J R, Mcglynn S P (1973). Electronic absorptionspectrum of nitrate ion and boron trihalides. Journal of the American Chemical Society, 95(4): 1050–1056
Mark G, Korth H G, Schuchmann H P, Vonsonntag C (1996). The photochemistry of aqueous nitrate ion revisited. Journal of Photochemistry and Photobiology A-Chemistry, 101(2–3): 89–103
McFall A S, Edwards K C, Anastasio C (2018). Nitrate photochemistry at the air-ice interface and in Oher Ice reservoirs. Environmental Science & Technology, 52(10): 5710–5717
Miller T M, Grassian V H (1998). Heterogeneous chemistry of NO2 on mineral oxide particles: spectroscopic evidence for oxide-coordinated and water-solvated surface nitrate. Geophysical Research Letters, 25(20): 3835–3838
Mochida M, Finlayson-Pitts B J (2000). FTIR studies of the reaction of gaseous NO with HNO3 on porous glass: Implications for conversion of HNO3 to photochemically active NOx in the atmosphere. Journal of Physical Chemistry A, 104(43): 9705–9711
Monge M E, D’Anna B, Mazri L, Giroir-Fendler A, Ammann M, Donaldson D J, George C (2010). Light changes the atmospheric reactivity of soot. Proceedings of the National Academy of Sciences of the United States of America, 107(15): 6605–6609
Morenz K J, Shi Q, Murphy J G, Donaldson D J (2016). Nitrate photolysis in salty snow. Journal of Physical Chemistry A, 120(40): 7902–7908
Neuman J A, Nowak J B, Brock C A, Trainer M, Fehsenfeld F C, Holloway J S, Hubler G, Hudson P K, Murphy D M, Nicks D K, Orsini D, Parrish D D, Ryerson T B, Sueper D T, Sullivan A, Weber R (2003). Variability in ammonium nitrate formation and nitric acid depletion with altitude and location over California. Journal of Geophysical Research, 108(D17): 4557
Oswald R, Ermel M, Hens K, Novelli A, Ouwersloot H G, Paasonen P, Petaja T, Sipila M, Keronen P, Back J, et al. (2015). A comparison of HONO budgets for two measurement heights at a field station within the boreal forest in Finland. Atmospheric Chemistry and Physics, 15(2): 799–813
Pandit S, Garcia SLM, Grassian VH (2021). HONO production from gypsum surfaces following exposure to NO2 and HNO3: Roles of relative humidity and light source. Environmental Science & Technology, 55(14): 9761–9772
Peng X, Wang T, Wang W H, Ravishankara A R, George C, Xia M, Cai M, Li Q Y, Salvador C M, Lau C, et al. (2022). Photodissociation of particulate nitrate as a source of daytime tropospheric Cl2. Nature Communications, 13(1): 939
Perkins K K, Hanisco T F, Cohen R C, Koch L C, Stimpfle R M, Voss P B, Bonne G P, Lanzendorf E J, Anderson J G, Wennberg P O, et al. (2001). The NOx−HNO3 system in the lower stratosphere: insights from in situ measurements and implications of the J(HNO3)-OH relationship. Journal of Physical Chemistry A, 105(9): 1521–1534
Ravishankara A R (1997). Heterogeneous and multiphase chemistry in the troposphere. Science, 276(5315): 1058–1065
Richards-Henderson N K, Anderson C, Anastasio C, Finlayson-Pitts B J (2015). The effect of cations on NO2 production from the photolysis of aqueous thin water films of nitrate salts. Physical Chemistry Chemical Physics, 17(48): 32211–32218
Rivera-Figueroa A M, Finlayson-Pitts B J (2003). Nitric acid “renoxification” in the troposphere: from a modeling myth to a laboratory reality. In: American Meteorological Society 83rd Annual Meeting, California. Boston: American Meteorological Society
