Abstract
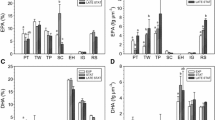
Aquaculture is a growing commercial activity worldwide, which resorts more and more often to microalgae as feed; the lipid composition of such microalgae is a critical factor with regard to the fish growth rate upon ingestion. The aim of this work was thus to study the influence of light intensity on the lipid profile of a known microalga, Pavlova lutheri. Several semi-continuous cultures were carried out, and biochemical parameters such as lipid, protein, carbohydrate, and chlorophyll contents were quantified. Lipids were specifically fractionated into classes by TLC, and those in each class were subjected to GC afterwards in an attempt to ascertain their fatty acid profile. Evidence was consequently provided which showed that cultures grown under low light intensity (9 W m−2) possess a higher fraction of eicosapentaenoic (EPA) and docosahexaenoic (DHA) acids esterified in polar classes—which are those with a more favorable role in aquaculture. It was also demonstrated that intermediate levels of light intensity (19 W m−2) may be misleading in terms of favorable effects upon EPA and DHA contents—because there is an increase in their total yields and productivities, but they appear mostly esterified into triacylglycerols; this may be a favorable deed for production and purification, but is metabolically not so effective in aquaculture. The highest EPA and DHA productivities attained were 1.29 and 0.69 mg L−1 day−1, respectively, at intermediate levels of light intensity (19 W m−2).



Similar content being viewed by others
References
Borowitzka MA (1997) Microalgae for aquaculture: opportunities and constraints. J Appl Phycol 9:393–401
Volkman JK, Jeffrey SW, Nichols PD, Rogers GI, Garland CD (1989) Fatty-acid and lipid-composition of 10 species of microalgae used in mariculture. J Exp Mar Biol Ecol 128:219–240
Dyerberg J (1986) Linolenate-derived polyunsaturated fatty-acids and prevention of atherosclerosis. Nut Rev 44:125–134
Radwan SS (1991) Sources of C-20-polyunsaturated fatty-acids for biotechnological use. Appl Microbiol Biotechnol 35:421–430
Reis A, Gouveia L, Veloso V, Fernandes HL, Empis JA, Novais JM (1996) Eicosapentaenoic acid-rich biomass production by the microalga Phaeodactylum tricornutum in a continuous-flow reactor. Biores Technol 55:83–88
Grima EM, Perez JAS, Camacho FG, Sanchez JLG, Alonso DL (1993) n-3 PUFA productivity in chemostat cultures of microalgae. Appl Microbiol Biotechnol 38:599–605
Yongmanitchai W, Ward O (1991) Screening of algae for potential alternative sources of eicosapentaenoic acid. Phytochemistry 30:2963–2967
Alonso DL, Belarbi EL, Rodriguez-Ruiz J, Segura CI, Gimenez A (1998) Acyl lipids of three microalgae. Phytochemistry 47:1473–1481
Spektorova LV, Goronkova OI, Nosova LP, Albitskaya ON, Danilova GY (1986) High-density culture of marine microalgae promising items for mariculture. 3. Mass-culture of Monochrysis-lutheri Droop. Aquaculture 55:231–240
Thompson PA, Guo M, Harrison PJ (1996) Nutritional value diets that vary in fatty acid composition for larval Pacific oyster (Crassostres gigas). Aquaculture 143:379–391
Volkman JK, Dunstan GA, Jeffrey SW, Kearney PS (1991) Fatty-acids from microalgae of the genus Pavlova. Phytochemistry 30:1855–1859
Borowitzka MA (1988) Algal growth media and sources of algal cultures. In: Borowitzka MA, Borowitzka LJ (eds) Micro-algal Biotechnology. Cambridge University Press, Cambridge, pp 456–465
Ballantine JA, Lavis A, Morris RJ (1979) Sterols of the phytoplankton effects of illumination and growth stage. Phytochemistry 18:1459–1466
Guihéneuf F, Mimouni V, Ulmann L, Tremblin G (2009) Combined effects of irradiance level and carbon source on fatty acid and lipid class composition in the microalga Pavlova lutheri commonly used in mariculture. J Exp Mar Biol Ecol 369:136–143
Liang Y, Beardall J, Heraud P (2006) Changes in growth, chlorophyll fluorescence and fatty acid composition with culture age in batch cultures of Phaeodactylum tricornutum and Chaetoceros muelleri (Bacillariophyceae). Bot Mar 49:165–173
Rao AR, Dayananda C, Sarada R, Shamala TR, Ravishankar GA (2007) Effect of salinity on growth of green alga Botryococcus braunii and its constituents. Bioresour Technol 98:560–564
Carvalho AP, Pontes I, Gaspar H, Malcata FX (2006) Metabolic relationships between macro- and micronutrients, and the eicosapentaenoic acid and docosahexaenoic acid contents of Pavlova lutheri. Enzyme Microb Technol 38:358–366
Petkov G, Garcia G (2007) Which are fatty acids of the green alga Chlorella? Biochem Syst Ecol 35:281–285
