Abstract
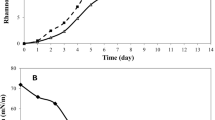
Rhamnolipids are interesting microbial surfactants having great industrial importance. However, the main obstacles towards an economic production of rhamnolipids are low productivity and high raw-material costs. Therefore, this study aimed at optimization of the culture media as well as culture conditions using response surface methodology for maximum rhamnolipid production by Pseudomonas aeruginosa isolate P6, a promising rhamnolipid-producing isolate. The optimum medium for maximum rhamnolipid production was found to be a mineral salts medium with glycerol 2 % v/v as the carbon source. The optimum cultivation conditions using response surface methodology were found to be an incubation temperature of 30 °C, an agitation rate of 250 rpm, an inoculum size of 5 % v/v and unlike most studies, an initial pH of 7.5. The resulting model predicted data points that corresponded well to the experimental values. Optimization resulted in a threefold increase in rhamnolipid production reaching 7.54 g/L. The data are potentially useful for further industrial exploitation of rhamnolipid production by the studied isolate.






Similar content being viewed by others
Abbreviations
- MSM:
-
Mineral salts media
- RSM:
-
Response surface methodology
- RL:
-
Rhamnolipids
References
Luo Z, Yuan XZ, Zhong H, Zeng GM, Liu ZF, Ma XL, Zhu YY (2013) Optimizing rhamnolipid production by Pseudomonas aeruginosa ATCC 9027 grown on waste frying oil using response surface method and batch-fed fermentation. J Cent South Univ 20:1015–1021
Cameotra SS, Makkar RS, Kaur J, Mehta SK (2010) Synthesis of biosurfactants and their advantages to microorganisms and mankind. Adv Exp Med Biol 672:261–280
Rikalovic MG, Vrvic MM, Karadzic IM (2015) Rhamnolipid biosurfactant from Pseudomonas aeruginosa—from discovery to application in contemporary technology. J Serb Chem Soc 80:279–304
Silva SNRL, Farias CBB, Rufino RD, Luna JM, Sarubbo LA (2010) Glycerol as substrate for the production of biosurfactant by Pseudomonas aeruginosa UCP0992. Colloids Surf B 79:174–183
Sinumvayo JP, Ishimwe N (2015) Agriculture and Food Applications of Rhamnolipids and its Production by Pseudomonas aeruginosa. J Chem Eng Process Technol 6:223
Md Noh NA, Mohd Salleh S, Yahya ARM (2015) Enhanced rhamnolipid production by Pseudomonas aeruginosa USM-AR2 via fed-batch cultivation based on maximum substrate uptake rate. Lett Appl Microbiol 58:617–623
Sodagari M, Ju LK (2014) Cells were a more important foaming factor than free rhamnolipids in fermentation of Pseudomonas aeruginosa E03−40 for high rhamnolipid production. J Surfactants Deterg 17:573–582
Mukherjee S, Das P, Sen R (2006) Towards commercial production of microbial surfactants. Trends Biotechnol 24:509–515
Pornsunthorntawee O, Wongpanit P, Rujiravanit R (2010) Rhamnolipid biosurfactants: production and their potential in environmental biotechnology. In: Ramkrishna S (ed) Biosurfactants. Landes Bioscience and Springer Science and Business Media, New York, pp 211–221
AL-Araji LIY, Abd. Rahman RNZR, Basri M, Salleh AB (2007) Optimisation of rhamnolipids produced by Pseudomonas aeruginosa 181 using response surface modeling. Ann Microbiol 57:571–575
Rahman KS, Rahman TJ, McClean S, Marchant R, Banat IM (2002) Rhamnolipid biosurfactant production by strains of Pseudomonas aeruginosa using low-cost raw materials. Biotechnol Prog 18:1277–1281