Rivera-Figueroa A M, Sumner A L, Finlayson-Pitts B J (2003). Laboratory studies of potential mechanisms of renoxification of tropospheric nitric acid. Environmental Science & Technology, 37(3): 548–554
Roberts J M (1990). The atmospheric chemistry of organic nitrates. Atmospheric Environment Part A-General Topics, 24(2): 243–287
Roca M, Zahardis J, Bone J, El-Maazawi M, Grassian V H (2008). 310 nm irradiation of atmospherically relevant concentrated aqueous nitrate solutions: nitrite production and quantum yields. Journal of Physical Chemistry A, 112(51): 13275–13281
Rollins A W, Browne E C, Min K E, Pusede S E, Wooldridge P J, Gentner D R, Goldstein A H, Liu S, Day D A, Russell L M, et al. (2012). Evidence for NOx control over nighttime SOA formation. Science, 337(6099): 1210–1212
Russell A G, Cass G R, Seinfeld J H (1986). On some aspects of nighttime atmospheric chemistry. Environmental Science & Technology, 20(11): 1167–1172
Scharko N K, Berke A E, Raff J D (2014). Release of nitrous acid and nitrogen dioxide from nitrate photolysis in acidic aqueous solutions. Environmental Science & Technology, 48(20): 11991–12001
Schuttlefield J, Rubasinghege G, El-Maazawi M, Bone J, Grassian V H (2008). Photochemistry of adsorbed nitrate. Journal of the American Chemical Society, 130(37): 12210–12211
Schwartz-Narbonne H, Jones S H, Donaldson D J (2019). Indoor lighting releases gas phase nitrogen oxides from indoor painted surfaces. Environmental Science & Technology Letters, 6(2): 92–97
Seinfeld J H, Pandis S N (2016). Atmospheric Chemistry and Physics: from Air Pollution to Climate Change. Hoboken: John Wiley & Sons
Shang D, Peng J, Guo S, Wu Z, Hu M (2021). Secondary aerosol formation in winter haze over the Beijing-Tianjin-Hebei Region, China. Frontiers of Environmental Science & Engineering, 15(2): 34
Shang H, Chen Z, Wang X, Li M, Li H, Mao C, Yu L, Sun J, Ai Z, Zhang L (2022). SO2-enhanced nitrate photolysis on TiO2 minerals: a vital role of photochemically reactive holes. Applied Catalysis B: Environmental, 308: 121217
Shi Q, Tao Y, Krechmer J E, Heald C L, Murphy J G, Kroll J H, Ye Q (2021). Laboratory investigation of renoxification from the photolysis of inorganic particulate nitrate. Environmental Science & Technology, 55(2): 854–861
Sörgel M, Regelin E, Bozem H, Diesch J M, Drewnick F, Fischer H, Harder H, Held A, Hosaynali-Beygi Z, Martinez M, Zetzsch C (2011). Quantification of the unknown HONO daytime source and its relation to NO2. Atmospheric Chemistry and Physics, 11(20): 10433–10447
Spindler G, Gruner A, Muller K, Schlimper S, Herrmann H (2013). Long-term size-segregated particle (PM10, PM2.5, PM1) characterization study at Melpitz-influence of air mass inflow, weather conditions and season. Journal of Atmospheric Chemistry, 70(2): 165–195
Stemmler K, Ammann M, Donders C, Kleffmann J, George C (2006). Photosensitized reduction of nitrogen dioxide on humic acid as a source of nitrous acid. Nature, 440(7081): 195–198
Sun Y L, Wang Z F, Du W, Zhang Q, Wang Q Q, Fu P Q, Pan X L, Li J, Jayne J, Worsnop D R (2015). Long-term real-time measurements of aerosol particle composition in Beijing, China: seasonal variations, meteorological effects, and source analysis. Atmospheric Chemistry and Physics, 15(17): 10149–10165
Tsai C, Spolaor M, Colosimo S F, Pikelnaya O, Cheung R, Williams E, Gilman J B, Lerner B M, Zamora R J, Warneke C, et al. (2018). Nitrous acid formation in a snow-free wintertime polluted rural area. Atmospheric Chemistry and Physics, 18(3): 1977–1996
