Abstract
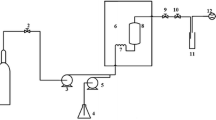
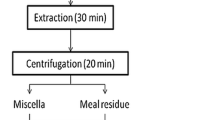
The extraction efficiency of microalgae lipids with aqueous isopropanol (IPA) was investigated and compared with the extraction of oil from full-fat soy flour. The effects of the type of microalgae (Scenedesmus sp. and Schizochytrium limacinum), cell rupture, and IPA concentration on the yield of oil and non-lipid biomass were determined. The oil yield from intact cells of Scenedesmus was 86–93 % with 70, 88, or 95 % (by wt) IPA. Ultrasonic cell rupture prior to oil extraction decreased the oil yield of Scenedesmus to 74 % when extracting with 70 % IPA. The oil yield from intact cells of S. limacinum was <23 % regardless of the IPA concentration, but ruptured cells gave a 94–96 % oil yield with 88 or 95 % IPA. The different response of the two microalgae to extraction with IPA is possibly caused by differences in the cell wall structure and type and amount of polar lipids. The oil yield from soy flour with 88 and 95 % IPA was 93–95 %, which was significantly greater than yields with 50 and 70 % IPA. Cell rupture had no effect on soy flour extraction. In general, the oil yield from the ruptured cells of both microalgae and soy flour increased with increasing IPA concentration.




Similar content being viewed by others
References
Baker EC, Sullivan DA (1983) Development of a pilot-plant process for the extraction of soy flakes with aqueous isopropyl alcohol. J Am Oil Chem Soc 60:1271–1277
Johnson LA, Lusas EW (1983) Comparison of alternative solvents for oils extraction. J Am Oil Chem Soc 60:229–242
Subbian E, Meinhold P, Buelter T, Hawkins AC (2008) Engineered microorganisms for producing isopropanol. U.S. patent, 20080293125
Yao L, Gerde JA, Wang T (2012) Oil extraction from microalga Nannochloropsis sp. with isopropyl alcohol. J Am Oil Chem Soc 89:2279–2287
Cooney M, Young G, Nagle N (2009) Extraction of bio-oils from microalgae. Sep Purif Rev 38:291–325
Kale A (2011) Methods of and systems for isolating nutraceutical products from algae. U.S. patent 8,084, 038
Wang G, Wang T (2012) Lipid and biomass distribution and recovery from two microalgae by aqueous and alcohol processing. J Am Oil Chem Soc 89:335–345
Harwood JL, Jones LA (1989) Lipids metabolism in algae. Adv Bot Res 16:1–53
Yao L, Gerde JA, Wang T, Wen Z, Lee S-L (2012) Characterization of lipids in five microalgae species. American Oil Chemists’ Society, 103rd American Oil Chemists’ Society Annual Meeting & Expo. Long Beach, CA, May 2012. Abstract
Lee J-Y, Yoo C, Jun S-Y, Ahn C-Y, Oh H-M (2010) Comparison of several methods for effective lipid extraction from microalgae. Biores Technol 101:S75–S77
Zheng H, Yin J, Gao Z, Huang H, Ji X, Dou C (2011) Disruption of Chlorella vulgaris cells for the release of biodiesel-producing lipids: a comparison of grinding, ultrasonication, Bead Milling, Enzymatic Lysis, and Microwaves. Appl Biochem Biotechnol 164:1215–1524
Ethier S, Woisard K, Vaughan D, Wen Z (2011) Continuous culture of the microalgae Schizochytrium limacinum on biodiesel-derived crude glycerol for producing docosahexaenoic acid. Biores Technol 102:88–93
Majoni S, Wang T, Johnson L (2011) Enzyme treatments to enhance oil recovery from condensed corn distillers solubles. J Am Oil Chem Soc 88:523–532
Ryckebosch E, Muylaert K, Foubert I (2012) Optimization of an analytical procedure for extraction of lipids from microalgae. J Am Oil Chem Soc 89:189–198
Wang T, Hammond EG, Fehr WR (1997) Phospholipid fatty acid composition and stereospecific distribution of soybeans with a wide range of fatty acid composition. J Am Oil Chem Soc 74:1587–1594
Wang G, Wang T (2012) Characterization of lipid components in two microalgae for biofuel application. J Am Oil Chem Soc 89:135–143
Allard B, Templier J (2000) Comparison of neutral lipid profile of various trilaminar outer cell wall (TLS)-containing microalgae with emphasis on algaenan occurrence. Photochemistry 54:369–380
Grossi V, Blokker P, Sinninghe Damsté JS (2001) Anaerobic biodegradation of lipids of the marine microalga Nannochloropsis salina. Org Gerochem 32:795–808
Staehelin AL, Pickett-Heaps JD (1975) The ultrastructure of Scenedesmus (Chlorophyceae). I. Species with the “reticulated” or “warty” type of ornamental layer. J Phycol 11:163–185
Atkinson AW, Gunning BES, John PCL (1972) Sporopollenin in the cell wall of Chlorella and other algae: ultrastructure, chemistry, and incorporation of 14C-acetate, studied in synchronous cultures. Planta 107:1–32
Honda D, Yokochi T, Nakahara T, Erata M, Higashihara T (1998) Schizochytrium limacinum sp. nov., a new thraustochytrid from a mangrove area in the west Pacific Ocean. Mycol Res 102:439–448
Wong MKM, Tsui CKM, Au DWT, Vrijmoed LLP (2008) Docosahexaenoic acid production and ultrastructure of the thraustochytrid Aurantiochytrium mangrovei MP2 under high glucose concentrations. Mycoscience 49:226–270
Röder T, Sixta H (2004) Thermal treatment of cellulose pulps and its influence to cellulose reactivity. Lenzinger Berichte 83:79–83
Acknowledgments
We thank Professor Zhiyou Wen for providing the microalga S. limacinum, Dr. Hui Wang for assisting in concentrating the algae cells in the CCUR pilot plant, and Phillips 66 for funding the research.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yao, L., Lee, SL., Wang, T. et al. Comparison of Lipid Extraction from Microalgae and Soybeans with Aqueous Isopropanol. J Am Oil Chem Soc 90, 571–578 (2013). https://doi.org/10.1007/s11746-012-2197-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-012-2197-5