Abstract
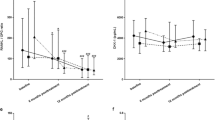
Tumor Necrosis Factor (TNF)-α and Interleukin (IL)-6 play a fundamental role in bone loss in rheumatoid arthritis (RA), partly due to the inhibition of the Wnt canonical pathway. The aim of our study was to investigate the short-term effects of three different treatments on Wnt inhibitors (Dkk-1 and sclerostin) and on bone turnover markers (BTMs): N-propeptide of type I collagen (PINP) and C-terminal telopeptide of type I collagen (β-CTX-I). We performed a retrospective analysis of prospectively collected data. We enrolled women affected by early RA (< 12 months) with active disease (DAS28 ≥ 2.6) despite a 6-month treatment with methotrexate (10–15 mg/week), who then started certolizumab pegol, tocilizumab, or methyl-prednisolone (8 mg/daily). Patients were divided into three groups according to the treatment. Blood samples were collected at baseline, week 1, and week 4. We selected 14 patients treated with certolizumab pegol, 14 patients with tocilizumab, and 20 patients with methyl-prednisolone. No difference between any of the tested parameters was found at baseline. β-CTX-I, Dkk-1, and sclerostin decreased after 1 week of treatment with certolizumab pegol (− 27% ± 21.5, − 50% ± 13.2, and − 30% ± 30.4, respectively, p < 0.05). Methyl-prednisolone induced similar changes, albeit less marked, on β-CTX-I and Wnt inhibitors, with a decrease in PINP (− 16.1% ± 16.5, p < 0.05). Tocilizumab did not significantly affect BTMs or Wnt inhibitors. No significant changes were found for PTH and 25OHD. In the first four weeks of treatment, TNFα inhibition showed strong effects on BTMs and Wnt inhibitors, differently from IL-6 blockade. Glucocorticoids induced similar changes; nonetheless, they showed undesired effects on bone formation.

Similar content being viewed by others
References
Schett G, Gravallese E (2012) Bone erosion in rheumatoid arthritis: mechanisms, diagnosis and treatment. Nat Rev Rheumatol 8:656–664. https://doi.org/10.1038/nrrheum.2012.153
Diarra D, Stolina M, Polzer K et al (2007) Dickkopf-1 is a master regulator of joint remodeling. Nat Med 13:156–163. https://doi.org/10.1038/nm1538
Fassio A, Idolazzi L, Viapiana O et al (2017) In psoriatic arthritis Dkk-1 and PTH are lower than in rheumatoid arthritis and healthy controls. Clin Rheumatol. https://doi.org/10.1007/s10067-017-3734-2
Ma Y, Zhang X, Wang M et al (2018) The serum level of Dickkopf-1 in patients with rheumatoid arthritis: a systematic review and meta-analysis. Int Immunopharmacol 59:227–232. https://doi.org/10.1016/j.intimp.2018.04.019
Rossini M, Viapiana O, Adami S et al (2015) In patients with rheumatoid arthritis, Dickkopf-1 serum levels are correlated with parathyroid hormone, bone erosions and bone mineral density. Clin Exp Rheumatol 33:77–83
Fassio A, Adami G, Gatti D et al (2018) Inhibition of tumor necrosis factor-alpha (TNF-alpha) in patients with early rheumatoid arthritis results in acute changes of bone modulators. Int Immunopharmacol 67:487–489. https://doi.org/10.1016/j.intimp.2018.12.050
Adami G, Orsolini G, Adami S et al (2016) Effects of TNF inhibitors on parathyroid hormone and Wnt signaling antagonists in rheumatoid arthritis. Calcif Tissue Int 99:360–364. https://doi.org/10.1007/s00223-016-0161-3
Terpos E, Fragiadaki K, Konsta M et al (2011) Early effects of IL-6 receptor inhibition on bone homeostasis: a pilot study in women with rheumatoid arthritis. Clin Exp Rheumatol 29:921–925
Briot K, Rouanet S, Schaeverbeke T et al (2015) The effect of tocilizumab on bone mineral density, serum levels of Dickkopf-1 and bone remodeling markers in patients with rheumatoid arthritis. Joint Bone Spine 82:109–115. https://doi.org/10.1016/j.jbspin.2014.10.015
Kawazoe M, Kaneko K, Shikano K et al (2018) Glucocorticoid therapy causes contradictory changes of serum Wnt signaling-related molecules in systemic autoimmune diseases. Clin Rheumatol 37:2169–2178. https://doi.org/10.1007/s10067-017-3689-3