Sahoo S, Datta S, Biswas D (2011) Optimization of culture conditions for biosurfactant production from Pseudomonas aeruginosa OCD1. J Adv Sci Res 2:32–36
Bodour AA, Drees KP, Maier RM (2003) Distribution of biosurfactant-producing bacteria in undisturbed and contaminated arid Southwestern soils. Appl Environ Microbiol 69:3280–3287
Abdel-Mawgoud AM (2008) Production and characterization of some microbial surfactants. Master degree dissertation. Ain Shams University, Cairo
Alfermann AW, Dombrowski K, Petersen M, Schmauder HP, Schweizer M, Völksch B, Günther T (1997) Basic scientific techniques for biotechnology-analytical methods—growth and cell viability. In: Schmauder HP, Schweizer M (eds) Methods in biotechnology. Taylor & Francis, London, pp 13–14
Kosaric N, Vardar-Sukan F (2014) Biosurfactants: production and utilization-processes, technologies, and economics. CRC Press, Boca Raton
Leitermann F, Syldatk C, Hausmann R (2008) Fast quantitative determination of microbial rhamnolipids from cultivation broths by ATR-FTIR Spectroscopy. J Biol Eng 2:13
Chandrasekaran EV, BeMiller JN (1980) Constituent analysis of glycosaminoglycans. In: Whistler RL, Wolfrom ML (eds) Methods in carbohydrate chemistry. Academic Press, New York, pp 89–96
Koch AK, Käppeli O, Fiechter A, Reiser J (1991) Hydrocarbon assimilation and biosurfactant production in Pseudomonas aeruginosa mutants. J Bacteriol 173:4212–4219
Wu J, Ju LK (1998) Extracellular particles of polymeric material formed in n-hexadecane fermentation by Pseudomonas aeruginosa. J Biotechnol 59:193–202
Wei YH, Chou CL, Chang JS (2005) Rhamnolipid production by indigenous Pseudomonas aeruginosa J4 originating from petrochemical wastewater. Biochem Eng J 27:146–154
Guerra-Santos L, Käppeli O, Fiechter A (1986) Dependence of Pseudomonas aeruginosa continuous culture biosurfactant production on nutritional and environmental factors. Appl Microbiol Biotechnol 24:443–448
Giani C, Wullbrandt D, Rothert R, Meiwes J (1996) Pseudomonas aeruginosa and its use in a process for the biotechnological preparation of L-rhamnose. US patent 5501966 A
Borges WS, Moura AAO, Filho UC, Cardoso VL et al (2015) Optimization of the operating conditions for rhamnolipid production using slaughterhouse-generated industrial float as substrate. Braz J Chem Eng 32:357–365
Santa Anna LM, Sebastian GV, Menezes EP, Alves TLM et al (2002) Production of biosurfactants from Pseudomonas aeruginosa PA1 isolated in oil environments. Braz J Chem Eng 19:159–166
Benincasa M, Contiero J, Manresa MA, Moraes IO (2002) Rhamnolipid production by Pseudomonas aeruginosa LBI growing on soapstock as the sole carbon source. J Food Eng 54:283–288
Da Rosa CFC, Michelon M, Burkert JFDM, Kalil SJ, Burkert CAV (2010) Production of a rhamnolipid-type biosurfactant by Pseudomonas aeruginosa LBM10 grown on glycerol. Afr J Biotechnol 9:9012–9017
Rocha C, San-Bias F, San-Bias G, Vierma L (1992) Biosurfactant production by two isolates of Pseudomonas aeruginosa. World J Microb Biot 8:125–128
Suryanti V, Hastuti S, Wahyuningsih TD, Mudasir M, Muliawati DI (2009) Biosurfactants production by pseudomonas aeruginosa using soybean oil as substrate. Indones J Chem 9:107–112
Pagliaro M, Ciriminna R, Kimura H, Rossi M, Della Pina C (2007) From glycerol to value-added products. Angew Chem Int Ed 46:4434–4440
Da Silva GP, Mack M, Contiero J (2009) Glycerol: a promising and abundant carbon source for industrial microbiology. Biotechnol Adv 27:30–39
