Abstract
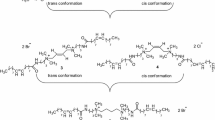
A series of rosin-based cationic gemini surfactants with different spacer length (n = 2, 3, 4) were synthesized and characterized. Surface activity and micellization parameters including the critical micelle concentration, the degree of counterion dissociation, and thermodynamic functions of micellization in aqueous solutions have been investigated. Free energy perturbation was performed to study the enthalpy-entropy compensation of the synthesized gemini surfactants in aqueous solutions. The experimental results showed that the micellization of rosin-based gemini surfactants in aqueous solutions is a spontaneous and entropy-driven process. The micellization process was found to follow the entropy–enthalpy compensation phenomenon.





Similar content being viewed by others
References
Menger FM, Littau C (1991) Gemini-surfactants: synthesis and properties. J Am Chem Soc 113:1451–1452
Zana R (2002) Dimeric and oligomeric surfactants. Behavior at interfaces and in aqueous solution: a review. Adv Colloid Interface 97:205–253
Nuraje N, Bai HY, Su K (2013) Bolaamphiphilic molecules: assembly and applications. Prog Polym Sci 38:302–343
Bhattacharya S, Haldar J (2001) Molecular design of surfactants to tailor its aggregation properties. Colloid Surf A 205:119–126
Negm NA, Kandile NG, Mohamad MA (2011) Synthesis, characterization and surface activity of new eco-friendly Schiff bases vanillin derived cationic surfactants. J Surf Deterg 14:325–331
Sahu NH, Mandaogade PM, Deshmukh AM, Meghre VS, Dorle AK (1999) Biodegradation studies of rosin-glycerol ester derivative. J Bioact Compat Pol 14:344–360
Satturwar PM, Mandaogade PM, Darwhekar GN, Fulzele SV, Joshi SB, Dorle AK (2003) Biodegradation studies of rosin-based polymers. Drug Dev Ind Pharm 29:669–677
Han SY, Song ZQ, Fang GZ, Li SJ (2009) Synthesis and analysis of rosin-based quaternary ammonium gemini surfactant. Chem Ind For Prod 29:110–112
Tan WX, Lin ZT, Bu HT, Tian YN, Jiang GB (2012) Nano-micelles based on a rosin derivative as potent sorbents and sinking agents with high absorption capabilities for the removal of metal ions. RSC Adv 2:7279–7289
Han CR, Zhu LW, Liu LJ, Jiang JX (2009) Controlled synthesis of Ni(OH)2 materials with rosin-based surfactant by microwave solvothermal method. Chem Ind For Prod 29:149–153
Jia WH, Rao XP, Song ZQ, Shang SB (2009) Microwave-assisted synthesis and properties of a novel cationic gemini surfactant with the hydrophenanthrene structure. J Surf Deterg 12:261–267
Chen ZJ, Sj Li, Tian B, Liang T, Yan J (2012) Synthesis of a rosin gemini surfactant and its properties. Environ Eng Sci 29:606–610
Liang T, Zhang YY, Li SJ, Nguyen TTH (2013) Synthesis, characterization, and bioactivity of rosin quaternary ammonium salt derivatives. Bioresources 8:735–742
Atta AM, Ramadan AM, Shaffei KA, Nassar AM, Ahmed NS, Fekry M (2009) Synthesis and properties of nonionic surfactants from rosin-imides maleic anhydride adduct. J Dispers Sci Technol 30:1100–1110
Atta AM, EI-Kafrawy AF, Abdel-Rauf ME, Maysour NE, Gafer AK (2010) Surface and thermodynamic properties of nonionic surfactants based on rosin-maleic anhydride and acrylic acid adducts. J Dispers Sci Technol 31:567–576
Oda R, Candau SJ, Oda R, Huc I (1997) Gemini surfactants, the effect of hydrophobic chain length and dissymmetry. Chem Commun 21:2105–2106
Danino D, Talmon Y, Zana R (1995) Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) surfactants (dimeric surfactants). 5. Aggregation and microstructure in aqueous solutions. Langmuir 11:1448–1456
Tsubone K, Arakawa Y, Rosen MJ (2003) Structural effects on surface and micellar properties of alkanediyl-α,ω-bis(sodium N-acyl-β-alaninate) gemini surfactants. J Colloid Interface Sci 262:516–524
Zana R (2010) Dimeric (gemini) surfactants: effect of the spacer group on the association behavior in aqueous solution. J Colloid Interface Sci 248:203–220
Zheng YJ, Yao KJ, Lee J, Chandler D, Wang JF, Wang CP, Chu FX, Tang CB (2010) Well-defined renewable polymers derived from gum rosin. Macromolecules 43:5922–5924
Wilbon PA, Zheng YJ, Yao KJ, Tang CB (2010) Renewable rosin acid-degradable caprolactone block copolymers by atom transfer radical polymerization and ring-opening polymerization. Macromolecules 43:8747–8754
Ross J, Miles GD (1941) An apparatus for comparison of foaming properties of soaps and detergents. J Am Oil Chem Soc 18:99–102
Rosen MJ, Tracy DJ (1998) Gemini surfactants. J Surf Deterg 1:547–554
Fruhner H, Wantle KD, Lunkenheimer K (2000) Relationship between surface dilational properties and foam stability. Colloids Surf A 162:193–202
Pugh RJ (1996) Foaming, foam films, antifoaming and defoaming. Adv Colloid Interface Sci 64:67–142
Tempel MV, Lucassen J, Lucassen-Reynders EH (1965) Application of surface thermodynamics to Gibbs elasticity. J Phys Chem 69:1798–1804
Phillips JN (1955) The energetics of micelle formation. Trans Faraday Soc 51:561–569
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 51363004), Guangxi Commission of Science and Technology (2012GXNSFAA053212), Funds from Key Laboratory of New Processing Technology for Nonferrous Metals and Materials (2KF-12), Guangxi Small Highland Innovation Team of Talents in Colleges and Universities, and Guangxi Funds for Specially-appointed Expert.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Deng, W., Zhang, Y., Zhong, Y. et al. Synthesis and Thermodynamic Properties of Rosin-Based Cationic Gemini Surfactants. J Surfact Deterg 17, 453–458 (2014). https://doi.org/10.1007/s11743-013-1559-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-013-1559-9