Gifre L, Ruiz-Gaspà S, Monegal A et al (2013) Effect of glucocorticoid treatment on Wnt signalling antagonists (sclerostin and Dkk-1) and their relationship with bone turnover. Bone 57:272–276. https://doi.org/10.1016/j.bone.2013.08.016
Colditz J, Thiele S, Baschant U et al (2019) Osteogenic Dkk1 mediates glucocorticoid-induced but not arthritis-induced bone loss. J Bone Miner Res. https://doi.org/10.1002/jbmr.3702
Aletaha D, Neogi T, Silman AJ et al (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 69:1580–1588. https://doi.org/10.1136/ard.2010.138461
Fassio A, Rossini M, Viapiana O et al (2017) New strategies for the prevention and treatment of systemic and local bone loss; from pathophysiology to clinical application. Curr Pharm Des 23:6241–6250. https://doi.org/10.2174/1381612823666170713104431
Brabnikova Maresova K, Pavelka K, Stepan JJ (2013) Acute effects of glucocorticoids on serum markers of osteoclasts, osteoblasts, and osteocytes. Calcif Tissue Int 92:354–361. https://doi.org/10.1007/s00223-012-9684-4
Saviola G, Abdi Ali L, Shams Eddin S et al (2007) Compared clinical efficacy and bone metabolic effects of low-dose deflazacort and methyl prednisolone in male inflammatory arthropathies: a 12-month open randomized pilot study. Rheumatology (Oxford) 46:994–998. https://doi.org/10.1093/rheumatology/kem030
Adami G, Saag KG (2019) Glucocorticoid-induced osteoporosis: 2019 concise clinical review. Osteoporos Int. https://doi.org/10.1007/s00198-019-04906-x
Landewé R, van der Heijde D, Klareskog L et al (2006) Disconnect between inflammation and joint destruction after treatment with etanercept plus methotrexate: results from the trial of etanercept and methotrexate with radiographic and patient outcomes. Arthritis Rheum 54:3119–3125. https://doi.org/10.1002/art.22143
Garnero P, Sornay-Rendu E, Claustrat B, Delmas PD (2000) Biochemical markers of bone turnover, endogenous hormones and the risk of fractures in postmenopausal women: the OFELY study. J Bone Miner Res 15:1526–1536. https://doi.org/10.1359/jbmr.2000.15.8.1526
Zerbini CAF, Clark P, Mendez-Sanchez L, et al (2017) Biologic therapies and bone loss in rheumatoid arthritis. Osteoporos Int 28:429–446. https://doi.org/10.1007/s00198-016-3769-2
Guañabens N, Gifre L, Peris P (2014) The role of Wnt signaling and sclerostin in the pathogenesis of glucocorticoid-induced osteoporosis. Curr Osteoporos Rep 12:90–97. https://doi.org/10.1007/s11914-014-0197-0
Compston J (2018) Glucocorticoid-induced osteoporosis: an update. Endocrine 61:7–16. https://doi.org/10.1007/s12020-018-1588-2
Sato AY, Cregor M, Delgado-Calle J et al (2016) Protection from glucocorticoid-induced osteoporosis by anti-catabolic signaling in the absence of sost/sclerostin. J Bone Miner Res 31:1791–1802. https://doi.org/10.1002/jbmr.2869
Yao W, Dai W, Jiang L et al (2016) Sclerostin-antibody treatment of glucocorticoid-induced osteoporosis maintained bone mass and strength. Osteoporos Int 27:283–294. https://doi.org/10.1007/s00198-015-3308-6
O’Brien CA, Jia D, Plotkin LI et al (2004) Glucocorticoids act directly on osteoblasts and osteocytes to induce their apoptosis and reduce bone formation and strength. Endocrinology 145:1835–1841. https://doi.org/10.1210/en.2003-0990
Zhou M, Li S, Pathak JL (2019) Pro-inflammatory cytokines and osteocytes. Curr Osteoporos Rep 17:97–104. https://doi.org/10.1007/s11914-019-00507-z
Heiland GR, Appel H, Poddubnyy D et al (2012) High level of functional dickkopf-1 predicts protection from syndesmophyte formation in patients with ankylosing spondylitis. Ann Rheum Dis 71:572–574. https://doi.org/10.1136/annrheumdis-2011-200216
Chen X-X, Baum W, Dwyer D et al (2013) Sclerostin inhibition reverses systemic, periarticular and local bone loss in arthritis. Ann Rheum Dis 72:1732–1736. https://doi.org/10.1136/annrheumdis-2013-203345
