Abstract
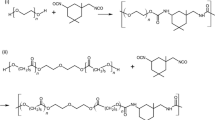
Well-defined graft copolymers based on poly(ε-caprolactone) (PCL) via poly(linoleic acid) (PLina), are derived from soybean oil. Poly(linoleic acid)-g-poly(ε-caprolactone) (PLina-g-PCL) and poly(linoleic acid)-g-poly(styrene)-g-poly(ε-caprolactone) (PLina-g-PSt-g-PCL) were synthesized by ring-opening polymerization of ε-caprolactone initiated by PLina and one-pot synthesis of graft copolymers, and by ring-opening polymerization and free radical polymerization by using PLina, respectively. PLina-g-PCL, PLina-g-PSt-g-PCL3, and PLina-g-PSt-g-PCL4 copolymers containing 96.97, 75.04 and 80.34 mol% CL, respectively, have been investigated regarding their enzymatic degradation properties in the presence of Pseudomonas lipase. In terms of weight loss, after 1 month, 51.5 % of PLina-g-PCL, 18.8 % of PLina-g-PSt-g-PCL3, and 38.4 % of PLina-g-PSt-g-PCL4 were degraded, leaving remaining copolymers with molecular weights of 16,140, 83,220 and 70,600 Da, respectively. Introducing the PLina unit into the copolymers greatly decreased the degradation rate. The molar ratio of [CL]/[Lina] dramatically decreased, from 21.3 to 8.4, after 30 days of incubation. Moreover, reduced PCL content in PLina-g-PSt-g-PCL copolymers decreased the degradation rate, probably due to the PSt enrichment within the structure, which blocks lipase contact with PCL units. Thus, copolymerization of PCL with PLina and PSt units leads to a controllable degradation profile, which encourages the use of these polymers as promising biomaterials for tissue engineering applications.




Similar content being viewed by others
References
Seyednejad H, Gawlitta D, Kuiper RV, Bruin A, van Nostrum CF, Vermonden T, Dhert WJA, Hennink WE (2012) In vivo biocompatibility and biodegradation of 3D-printed porous scaffolds based on a hydroxyl-functionalized poly(ε-caprolactone). Biomaterials 33:4309–4318
Causa F, Netti PA, Ambrosio L (2007) A multi-functional scaffold for tissue regeneration: the need to engineer a tissue analogue. Biomaterials 28:5093–5099
Ng KW, Hutmacher DW, Schantz JT, Ng CS, Too HP, Lim TC, Phan TT, Teoh SH (2001) Evaluation of ultra-thin poly(3-caprolactone) films for tissue-engineered skin. Tissue Eng 7:441–455
Ciapetti G, Ambrosi L, Savarino L, Granchi D, Cenni E, Baldini N, Pagani S, Guizzardi S, Causa F, Giunti A (2003) Osteoblast growth and function in porous poly 3-caprolactone matrices for bone repair: a preliminary study. Biomaterials 24:3815–3824
Gerçek I, Tığlı RS, Gümüşderelioğlu M (2008) A novel scaffold based on formation and agglomeration of PCL microbeads by freeze-drying. J Biomed Mater Res A 86A(4):1012–1022
Tığlı RS, Kazaroğlu NM, Maviş B, Gümüşderelioğlu M (2011) Cellular behaviour on epidermal growth factor (EGF) immobilized PCL/gelatin nanofibrous scaffolds. J Biomater Sci Polym Ed 22(1–3):207–223
Gümüşderelioğlu M, Dalkıranoğlu S, Aydın Tığlı RS, Çakmak S (2011) A novel dermal substitute based on biofunctionalized electrospun PCL nanofibrous matrix. J Biomed Mater Res A 98A(3):461–472
Peng H, Ling J, Liu J, Zhu N, Ni X, Shen Z (2010) Controlled enzymatic degradation of poly(3-caprolactone)-based copolymers in the presence of porcine pancreatic lipase. Polym Degrad Stabil 95:643–650
Zhu GX, Ling J, Shen ZQ (2003) Isothermal crystallization of random copolymers of 3-caprolactone with 2,2-dimethyltrimethylene carbonate. Polymer 44:5827–5832
Andjelkovic DD, Valverde M, Henna P, Li FK, Larock RC (2005) Novel thermosets prepared by cationic copolymerization of various vegetable oils: synthesis and their structure property relationships. Polymer 46:9674–9685
Cakmakli B, Hazer B, Tekin IO, Kizgut S, Koksal M, Menceloglu Y (2004) Synthesis and characterization of polymeric linseed oil grafted methyl methacrylate or styrene. Macromol Biosci 4:649–655
Soucek MD, Khattab T, Wu J (2012) Review of autoxidation and driers. Prog Org Coat 73:435–454
Xia Y, Larock RC (2010) Vegetable oil-based polymeric materials: synthesis, properties, and applications. Green Chem 12:1893–1909
Allı A, Allı S, Becer R, Hazer B (2014) One-pot synthesis of poly(linoleic acid)-g-poly(styrene)-g-poly(ε-caprolactone) graft copolymers. J Am Oil Chem Soc 91:849–858