Usher C R, Michel A E, Grassian V H (2003). Reactions on mineral dust. Chemical Reviews, 103(12): 4883–4940
van Donkelaar A, Martin R V, Li C, Burnett R T (2019). Regional estimates of chemical composition of fine particulate matter using a combined geoscience-statistical method with information from satellites, models, and monitors. Environmental Science & Technology, 53(5): 2595–2611
Wagner I, Strehlow H, Busse G (1980). Flash-photolysis of nitrate ions in aqueous-solution. Zeitschrift Fur Physikalische Chemie, 123(1): 1–33
Wang X, Dalton E Z, Payne Z C, Perrier S, Riva M, Raff J D, George C (2021). Superoxide and nitrous acid production from nitrate photolysis is enhanced by dissolved aliphatic organic matter. Environmental Science & Technology Letters, 8(1): 53–58
Wang Y, Chen Y, Wu Z J, Shang D J, Bian Y X, Du Z F, Schmitt S H, Su R, Gkatzelis G I, Schlag P, et al. (2020). Mutual promotion between aerosol particle liquid water and particulate nitrate enhancement leads to severe nitrate-dominated particulate matter pollution and low visibility. Atmospheric Chemistry and Physics, 20(4): 2161–2175
Warneck P, Wurzinger C (1988). Product quantum yields for the 305 nm photodecomposition of nitrate in aqueous solution. Journal of Physical Chemistry, 92(22): 6278–6283
Wen L A, Chen J M, Yang L X, Wang X F, Xu C H, Sui X A, Yao L, Zhu Y H, Zhang J M, Zhu T, et al. (2015). Enhanced formation of fine particulate nitrate at a rural site on the North China Plain in summer: the important roles of ammonia and ozone. Atmospheric Environment, 101: 294–302
Xing J, Mathur R, Pleim J, Hogrefe C, Gan C M, Wong D C, Wei C, Gilliam R, Pouliot G (2015). Observations and modeling of air quality trends over 1990–2010 across the Northern Hemisphere: China, the United States and Europe. Atmospheric Chemistry and Physics, 15(5): 2723–2747
Xu Q, Wang S, Jiang J, Bhattarai N, Li X, Chang X, Qiu X, Zheng M, Hua Y, Hao J (2019). Nitrate dominates the chemical composition of PM2.5 during haze event in Beijing, China. Science of the Total Environment, 689: 1293–1303
Xu W, Wu Q, Liu X, Tang A, Dore A J, Heal M R (2016). Characteristics of ammonia, acid gases, and PM2.5 for three typical land-use types in the North China Plain. Environmental Science and Pollution Research International, 23(2): 1158–1172
Xu W, Yang W, Han C, Yang H, Xue X (2021). Significant influences of TiO2 crystal structures on NO2 and HONO emissions from the nitrates photolysis. Journal of Environmental Sciences-China, 102(4): 198–206
Yang W, Han C, Yang H, Xue X (2018). Significant HONO formation by the photolysis of nitrates in the presence of humic acids. Environmental Pollution, 243(Pt A): 679–686
Yang X, Luo F, Li J, Chen D, e Y, Lin W, Jin J (2019). Alkyl and aromatic nitrates in atmospheric particles determined by gas chromatography tandem mass spectrometry. Journal of the American Society for Mass Spectrometry, 30(12): 2762–2770
Yao X H, Lau A P S, Fang M, Chan C K, Hu M (2003). Size distributions and formation of ionic species in atmospheric particulate pollutants in Beijing, China: 1 — Inorganic ions. Atmospheric Environment, 37(21): 2991–3000
Ye C, Gao H, Zhang N, Zhou X (2016). Photolysis of nitric acid and nitrate on natural and artificial surfaces. Environmental Science & Technology, 50(7): 3530–3536
Ye C, Heard D E, Whalley L K (2017a). Evaluation of novel routes for NOx formation in remote regions. Environmental Science & Technology, 51(13): 7442–7449
