Summary
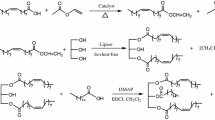
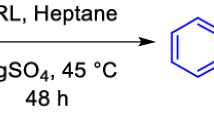
Using phenylboronic acid as solubilizing agent in nonpolar solvents the lipase-catalyzed monoacylation of glycerol with uncommon fatty acids like (S)-17-hydroxy stearic acid was successful. 1-monostearoyl-5,6-dihydroxy hexane was prepared by an alternative classical route.
Similar content being viewed by others
References
Akoh, C. C., Cooper, C., and Nwosu, C. V. (1992).J. Am. Oil Chem. Soc. 69 (3), 257–260.
Berger, M. and Schneider, M. P. (1992).J. Am. Oil Chem. Soc., in press.
Chopineau, J., McCafferty, F. D., Therisod, M., and Klibanov, A. M. (1988)Biotechnol. Bioeng. 31, 208–214.
Lang, S., Multzsch, R., Passeri, A., Schmeichel, A., Steffen, B., Wagner, F., Hamann, D., and Cammenga, H. K. (1991).Acta Biotechnologica 11 (4), 379–386.
McNeill, G. P., Shimizu, S., and Yamane, T. (1990).J. Am. Oil Chem. Soc. 67 (11), 779–783.
McNeill, G. P., Shimizu, S., and Yamane, T. (1991).J. Am. Oil Chem. Soc. 68 (1), 1–5.
Metzger, J. O. and Linker, U. (1991).Fat Sci. Technol. 93 (7), 244–249.
Miller, C., Austin, H., Pesorske, L., and Gonzalez, J. (1988).J. Am. Oil Chem. Soc. 65, 927–931.
Omar, I. C., Saeki, H., Nishio, N., and Nagai, S. (1989).Biotechnol. Letters 11 (3), 161–166.
Park, H.-O., Lee, D.-S., and Shim, S. C. (1992).Biotechnol. Letters 14 (2), 111–116.
Schuch, R. and Mukherjee, K. D. (1989).Appl. Microbiol. Biotechnol. 30, 332–336.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Steffen, B., Ziemann, A., Lang, S. et al. Enzymatic monoacylation of trihydroxy compounds. Biotechnol Lett 14, 773–778 (1992). https://doi.org/10.1007/BF01029137
Issue Date:
DOI: https://doi.org/10.1007/BF01029137