Metzger CE, Narayanan SA (2019) The role of osteocytes in inflammatory bone loss. Front Endocrinol (Lausanne) 10:285. https://doi.org/10.3389/fendo.2019.00285
Yeremenko N, Zwerina K, Rigter G et al (2015) Tumor necrosis factor and interleukin-6 differentially regulate Dkk-1 in the inflamed arthritic joint. Arthritis Rheumatol (Hoboken, NJ) 67:2071–2075. https://doi.org/10.1002/art.39183
Appel H, Ruiz-Heiland G, Listing J et al (2009) Altered skeletal expression of sclerostin and its link to radiographic progression in ankylosing spondylitis. Arthritis Rheum 60:3257–3262. https://doi.org/10.1002/art.24888
Seeman E, Martin TJ (2019) Antiresorptive and anabolic agents in the prevention and reversal of bone fragility. Nat Rev Rheumatol 15:225–236. https://doi.org/10.1038/s41584-019-0172-3
Smolen JS, Landewé R, Bijlsma J et al (2017) EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 76:960–977. https://doi.org/10.1136/annrheumdis-2016-210715
Chen Y-M, Chen H-H, Huang W-N et al (2017) Tocilizumab potentially prevents bone loss in patients with anticitrullinated protein antibody-positive rheumatoid arthritis. PLoS ONE 12:e0188454. https://doi.org/10.1371/journal.pone.0188454
Kume K, Amano K, Yamada S et al (2014) The effect of tocilizumab on bone mineral density in patients with methotrexate-resistant active rheumatoid arthritis. Rheumatology (Oxford) 53:900–903. https://doi.org/10.1093/rheumatology/ket468
Shim J-H, Stavre Z, Gravallese EM (2018) Bone loss in rheumatoid arthritis: basic mechanisms and clinical implications. Calcif Tissue Int 102:533–546. https://doi.org/10.1007/s00223-017-0373-1
Fassio A, Idolazzi L, Jaber MA et al (2016) The negative bone effects of the disease and of chronic corticosteroid treatment in premenopausal women affected by rheumatoid arthritis. Reumatismo 68:65–71. https://doi.org/10.4081/reumatismo.2016.878
Güler-Yüksel M, Hoes JN, Bultink IEM, Lems WF (2018) Glucocorticoids, inflammation and bone. Calcif Tissue Int 102:592–606. https://doi.org/10.1007/s00223-017-0335-7
Sambrook PN, Eisman JA, Yeates MG et al (1986) Osteoporosis in rheumatoid arthritis: safety of low dose corticosteroids. Ann Rheum Dis 45:950–953
Blavnsfeldt A-BG, de Thurah A, Thomsen MD et al (2018) The effect of glucocorticoids on bone mineral density in patients with rheumatoid arthritis: a systematic review and meta-analysis of randomized, controlled trials. Bone 114:172–180. https://doi.org/10.1016/j.bone.2018.06.008
Robling AG, Niziolek PJ, Baldridge LA et al (2008) Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/sclerostin. J Biol Chem 283:5866–5875. https://doi.org/10.1074/jbc.M705092200
Acknowledgements
The authors would like to thank the LURM (Laboratorio Universitario di Ricerca Medica) Research Center, University of Verona, and especially Eliana Amato for performing the biochemical analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human and Animal Rights and Informed Consent
The study was conducted according to the protocol 1483CESC approved by our local Ethics Committee, in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from all individual participants included.
Conflict of interest
Maurizio Rossini reports personal fees from AbbVie, Abiogen, Eli-Lilly, Merck Sharp & Dohme, Novartis, Sanofi, UCB, outside the submitted work. Angelo Fassio, Giovanni Adami, Alessandro Giollo, Ombretta Viapiana, Nazzarena Malavolta, Gianantonio Saviola, Roberto Bortolotti, Luca Idolazzi, Francesco Bertoldo and Davide Gatti have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fassio, A., Adami, G., Giollo, A. et al. Acute Effects of Glucocorticoid Treatment, TNFα or IL-6R Blockade on Bone Turnover Markers and Wnt Inhibitors in Early Rheumatoid Arthritis: A Pilot Study. Calcif Tissue Int 106, 371–377 (2020). https://doi.org/10.1007/s00223-019-00649-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-019-00649-3