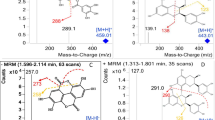
Abstract
A high-performance liquid chromatographic (HPLC) method has been developed to separate sinapine and sinapic acid from other phenolics in canola seed and meal in a single run. The separation was achieved with a reverse-phase C18 column. Owing to the higher recovery of phenolics and ease of use, refluxing with 100% methanol for 20 min was selected as the extraction method for HPLC analysis and determination of total phenolics using Folin-Ciocalteu reagent. A 10-min isocratic/linear/concave gradient and a 15-min isocratic/linear gradient were selected as the best gradients for the separation of these phenolic compounds. Peak identities for sinapine and sinapic acid were verified with ion exchange separation followed by HPLC analysis. The method was calibrated using sinapine bisulfate and sinapic acid standards; correlation coefficients (R 2) for the calibration curves were 0.997 and 0.999 for sinapine bisulfate and sinapic acid, respectively. The extinction coefficient of sinapine was determined to be 1.16 times that of sinapic acid at the detector wavelength (330 nm). Applying this method to routine canola phenolic analyses can greatly reduce the cost by simplifying the procedures and reducing the time required for each determination.
Similar content being viewed by others
References
Austin, F.L., and I.A. Wolff, Sinapine and Related Esters in Seed Meal of Crambe abyssinica J. Agric. Food Chem. 16:132–135 (1968).
Krygier, K., F. Sosulski, and L. Hogge, Free, Esterified and Insoluble Phenolic Acids. II. Composition of Phenolic Acids in Rapeseed Flour and Hulls, —Ibid. 30:334–336 (1982).
Bouchereau, A., J. Hamelin, M. Renard, and F. Larher, Structural Changes in Sinapic Acid Conjugates During Seedling Development of Rape, Plant Physiol. Biochem. 30:467–475 (1992).
Shahidi, F., and M. Naczk, Food Phenolics: Sources, Chemistry, Effects and Applications, Technomic Publishing, Lancaster, PA, 1995.
Hobson, F.A., G.R. Fenwick, R.K. Heaney, D.G. Land and R.F. Curis, Rapeseed Meal and Egg Tainting: Association with Sinapine, Br. Poult. Sci. 18:539–541 (1977).
Goh, Y.K., D.R. Clandinin, A.R. Robblee, and K. Darlington, The Effect of Level of Sinapine in a Laying Ration on the Incidence of Fishy Odor in Eggs from Brown-Shelled Egg Layers, Can. J. Anim. Sci. 59:313–316 (1979).
Shahidi, F., and M. Naczk, An Overview of Phenolics of Canola and Rapeseed: Chemical, Sensory and Nutritional Significance, J. Am. Oil Chem. Soc. 69:917–924 (1992).
Keshavarz, E., R.K.M. Cheung, R.C.M. Liu, and S. Nakai, Adaptation of the Three Stage Extraction Process to Rapeseed Meal for Preparation of Colorless Protein Extracts, Can. Inst. Food Sci. Technol. J. 10:73–77 (1977).
Sosulski, F.W., Organoleptic and Nutritional of Phenolic Compounds on Oilseed Protein Products: A Review, J. Am. Oil Chem. Soc. 56:711–715 (1979).
Youngs, C.G., Technical Status Assessment of Food Protein from Canola, in Research on Canola Seed, Oil and Meal, 9th Project Report, Canola Council of Canada, Winnipeg, 1991, pp. 309–341.
Blair, R., and R.D. Reichert, Carbohydrate and Phenolic Constituents in a Comprehensive Range of Rapeseed and Canola Fractions: Nutritional Significance for Animals, J. Sci. Food Agric. 35:29–35 (1984).
Sosulski, F.W., and K.J. Dabrowski, Composition of Free and Hydrolyzable Phenolic Acids in the Flours and Hulls of Ten Legume Species, J. Agric. Food Chem. 32:131–133 (1984).
Ribereau-Gayon, P., Plant Phenolics, Oliver & Boyd, Edinburgh, United Kingdom 1972.
