Abstract
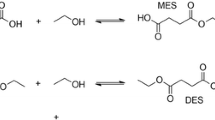
Dihydroxystearic acid (DHSA) ester was synthesized enzymatically to overcome the problems associated with chemical processes. Immobilized enzyme, Lipozyme IM and Novozym 435, were employed as catalysts in the esterification reaction between DHSA and monohydric alcohol. Various factors that may affect the esterification reaction were studied, such as initial water content (a w ), organic solvent, substrate concentration and the influence of alcohol chain length. It was found that the percent conversion was higher in organic solvents with log P (the logarithm of the partition coefficient of solvent in octanol/water system) from 2.0 to 4.0. The reaction was not affected by a w from 0.09 to 0.96. Increasing the mole ratio of alcohol to acid above 2.0 did not increase the percent converions of ester. The ester was identified by Fourier transform infrared and 13C nuclear magnetic resonance spectroscopy.
Similar content being viewed by others
References
Lokotsch, W., S. Lang, D. Mobius, and F. Wagner, Biocatalytical Synthesis and Monolayer Studies of Multiple Hydroxylated Wax Esters, J. Am. Oil. Chem. Soc. 73:1459–1464 (1996).
Hayes, D.G., and R. Kleiman, Lipase-Catalyzed Synthesis of Lesquerolic Acid Wax and Diol Esters and Their Properties, ——Ibid., 73:1385–1392 (1996).
Ghoshray, S., and D.K. Bhattacharya, Enzymatic Preparation of Ricinoleic Acid Esters of Long-Chain Monohydric Alcohols and Properties of the Esters, ——Ibid. 69:85–88 (1992).
Ghosh, M., and D. K. Bhattacharyya, Lipase-Catalyzed Synthesis of Hydroxy Stearates and Their Properties, ——Ibid. 75:1057–1059 (1998).
Mukesh, D., R.S. Iyer, J.S. Wagh, A.A. Mikashi, A.A. Banerji, R.V. Newadkar, and H.S. Bevinakatti, Lipase-Catalysed Transesterification of Castor Oil, Biotechnol. Lett. 15:251–256 (1993).
Wagner, F., F. Kleppe, W. Lokotsch, A. Ziemann, and S. Lang, Synthesis of Uncommon Wax Ester with Immobilized Lipases, Ann. NY Acad. Sci. 672:484–491 (1992)
Hayes, D.G. The Catalytic Activity of Lipases Toward Hydroxy Fatty Acids—A Review, J. Am. Oil Chem. Soc. 73:543–549 (1996).
Awang, R., S. Ahmad and Y.B. Kang, Preparation of Dihydroxy Fatty Acid from Oleic Acid, Malaysian Patent PI 9804456 (1998).
Steffen, B., A. Ziemann, S. Lang, and F. Wagner, Monoacylation of Trihydroxy Compounds, Biotechnol. Lett. 14:773–778 (1992).
Lang, S., R. Multzch, A. Passeri, A. Schmeichel, B. Steffen, F. Wagner, D. Hamman, and H.K. Cammage, Unusual Wax Esters and Glycolipids: Biocatalytic Formation and Physico Chemical Characterization, Acta Biotechnol. 11:379–386 (1991).
Laane, C., and S. Boeren, K. Vos, and C. Veeger, Rules for Optimization of Biocatalysis in Organic Solvents, Biotechnol. Biolengin. 30:81–87 (1986).
Katchalski-Katzir, E., Medium and Biocatalytic Engineering, in Biocatalysis in Non-Conventional Media, edited by J. Tramper, M.H. Vermue, H.H. Beefink, and U. von Stockar, Elsevier Science Publishers B.V., New York, Vol. 8, 1992, pp. 3–9.
Basri, M., W.M.Z. Wan Yunus, S.Y. Wong, K. Ampon, C.N.A. Razak, and A.B. Salleh, Immobilization of Lipase from Candida rugosa on Synthetic Polymer Beads for Use in the Synthesis of Fatty Esters, J. Chem. Tech. Biotechnol. 66:169–173 (1996).
Valivety, R.H., P.J. Halling, A.D. Peilow, and A.R. Macrae, Lipases from Different Sources Vary Widely in Dependence of Catalytic Activity on Water Activity, Biochim. Biophys. Acta, 1122:143–146 (1992).
Halling, P.J., Salts Hydrates for Water Activity Control with Biocatalyst in Organic Media, Biotechnol. Tech. 6:271–276 (1992).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Awang, R., Basri, M., Ahmad, S. et al. Enzymatic esterification of dihydroxystearic acid. J Amer Oil Chem Soc 77, 609–612 (2000). https://doi.org/10.1007/s11746-000-0098-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-000-0098-1