Abstract
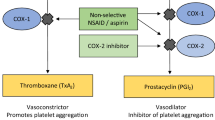
Elucidation of differences between the active sites of COX-1 and COX-2 allowed the targeted design of the selective COX-2 inhibitors known as coxibs. They were marketed as non-steroidal anti-inflammatory drugs (NSAIDs) that had improved upper gastrointestinal (GI) safety compared with older non-selective NSAIDs such as diclofenac and naproxen. Two GI safety studies conducted with arthritis patients demonstrated that in terms of upper GI safety, celecoxib was not superior to diclofenac (CLASS study) but rofecoxib was superior to naproxen (VIGOR study). However, the VIGOR study revealed also that rofecoxib had increased cardiovascular (CV) risk compared with naproxen. This clinical outcome was supported by the existence of plausible eicosanoid-based biological mechanisms whereby selective COX-2 inhibition could increase CV risk. Nevertheless, the existence of CV risk with rofecoxib was successfully discounted by its pharmaceutical company owner, Merck & Co, with the assistance of specialist opinion leaders and rofecoxib achieved widespread clinical use for 4–5 years. Rofecoxib was withdrawn from the market when several clinical trials in colorectal cancer and post-operative pain revealed increased CV risk with not only rofecoxib, but also coxibs. The commercial success of rofecoxib provides a case-study of failure of the medical journal literature to guide drug usage. Attention to ethical issues may have provided a more useful guide for prescribers.



Similar content being viewed by others
References
Moncada S, Ferreira SH, Vane JR (1975) Inhibition of prostaglandin biosynthesis as the mechanism of analgesia of aspirin-like drugs in the dog knee joint. Eur J Pharmacol 31:250–260
Vane JR, Bakhle YS, Botting RM (1998) Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol 38:97–120
Fries JF, Murtagh KN, Bennett M, Zatarain E, Lingala B, Bruce B (2004) The rise and decline of nonsteroidal antiinflammatory drug-associated gastropathy in rheumatoid arthritis. Arthritis Rheum 50:2433–2440
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, Gildehaus D, Miyashiro JM, Penning TD, Seibert K, Isakson PC, Stallings WC (1996) Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 384:644–648
Hawkey CJ (1999) COX-2 inhibitors. Lancet 353:307–314
Warner TD, Giuliano F, Vojnovic I, Bukasa A, Mitchell JA, Vane JR (1999) Nonsteroid drug selectivities for cyclo-oxygenase-1 rather than cyclo-oxygenase-2 are associated with human gastrointestinal toxicity: a full in vitro analysis. Proc Natl Acad Sci USA 96:7563–7568
Demasi M, Cleland LG, Cook-Johnson RJ, Caughey GE, James MJ (2003) Effects of hypoxia on monocyte inflammatory mediator production: dissociation between changes in cyclooxygenase-2 expression and eicosanoid synthesis. J Biol Chem 278:38607–38616
Caughey GE, Cleland LG, Penglis PS, Gamble JR, James MJ (2001) Roles of cyclooxygenase (COX) -1 and -2 in prostanoid production by human endothelial cells: selective upregulation of prostacyclin synthesis by COX-2. J Immunol 167:2831–2838
Cook-Johnson RJ, Demasi M, Cleland LG, Gamble JR, Saint DA, James MJ (2006) Endothelial cell COX-2 expression and activity in hypoxia: transcriptional control mechanisms. BBA-Cell Mol Biol Lipids 1761:1443–1449
Caughey GE, Cleland LG, Gamble JR, James MJ (2001) Up-regulation of endothelial cyclooxygenase-2 and prostanoid synthesis by platelets: role of thromboxane A2. J Biol Chem 276:37839–37845
