Abstract
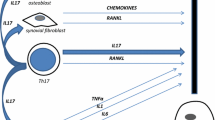
There is an emerging interest in the role of anti-TNF-α therapy in reducing bone damage in chronic arthritis with special regard to rheumatoid arthritis. Accumulation of osteoclasts in rheumatoid synovial tissues, and their activation due to osteoclastogenic cytokines and chemokines at cartilage erosion sites suggest that they may advantageously be considered as therapeutic targets. Given that the primary role of TNF-α in osteoclastogenesis, the inhibition of TNF-α represents an important strategy for reducing bone damage in rheumatoid arthritis. In point of fact, there is evidence that treatment with anti-TNF-α agents may avoid or reduce bone damage in rheumatoid arthritis, even if further studies are required to provide a biological explanation and a link for the observation of the advantageous effects of TNF-α inhibitors on the progression of bone damage in chronic arthritis. The existence of factors involved in osteoclast activation, including IL-1, IL-6, IL-7, IL-11, IL-17, M-CSF, TGF-β, MIP-1α, MIP-1β, IP-10, MIG, and OSCAR, indicates that TNF-α is only a single player in the great molecular cauldron of osteoclastogenesis. The presence of mediators behind the TNF-α and RANK-RANKL complex that may be independent in inducing osteoclastogenesis, such as NFATc1, suggests that the anti-TNF-α therapy will not provide a complete reduction of bone damage in chronic arthritis.

Similar content being viewed by others
References
Bromley M, Woolley DE (1984) Chondroclasts and osteoclasts at subchondral sites of erosion in the rheumatoid joint. Arthritis Rheum 27:968–975
Gravallese EM, Manning C, Tsay A et al (2000) Synovial tissue in rheumatoid arthritis is a source of osteoclast differentiation factor. Arthritis Rheum 43:250–258
Colucci S, Brunetti G, Cantatore FP et al (2007) Lymphocytes and synovial fluid fibroblasts support osteoclastogenesis through RANKL, TNFalpha, and IL-7 in an in vitro model derived from human psoriatic arthritis. J Pathol 212:47–55
Kim HH, Lee DE, Shin JN et al (1999) Receptor activator of NF-κB recruits multiple TRAF family adaptors and activates c-Jun N-terminal kinase. FEBS Lett 443:297–302
Matsumoto M, Sudo T, Saito T, Osada H, Tsujimoto M (2000) Involvement of p38 mitogen-activated protein kinase signaling pathway in osteoclastogenesis mediated by receptor activator of NF-κB ligand (RANKL). J Biol Chem 275:31155–31161
Kobayashi N, Kadono Y, Naito A et al (2001) Segregation of TRAF6-mediated signaling pathways clarifies its role in osteoclastogenesis. EMBO J 20:1271–1280
Bai S, Kitaura H, Zhao H et al (2005) FHL2 inhibits the activated osteoclast in a TRAF6 dependent manner. J Clin Invest 115:2742–2751
Wada T, Nakashima T, Oliveira-dos-Santos AJ et al (2005) The molecular scaffold Gab2 is a crucial component of RANK signaling and osteoclastogenesis. Nat Med 11:394–399
David J-P, Sabapathy K, Hoffmann O, Idarraga MH, Wagner EF (2002) JNK1 modulates osteoclastogenesis through both c-Jun phosphorylation- dependent and -independent mechanisms. J Cell Sci 115:4317–4325
Asagiri M, Takayanagi H (2007) The molecular understanding of osteoclast differentiation. Bone 40:251–264
Takayanagi H (2007) Osteoimmunology: shared mechanisms and crosstalk between the immune and bone systems. Nat Rev Immunol 7:292–304
Koga T, Inui M, Inoue K, Kim S, Suematsu A, Kobayashi E, Iwata T, Ohnishi H, Matozaki T, Kodama T et al (2004) Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428:758–763
Takayanagi H, Kim S, Koga T, Nishina H, Isshiki M, Yoshida H, Saiura A, Isobe M, Yokochi T, Inoue J et al (2002) Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell 3:889–901
Simonet WS, Lacey DL, Dunstan CR et al (1997) Osteoprotegerin: a novel-secreted protein involved in the regulation of bone density. Cell 89:309–319
Pageau SC (2009) Denosumab. MAbs 1:210–215
Weitzmann MN, Cenci S, Rifas L, Brown C, Pacifici R (2000) Interleukin-7 stimulates osteoclast formation by up-regulating the T-cell production of soluble osteoclastogenic cytokines. Blood 96:1873–1878