Cakmakli B, Hazer B, Tekin IÖ, Cömert FB (2005) Synthesis and characterization of polymeric soybean oil-g-methyl methacrylate (and n-butyl methacrylate) graft copolymers: biocompatibility and bacterial adhesion. Biomacromolecules 6:1750–1758
Allı A, Hazer B (2008) Poly(N-isopropyl acrylamide) thermoresponsive cross-linked conjugates containing polymeric soybean oil and/or polypropylene glycol. Eur Poly J 44:1701–1713
Miao S, Sun L, Wang P, Liu R, Su Z, Zhang S (2012) Soybean oil-based polyurethane networks as candidate biomaterials: synthesis and biocompatibility. Eur J Lipid Sci Technol 114:1165–1174
Aydın Tığlı RS, Hazer B, Acar M, Gümüşderelioğlu M (2013) Osteogenic activities of polymeric soybean oil-g-polystyrene membranes. Polym Bull 70(7):2065–2082
Gan Z, Liang Q, Zhang J, Jing X (1997) Enzymatic degradation of poly(3-caprolactone) film in phosphate buffer solution containing lipases. Polym Degrad Stabil 56:209–213
Li S, Liu L, Garreau H, Vert M (2003) Lipase-catalyzed biodegradation of poly(3-caprolactone) blended with various polylactide-based polymers. Biomacromolecules 4:372–377
He F, Li SM, Vert M, Zhuo RX (2003) Enzyme-catalyzed polymerization and degradation of copolymers prepared from ε-caprolactone and poly(ethylene glycol). Polymer 44:5145–5153
Allı A, Hazer B (2011) Synthesis and characterization of poly(N-isopropyl acryl amide)-g-poly(linoleic acid)/poly(linolenic acid) graft copolymers. J Am Oil Chem Soc 88:255–263
Ma Y, Zheng Y, Zeng X, Jiang L, Chen H, Liu R, Huang L, Mei L (2011) Novel docetaxel-loaded nanoparticles based on PCL-Tween 80 copolymer for cancer treatment. Int J Nanomed 6:2679–2688
Li S, Vert M (1999) Biodegradable polymers: polyesters. In: Mathiowitz E (ed) the encyclopedia of controlled drug delivery. Wiley, New York, pp 71–93
Takemura R, Werb Z (1984) Secretory products of macrophages and their physiological functions. Am J Physiol Cell Physiol 15:C1–C9
Kulkarni A, Reiche J, Hartmann J, Kratz K, Lendlein A (2008) Selective enzymatic degradation of poly(ε-caprolactone) containing multiblock copolymers. Eur J Pharm Biopharm 68:46–56
Li S, Garreau H, Pauvert B, McGrath J, Toniolo A, Vert M (2002) Enzymatic degradation of block copolymers prepared from 3-caprolactone and poly(ethylene glycol). Biomacromolecules 3:525–530
Cho K, Lee J, Xing P (2002) Enzymatic degradation of blends of poly(ε-caprolactone) and poly(styrene-co-acrylonitrile) by Pseudomonas lipase. J Appl Polym Sci 83:868–879
Gan Z, Yu D, Zhong Z, Liang Q, Jing X (1999) Enzymatic degradation of poly(3-caprolactone)/poly(dl-lactide) blends in phosphate buffer solution. Polymer 40:2859–2862
Azevedo HS, Reis RL (2005) Understanding the enzymatic degradation of biodegradable polymers and strategies to control their degradation rate. In: Reis RL, San Román J (eds) Biodegradable systems in tissue engineering and regenerative medicine. CRC Press, Florida, pp 177–197
He F, Li S, Garreau H, Vert M, Zhuo R (2005)Enzyme-catalyzed polymerization and degradation of copolyesters of ε-caprolactone and γ-butyrolactone. Polymer 46:12682–12688
Darwis D, Mitomo H, Enjoji T, Yoshi F, Makuuchi K (1998) Enzymatic degradation of radiation crosslinked poly(ε-caprolactone). Polym Degrad Stab 62:259–265
Acknowledgments
This work was supported financially by the Turkish Scientific Research Council (Grants Numbers: 110T884, 211T016) and Bülent Ecevit University Research Fund (Grant Number: 2012-17-21-03).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
11746_2015_2611_MOESM1_ESM.tif
Supplementary material 1: 1H NMR spectra of (a) PLina-g-PCL copolymer degraded after (b) 1 day, (c) 3 days, (d) 5 days, (e) 18 days and (f) 30 days (TIFF 2382 kb)
11746_2015_2611_MOESM2_ESM.tif
Supplementary material 2: 1H NMR spectra of (a) PLina-g-PSt-g-PCL3 copolymer degraded after (b) 1 day, (c) 3 days, (d) 5 days, (e) 18 days and (f) 30 days (TIFF 2265 kb)
11746_2015_2611_MOESM3_ESM.tif
Supplementary material 3: 1H NMR spectra of (a) PLina-g-PSt-g-PCL4 copolymer degraded after (b) 1 day, (c) 3 days, (d) 5 days, (e) 18 days and (f) 30 days (TIFF 2634 kb)
About this article
Cite this article
Allı, S., Tığlı Aydın, R.S., Allı, A. et al. Biodegradable Poly(ε-Caprolactone)-Based Graft Copolymers Via Poly(Linoleic Acid): In Vitro Enzymatic Evaluation. J Am Oil Chem Soc 92, 449–458 (2015). https://doi.org/10.1007/s11746-015-2611-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-015-2611-x