Nagashima H, Matsumoto GI, Ohtani S, Momose H (1995) Temperature acclimation and the fatty acid composition of an antarctic green alga Chlorella. Proc NIPR Symp Polar Biol 8:194–199
Rousch JM, Bingham SE, Sommerfeld MR (2003) Changes in fatty acid profiles of thermo-intolerant and thermo-tolerant marine diatoms during temperature stress. J Exp Mar Biol Ecol 295:145–156
Tatsuzawa H, Takizawa E (1995) Changes in lipid and fatty acid composition of Pavlova lutheri. Phytochem 40:397–400
Jiang H, Gao K (2004) Effects of lowering temperature during culture on the production of polyunsaturated fatty acids in the marine diatom Phaeodactylum tricornutum (Bacillariophyceae). J Phycol 40:651–654
Blanchemain A, Grizeau D (1996) Eicosapentaenoic acid content of Skeletonema costatum as a function of growth and irradiance; relation with chlorophyll a content and photosynthetic capacity. J Exp Mar Biol Ecol 196:177–188
Guihéneuf F, Mimouni V, Ulmann L, Tremblin G (2008) Environmental factors affecting growth and omega 3 fatty acid composition in Skeletonema costatum. The influences of irradiance and carbon source. Diatom Res 28:93–103
Thompson PA, Harrison PJ, Whyte JNC (1990) Influence of irradiance on the fatty acid composition of phytoplankton. J Phycol 26:278–288
Brown MR, Dunstan GA, Jeffrey SW, Volkman JK, Barrett SM, LeRoi JM (1993) The influence of irradiance on the biochemical composition of the prymnesiophyte Isochrysis sp. (Clone T-ISO). J Phycol 29:601–612
Khotimchenko SV, Yakovleva IM (2005) Lipid composition of the red alga Tichocarpus crinitus exposed to different levels of photon irradiance. Phytochemistry 66:73–79
Meireles LA, Guedes AC, Barbosa CR, Azevedo JL, Cunha JP, Malcata FX (2008) On-line control of light intensity in a microalgal bioreactor using a novel automatic system. Enzyme Microb Technol 42:554–559
Meireles LA, Azevedo JL, Cunha JP, Malcata FX (2002) On-line determination of biomass in a microalga bioreactor using a novel computerized flow injection analysis system. Biotechnol Prog 18:1387–1391
Hara A, Radin NS (1978) Lipid extraction of tissues with low-toxicity solvent. Anal Biochem 90:420–426
Meireles LA, Guedes AC, Malcata FX (2003) Lipid class composition of the microalga Pavlova lutheri: eicosapentaenoic and docosahexaenoic acids. J Agric Food Chem 51:2237–2241
Lowry O, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Jeffrey SW, Humprey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem Physiol Pflanzen 167:191–194
Madariaga I, Joint I (1992) A comparative study of phytoplankton physiological indicators. J Exp Mar Biol Ecol 158:149–165
Grima EM, Camacho FG, Perez JAS, Sanchez JLG (1994) Biochemical productivity and fatty acid profiles of Isochrysis galbana Parke and Tetraselmis sp. as a function of incident light intensity. Proc Biochem 29:119–126
Eichenberger W, Gribi C (1997) Lipids of Pavlova lutheri: cellular site and metabolic role of DGCC. Phytochemistry 45:1561–1567
Kato M, Sakai M, Adachi K, Ikemoto H, Sano H (1996) Distribution of betaine lipids in marine algae. Phytochemistry 42:1341–1345
Alonso DL, Belarbi EH, Fernandez-Sevilla JM, Rodriguez-Ruiz J, Grima EM (2000) Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry 54:461–471
Dubinsky Z, Matsukawa R, Karube I (1985) Photobiological aspects of algal mass culture. J Mar Biotechol 2:61–65
Kato M, Hajiro NK, Sano H, Miyachi S (1995) Polyunsaturated fatty acids and betaine lipids from Pavlova lutheri. Plant Cell Physiol 36:1607–1611
Chen GQ, Jiang Y, Chen F (2007) Fatty acid and lipid class composition of the eicosapentaenoic acid-producing microalga, Nitzschia laevis. Food Chem 104:1580–1585
Mouget JL, Tremblin G, Morant-Manceau A, Morançais M, Robert JM (1999) Long-term photoacclimation of Haslea ostrearia (Bacillariophyta): effect of irradiance on growth rates, pigment content and photosynthesis. Eur J Phycol 34:109–115
Acknowledgments
Laboratory support from Dr. Ana P. Carvalho is hereby gratefully acknowledged. This work was partially funded by project MICROPESCA (ref. MARE/0072/04), coordinated by author F. X. Malcata, and BIOALGA (ref. POCTI/CTA/44600/2002), both under the auspices of ESF (III Quadro Comunitário de Apoio), and funded by the EC and the Portuguese Government; as well as by a Ph.D. fellowship granted to author A. C. Guedes (ref. SFRH/BD/8115/2002), which was also supervised by author F.X. Malcata and funded by the EC and the Portuguese Government.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Guedes, A.C., Meireles, L.A., Amaro, H.M. et al. Changes in Lipid Class and Fatty Acid Composition of Cultures of Pavlova lutheri, in Response to Light Intensity. J Am Oil Chem Soc 87, 791–801 (2010). https://doi.org/10.1007/s11746-010-1559-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-010-1559-0