Hwang SY, Kim JY, Kim KW, Park MK, Moon Y, Kim WU et al (2004) IL-17 induces production of IL-6 and IL-8 in rheumatoid arthritis synovial fibroblasts via NF-kappaB- and PI3-kinase/Aktdependent pathways. Arthritis Res Ther 6:R120–R128
Wei S, Kitaura H, Zhou P, Ross FP, Teitelbaum SL (2005) IL-1 mediates TNF-induced osteoclastogenesis. J Clin Invest 115:282–290
Hase H, Kanno Y, Kojima H, Sakurai D, Kobata T (2008) Coculture of osteoclast precursors with rheumatoid synovial fibroblasts induces osteoclastogenesis via transforming growth factor-β mediated down-regulation of osteoprotegerin. Arthritis Rheum 58:3356–3365
Yago T, Nanke Y, Kawamoto M, Furuya T, Kobashigawa T, Kamatani N et al (2007) IL-23 induces human osteoclastogenesis via IL-17 in vitro, and anti-IL-23 antibody attenuates collagen-induced arthritis in rats. Arthritis Res Ther 9:R96
Abe M, Hiura K, Wilde J, Moriyama K, Hashimoto T, Ozaki S et al (2002) Role for macrophage inflammatory protein (MIP)-1α and MIP-1β in the development of osteolytic lesions in multiple myeloma. Blood 100:2195–2202
Kwak HB, Lee SW, Jin HM, Ha H, Lee SH, Takeshita S et al (2005) Monokine induced by interferon-γ is induced by receptor activator of nuclear factor κB ligand and is involved in osteoclast adhesion and migration. Blood 105:2963–2969
Kwak HB, Ha H, Kim HN, Lee JH, Kim HS, Lee S et al (2008) Reciprocal cross-talk between RANKL and interferon-γ-inducible protein 10 is responsible for bone-erosive experimental arthritis. Arthritis Rheum 58:1332–1342
So H, Rho J, Jeong D, Park R, Fisher DE, Ostrowski MC et al (2003) Microphthalmia transcription factor and PU.1 synergistically induce the leukocyte receptor osteoclast-associated receptor gene expression. J Biol Chem 278:24209–24216
Kim K, Kim JH, Lee J, Jin HM, Kook H, Kim KK et al (2007) MafB negatively regulates RANKL-mediated osteoclast differentiation. Blood 109:3253–3259
Herman S, Müller RB, Krönke G, Zwerina J, Redlich K, Hueber AJ et al (2008) Induction of osteoclast-associated receptor, a key osteoclast costimulation molecule, in rheumatoid arthritis. Arthritis Rheum 58:3041–3050
Polzer K, Diarra D, Zwerina J, Schett G (2008) Inflammation and destruction of the joints-the Wnt pathway. Joint Bone Spine 75:105–107
Baron R, Rawadi G, Roman-Roman S (2006) Wnt signaling: a key regulator of bone mass. Curr Top Dev Biol 76:103–127
Sen M, Lauterbach K, El-Gabalawy H, Firestein GS, Corr M, Carson DA (2000) Expression and function of wingless and frizzled homologs in rheumatoid arthritis. Proc Natl Acad Sci USA 97:2791–2796
Heiland GR, Zwerina K, Baum W, Kireva T, Distler JH, Grisanti M, Asuncion F et al (2010) Neutralisation of Dkk-1 protects from systemic bone loss during inflammation and reduces sclerostin expression. Ann Rheum Dis 69:2152–2159
Diarra D, Stolina M, Polzer K, Zwerina J, Ominsky MS, Dwyer D et al (2007) Dickkopf-1 is a master regulator of joint remodeling. Nat Med 13:156–163
Nakao A, Fukushima H, Kajiya H, Ozeki S, Okabe K (2007) RANKL-stimulated TNFa production in osteoclast precursor cells promotes osteoclastogenesis by modulating RANK signaling pathways. Biochem Biophysl Res Com 357:945–950
Fuller K, Kirstein B, Chambers TJ (2006) Murine osteoclast formation and function: differential regulation by humoral agents. Endocrinology 147:1979–1985
Abu-Amer Y, Erdmann J, Kollias G, Alexopoulou L, Ross FP, Teitelbaum SL (2000) Tumor necrosis factor receptors types 1 and 2 differentially regulate osteoclastogenesis. J Biol Chem 275:27307–27310
Kaji K, Katogi R, Azuma Y, Naito A, Inoue JI, Kudo A (2001) Tumor necrosis factor alpha-induced osteoclastogenesis requires tumor necrosis factor receptor-associated factor 6. J Bone Miner Res 16:1593–1599
Ochi S, Shinohara M, Sato K, Gober HJ, Koga T, Kodama T, Takai T, Miyasakab N, Takayanagi H (2007) Pathological role of osteoclast costimulation in arthritis-induced bone loss. PNAS 104:11394–11399