Maier RM, Soberón-Chávez G (2000) Pseudomonas aeruginosa rhamnolipids: biosynthesis and potential applications. Appl Microbiol Biotechnol 54:625–633
Henkel M, Müller MM, Kügler JH, Lovaglio RB, Contiero J, Syldatk C, Hausmann R (2012) Rhamnolipids as biosurfactants from renewable resources: concepts for next-generation rhamnolipid production. Process Biochem 47:1207–1219
Wu JY, Yeh KL, Lu WB, Lin CL, Chang JS (2008) Rhamnolipid production with indigenous Pseudomonas aeruginosa EM1 isolated from oil-contaminated site. Bioresour Technol 99:1157–1164
Murthy MSRC, Swaminathan T, Rakshit SK, Kosugi Y (2000) Statistical optimization of lipase catalyzed hydrolysis of methyloleate by response surface methodology. Bioprocess Eng 22:35–39
Nalini S, Parthasarathi R (2014) Production and characterization of rhamnolipids produced by Serratia rubidaea SNAU02 under solid-state fermentation and its application as biocontrol agent. Bioresour Technol 173:231–238
Turbekar R, Malik N, Thakare D (2015) Optimization studies for enhancing rhamnolipid production by Pseudomonas aeruginosa RT using response surface methodology. Int J Res Stud Biosci 3:65–73
Ghribi D, Abdelkefi-Mesrati L, Mnif I, Kammoun R, Ayadi I, Saadaoui I, Maktouf S, Chaabouni-Ellouze S (2012) Investigation of antimicrobial activity and statistical optimization of Bacillus subtilis SPB1 biosurfactant production in solid-state fermentation. J Biomed Biotechnol 2012:373682. doi:10.1155/2012/373682
Abdel-Hafez SM, Hathout RM, Sammour OA (2014) Towards better modeling of chitosan nanoparticles production: screening different factors and comparing two experimental designs. Int J Biol Macromol 64:334–340
Abalos A, Maximo F, Manresa MA, Bastida J (2002) Utilization of response surface methodology to optimize the culture media for the production of rhamnolipids by Pseudomonas aeruginosa AT10. J Chem Technol Biotechnol 77:777–784
Kashyap P, Sabu A, Pandey A, Szakacs G, Soccol CR (2002) Extra-cellular l-glutaminase production by Zygosaccharomyces rouxii under solid-state fermentation. Process Biochem 38:307–312
Zhu L, Yang X, Xue C, Chen Y, Qu L, Lu W (2012) Enhanced rhamnolipids production by Pseudomonas aeruginosa based on a pH stage-controlled fed-batch fermentation process. Bioresour Technol 117:208–213
Chen SY, Wei YH, Chang JS (2007) Repeated pH-stat fed-batch fermentation for rhamnolipid production with indigenous Pseudomonas aeruginosa S2. Appl Microbiol Biotechnol 76:67–74
Chen SY, Lu WB, Wei YH, Chen WM, Chang JS (2007) Improved production of biosurfactant with newly isolated Pseudomonas aeruginosa S2. Biotechnol Prog 23:661–666
Mulligan CN, Sharma SK, Mudhoo A (eds) (2014) Biosurfactants: research trends and applications. CRC Press, Boca Raton
Moussa TAA, Mohamed MS, Samak N (2014) Production and characterization of di-rhamnolipid produced by Pseudomonas aeruginosa TMN. Braz J Chem Eng 31:867–880
Lotfabad TB, Shourian M, Roostaazad R, Najafabadi AR, Adelzadeh MR, Noghabi KA (2009) An efficient biosurfactant-producing bacterium Pseudomonas aeruginosa MR01, isolated from oil excavation areas in south of Iran. Colloids Surf B 69:183–193
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
El-Housseiny, G.S., Aboulwafa, M.M., Aboshanab, K.A. et al. Optimization of Rhamnolipid Production by P. aeruginosa Isolate P6. J Surfact Deterg 19, 943–955 (2016). https://doi.org/10.1007/s11743-016-1845-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-016-1845-4