Swain, T., and W.E. Hillis, The Phenolic Constituents of Prunus domestica I. The Quantitative Analysis of Phenolic Constituents, J. Sci. Food Agric. 10:63–68 (1959).
Wang, S.X., B.D. Oomah, and D.I. McGregor, Application and Evaluation of Ion-Exchange UV Spectrophotometric Method for Determination of Sinapine in Brassica Seeds and Metals, J. Agric. Food Chem. 46:575–579 (1998).
Ismail, F., and N.A.M. Eskin, A New Quantitative Procedure for Determination of Sinapine, —Ibid. 46:575–579 (1998).
Hartley, R.D., and E.C. Jones, Effect of Ultraviolet Light on Substituted Cinnamic Acids and the Estimation of Their Cis and Trans Isomers by Gas Chromatography, J. Chromatogr. 107:213–218 (1975).
Andersen, J.M., Analysis of Plant Phenolics by High Performance Liquid Chromatography, —Ibid. 259:131–139 (1983).
Clausen, S., O. Olsen, and H. Sorensen, Separation of Aromatic Choline Esters by High-Performance Liquid Chromatography, —Ibid. 260:193–199 (1983).
Hagerman, A.E., and R.L. Nicholson, High-Performance Liquid Chromatographic Determination of Hydroxycinnamic Acids in the Maize Mesocotyl, J. Agric. Food Chem. 30:1098–1102 (1982).
Lattanzio, V., High-Performance Liquid Chromatography of Free and Bound Phenolic Acid in the Eggplant, J. Chromatogr. 250:143–148 (1982).
Bjerg, B., O. Olsen, K.W. Rasmussen, and H. Sorensen, New Principles of Ion-Exchange Techniques Suitable to Sample Preparation and Group Separation of Natural Products Prior to Liquid Chromatography, J. Liquid Chromatogr. 7:691–707 (1984).
Bouchereau, A., J. Hamelin, I. Lamour, M. Renard, and F. Larher, Distribution of Sinapine and Related Compounds in Seeds of Brassica and Allied Genera, Phytochemistry 30:1873–1881 (1991).
Mailer, R.J., J. Daun, and R. Scarth, Cultivar Identification in Brassica napus L. by Reversed-Phase High-Performance Liquid Chromatography of Ethanol Extracts, J. Am. Oil Chem. Soc. 70:863–866 (1993).
Clandinin, D.R., Rapeseed Oil Meal Studied. 4. Effect of Sinapine, the Bitter Substance in Rapeseed Oil Meal, on the Growth of Chickens, Poult. Sci. J. 40:484–487 (1961).
Murray, E.D., C.D. Myers, and L.D. Barker, Protein Product and Process for Preparing Same, Canadian Patent 1,028,522 (1978).
Schanderl, S.H., Tannins and Related Phenolics, in Methods in Food Analysis, Physical Chemical and Instrumental Methods of Analysis, edited by M.A. Joslyn, Academic Press, New York, 1970 pp. 701–725.
SAS, SAS User's Guide, Version 6, SAS Institute Inc., Cary, NC, 1996.
Naczk, M., F. Shahidi, and A. Sullivan, Recovery of Rapeseed Tannins by Various Solvent Systems, Food Chem. 45:51–54 (1992).
Macrae, R., HPLC in Food Analysis, Academic Press, London, 1982, p. 4.
Larsen, L.M., O. Olsen, A. Ploger, and H. Sorensen, Sinapine-O-β-d-glucopyranoside in Seeds of Alliaria officinalis, Phytochemistry 22:219–222 (1983).
Wanasundara, J., R. Amarowicz, and F. Shahidi, Isolation and Identification of an Antioxidative Component in Canola Meal, J. Agric. Food Chem. 42:1285–1290 (1994).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Cai, R., Arntfield, S.D. A rapid high-performance liquid chromatographic method for the determination of sinapine and sinapic acid in canola seed and meal. J Amer Oil Chem Soc 78, 903–910 (2001). https://doi.org/10.1007/s11746-001-0362-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-001-0362-4