Penglis PS, Cleland LG, Demasi M, Caughey GE, James MJ (2000) Differential regulation of prostaglandin E2 and thromboxane A2 production in human monocytes: implications for the use of cyclooxygenase inhibitors. J Immunol 165:1605–1611
Catella-Lawson F, McAdam B, Morrison BW, Kapoor S, Kujubu D, Antes L, Lasseter KC, Quan H, Gertz BJ, Fitzgerald GA (1999) Effects of specific inhibition of cyclooxygenase-2 on sodium balance, hemodynamics, and vasoactive eicosanoids. J Pharmacol Exp Ther 289:735–741
McAdam BF, Catella-Lawson F, Mardini IA, Kapoor S, Lawson JA, Fitzgerald GA (1999) Systemic biosynthesis of prostacyclin by cyclooxygenase (COX)-2: the human pharmacology of a selective inhibitor of COX-2. Proc Natl Acad Sci USA 96:272–277
Stemme V, Swedenborg J, Claesson H-E, Hansson GK (2000) Expression of cyclo-oxygenase-2 in human atherosclerotic carotid arteries. Eur J Vasc Endovasc Surg 20:146–152
Belton O, Byrne D, Kearney D, Leahy A, Fitzgerald DJ (2000) Cyclooxygenase-1 and -2-dependent prostacyclin formation in patients with atherosclerosis. Circulation 102:840–845
Baker CS, Hall RJ, Evans TJ, Pomerance A, Maclouf J, Creminon C, Yacoub MH, Polak JM (1999) Cyclooxygenase-2 is widely expressed in atherosclerotic lesions affecting native and transplanted human coronary arteries and colocalizes with inducible nitric oxide synthase and nitrotyrosine particularly in macrophages. Arterioscler Thromb Vasc Biol 19:646–655
Topper JN, Cai J, Falb D, Gimbrone MA (1996) Identification of vascular endothelial genes differentially responsive to fluid mechanical stimuli: cyclooxygenase-2, manganese superoxide dismutase, and endothelial nitric oxide synthase are selectively up-regulated by steady laminar shear stress. Proc Natl Acad Sci USA 93:10417–10422
Dowd NP, Scully M, Adderley SR, Cunningham AJ, Fitzgerald DJ (2001) Inhibition of cyclooxygenase-2 aggravates doxorubicin-mediated cardiac injury in vivo. J Clin Invest 108:585–590
Adderley SR, Fitzgerald DJ (1999) Oxidative damage of cardiomyocytes is limited by extracellular regulated kinases 1/2-mediated induction of cyclooxygenase-2. J Biol Chem 274:5038–5046
Bolli R, Shinmura K, Tang XL, Kodani E, Xuan YT, Guo Y, Dawn B (2002) Discovery of a new function of cyclooxygenase (COX)-2: COX-2 is a cardioprotective protein that alleviates ischemia/reperfusion injury and mediates the late phase of preconditioning. Cardiovasc Res 55:506–519
Patrono C, Garcia Rodriguez LA, Landolfi R, Baigent C (2005) Low-dose aspirin for the prevention of atherothrombosis. N Engl J Med 353:2373–2383
Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A, Makuch R, Eisen G, Agrawal NM, Stenson WF, Burr AM, Zhao WW, Kent JD, Lefkowith JB, Verburg KM, Geis GS (2000) Gastrointestinal toxicity with celecoxib vs. nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: a randomized controlled trial. Celecoxib long-term arthritis safety study. JAMA 284:1247–1255
US Food and Drug Authority (accessed April 2007) FDA Advisory Committee briefing document. NDA 20–998/S-009. Celebrex capsules (Celecoxib). Medical Officer Review. http://www.fda.gov/ohrms/dockets/ac/01/briefing/3677b1_03_med.doc
Bertagnolli MM, Eagle CJ, Zauber AG, Redston M, Solomon SD, Kim K, Tang J, Rosenstein RB, Wittes J, Corle D, Hess TM, Woloj GM, Boisserie F, Anderson WF, Viner JL, Bagheri D, Burn J, Chung DC, Dewar T, Foley TR, Hoffman N, Macrae F, Pruitt RE, Saltzman JR, Salzberg B, Sylwestrowicz T, Gordon GB, Hawk ET (2006) Celecoxib for the prevention of sporadic colorectal adenomas. N Engl J Med 355:873–884