Ye C, Zhang N, Gao H, Zhou X (2017b). Photolysis of particulate nitrate as a source of HONO and NOx. Environmental Science & Technology, 51(12): 6849–6856
Ye C, Zhang N, Gao H, Zhou X (2019). Matrix effect on surface-catalyzed photolysis of nitric acid. Scientific Reports, 9(1): 4351
Zafiriou O C, Bonneau R (1987). Wavelength-dependent quantum yield of OH radical formation from photolysis of nitrite ion in water. Photochemistry and Photobiology, 45(6): 723–727
Zellner R, Exner M, Herrmann H (1990). Absolute OH quantum yields in the laser photolysis of nitrate, nitrite and dissolved H2O2 at 308 and 351 nm in the temperature-range 278–353 K. Journal of Atmospheric Chemistry, 10(4): 411–425
Zepp R G, Hoigne J, Bader H (1987). Nitrate-induced photooxidation of trace organic chemicals in water. Environmental Science & Technology, 21(5): 443–450
Zhang N, Zhou X L, Shepson P B, Gao H L, Alaghmand M, Stirm B (2009). Aircraft measurement of HONO vertical profiles over a forested region. Geophysical Research Letters, 36(15): L15820
Zheng H T, Song S J, Sarwar G, Gen M S, Wang S X, Ding D, Chang X, Zhang S P, Xing J, Sun Y L, Ji D S, Chan C K, Gao J, McElroy M B (2020). Contribution of particulate nitrate photolysis to heterogeneous sulfate formation for winter haze in China. Environmental Science & Technology Letters, 7(9): 632–638
Zhou X L, Gao H L, He Y, Huang G, Bertman S B, Civerolo K, Schwab J (2003). Nitric acid photolysis on surfaces in low-NOx environments: significant atmospheric implications. Geophysical Research Letters, 30(23): 2217
Zhu C, Xiang B, Chu L T, Zhu L (2010). 308 nm photolysis of nitric acid in the gas phase, on aluminum surfaces, and on ice films. Journal of Physical Chemistry A, 114(7): 2561–2568
Zhu L, Sangwan M, Huang L, Du J, Chu L T (2015). Photolysis of nitric acid at 308 nm in the absence and in the presence of water vapor. Journal of Physical Chemistry A, 119(20): 4907–4914
Zhuang H, Chan C K, Fang M, Wexler A S (1999). Size distributions of particulate sulfate, nitrate, and ammonium at a coastal site in Hong Kong (China). Atmospheric Environment, 33(6): 843–853
Zou J, Lu J, Sun Y, Zhu C (2015). UV photolysis of HNO3 in the gas phase and on the SiO2 film. Environmental Chemistry, 34(4): 748–753 (in Chinese)
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Nos. 22188102 and 21922610). The authors appreciate the Ozone Formation Mechanism and Control Strategies Project of the Research Center for Eco-Environmental Sciences, CAS (China) (No. RCEES-CYZX-2020), the Cultivating Project of Strategic Priority Research Program of Chinese Academy of Sciences (China) (No. XDPB1901), and the Young Talent Project of the Center for Excellence in Regional Atmospheric Environment, CAS (China) (No. CERAE201801).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Highlights
• Recent advances in the photolysis of nitrate/HNO3 are reviewed.
• Mechanisms and key factors affecting the photolysis of nitrate/HNO3 are summarized.
• Atmospheric implications and future research recommendations are provided.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cao, Y., Ma, Q., Chu, B. et al. Homogeneous and heterogeneous photolysis of nitrate in the atmosphere: state of the science, current research needs, and future prospects. Front. Environ. Sci. Eng. 17, 48 (2023). https://doi.org/10.1007/s11783-023-1648-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11783-023-1648-6