Smolen JS, Han C, Bala M, Maini RN, Kalden JR, van der Heijde D, Breedveld FC, Furst DE, Lipsky PE (2005) Evidence of radiographic benefit of treatment with infliximab plus methotrexate in rheumatoid arthritis patients who had no clinical improvement: a detailed subanalysis of data from the anti-tumor necrosis factor trial in rheumatoid arthritis with concomitant therapy study. Arthritis Rheum 52:1020–1030
Lange U, Teichmann J, Muller-Ladner U, Strunk J (2005) Increase in bone mineral density of patients with rheumatoid arthritis treated with anti-TNF-α antibody: a prospective open-label pilot study. Rheumatology (Oxford) 44:1546–1548
Catrina AI, Trollmo C, af Klint E et al (2005) Evidence that anti-tumor necrosis factor therapy with both etanercept and infliximab induces apoptosis in macrophages, but not lymphocytes, in rheumatoid arthritis joints: extended report. Arthritis Rheum 52:61–72
Saito H, Kojima T, Takahashi M, Horne WC, Baron R, Amagasa T, Ohya K, Kazuhiro Aoki K (2007) Tumor necrosis factor receptor loop peptide mimic inhibits bone destruction to the same extent as anti-tumor necrosis factor monoclonal antibody in murine collagen-induced arthritis. Arthr Rheum 56:1164–1174
Takasaki W, Kajino Y, Kajino K, Murali R, Greene MI (1997) Structure-based design and characterization of exocyclic peptidomimetics that inhibit TNF α binding to its receptor. Nat Biotechnol 15:1266–1270
Aoki K, Saito H, Itzstein C, Ishiguro M, Shibata T, Blanque R et al (2006) TNF-α receptor loop peptide mimic blocks RANK ligand-induced signaling, bone resorption, and bone loss. J Clin Invest 116:1525–1534
Redlich K, Gortz B, Hayer S, Zwerina J, Doerr N, Kostenuik P et al (2004) Repair of local bone erosions and reversal of systemic bone loss upon therapy with anti-tumor necrosis factor in combination with osteoprotegerin or parathyroid hormone in tumor necrosis factor mediated arthritis. Am J Pathol 164:543–555
Saidenberg-Kermanac’h N, Corrado A, Lemeiter D, de Vernejoul MC, Boissier MC, Cohen-Solal ME (2004) TNF-α antibodies and osteoprotegerin decrease systemic bone loss associated with inflammation through distinct mechanisms in collagen-induced arthritis. Bone 35:1200–1207
Zwerina J, Hayer S, Tohidast-Akrad M, Bergmeister H, Redlich K, Feige U, Dunstan C, Kollias G, Steiner G, Smolen J, Schett G (2004) Single and combined inhibition of tumor necrosis factor, interleukin-1, and RANKL pathways in tumor necrosis factor-induced arthritis. Arthr Rheum 50:277–290
Seriolo B, Paolino S, Sulli A, Cutolo M (2006) Are there any positive effects of TNF-alpha blockers on bone metabolism? Reumatismo 58:199–205
Hirose W, Nishikawa K, Hirose M, Nanki T, Sugimoto H (2009) Response of early active rheumatoid arthritis to tumor necrosis factor inhibitors: evaluation by magnetic resonance imaging. Mod Rheumatol 19:20–26
Anandarajah AP, Ory P, Salonen D, Feng C, Wong RL, Ritchlin CT (2010) Effect of adalimumab on joint disease: features of patients with psoriatic arthritis detected by magnetic resonance imaging. Ann Rheum Dis 69:206–209
Yao Z, Li P, Zhang Q, Schwarz EM, Keng P, Arbini A, Boyce BF, Xing L (2006) Tumor necrosis factor-alpha increases circulating osteoclast precursor numbers by promoting their proliferation and differentiation in the bone marrow through up-regulation of c-Fms expression. J Biol Chem 281:11846–11855
Musacchio E, Valvason C, Botsios C, Ostuni F, Furlan A, Ramonda R, Modesti V, Sartori L, Punzi L (2009) The tumor necrosis factor-alpha-blocking agent infliximab inhibits interleukin 1beta (IL-1beta) and IL-6 gene expression in human osteoblastic cells. J Rheumatol 36:1575–1579
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maruotti, N., d’Onofrio, F., Corrado, A. et al. Why TNF-α inhibition is not sufficient to avoid juxta-articular erosions in chronic arthritis?. Intern Emerg Med 7, 15–20 (2012). https://doi.org/10.1007/s11739-011-0526-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-011-0526-6