US Food and Drug Authority (accessed April 2007) FDA Advisory Committee briefing document. NDA 21–042, s007. VIOXX gastrointestinal safety. http://www.fda.gov/ohrms/dockets/ac/01/briefing/3677b2_03_med.pdf
Bombardier C, Laine L, Reicin A, Shapiro D, Burgos-Vargas R, Davis B, Day R, Ferraz MB, Hawkey CJ, Hochberg MC, Kvien TK, Schnitzer TJ, Weaver A (2000) Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. N Engl J Med 343:1520–1528
Bresalier RS, Sandler RS, Quan H, Bolognese JA, Oxenius B, Horgan K, Lines C, Riddell R, Morton D, Lanas A, Konstam MA, Baron JA (2005) Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 352:1092–1102
Nussmeier NA, Whelton AA, Brown MT, Langford RM, Hoeft A, Parlow JL, Boyce SW, Verburg KM (2005) Complications of the COX-2 inhibitors parecoxib and valdecoxib after cardiac surgery. N Engl J Med 352:1081–1091
McGettigan P, Henry D (2006) Cardiovascular risk and inhibition of cyclooxygenase: a systematic review of the observational studies of selective and nonselective inhibitors of cyclooxygenase 2. JAMA 296:1633–1644
Couzin J (2004) Drug safety: withdrawal of vioxx casts a shadow over COX-2 inhibitors. Science 306:384–385
Juni P, Rutjes AWS, Dieppe PA (2002) Are selective COX-2 inhibitors superior to traditional non-steroidal anti-inflammatory drugs? Br Med J 324:1287–1288
Ray WA, Stein CM, Hall K, Daugherty JR, Griffin MR (2002) Non-steroidal anti-inflammatory drugs and risk of serious coronary heart disease: an observational cohort study. Lancet 359:118–123
Ray WA, Stein CM, Daugherty JR, Hall K, Arbogast PG, Griffin MR (2002) COX-2 selective non-steroidal anti-inflammatory drugs and risk of serious coronary heart disease. Lancet 360:1071–1073
Merck & Co (accessed April 2007) Annual Report. 2001–04. http://phx.corporate-ir.net/phoenix.zhtml?c=73184&p=irol-reportsannual
Lexchin J (2002) Should doctors be prescribing new drugs? Int J Risk Safety Med 15:213–222
National Institute for Health Care Management (accessed April 2007) Prescription Drugs and Mass Media Advertising, 2000. http://www.nihcm.org/finalweb/DTCbrief2001.pdf
Waxman HA (accessed April 2007) Statement of Rep. Henry A. Waxman for the hearing “The role of FDA and pharmaceutical companies in ensuring the safety of approved drugs, like Vioxx.” http://democrats.reform.house.gov/Documents/20050505113149–41995.pdf
Konstam MA, Weir MR, Reicin A, Shapiro D, Sperling RS, Barr E, Gertz BJ (2001) Cardiovascular thrombotic events in controlled, clinical trials of rofecoxib. Circulation 104:2280–2288
Topol EJ (2004) Failing the public health-rofecoxib, Merck, and the FDA. N Engl J Med 351:1707–1709
Mukherjee D, Nissen SE, Topol EJ (2001) Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA 286:954–959
Cleland LG, James MJ, Stamp LK, Penglis PS (2001) COX-2 inhibition and thrombotic tendency: a need for surveillance. Med J Aust 175:214–217
Graham DJ (accessed April 2007) Testimony of Dr David Graham, US Senate Committee on Finance. http://www.senate.gov/∼finance/hearings/testimony/2004test/111804dgtest.pdf
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
James, M.J., Cook-Johnson, R.J. & Cleland, L.G. Selective COX-2 Inhibitors, Eicosanoid Synthesis and Clinical Outcomes: A Case Study of System Failure. Lipids 42, 779–785 (2007). https://doi.org/10.1007/s11745-007-3069-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-007-3069-x