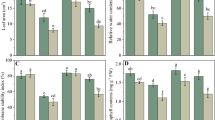
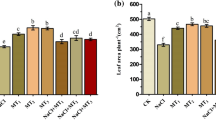
Abstract
Salt stress usually results in severe physiological damage to plants. Melatonin (N-acetyl-5-methoxytryptamine) is an important growth regulator that adapts plants to abiotic stress. The present study evaluated the role of melatonin application on inducing salt tolerance in sugar beet (Beta vulgaris L.). The protective role of melatonin (0, 30, 60, and 90 µM) was examined by measuring leaf photosynthetic characteristics, antioxidant system, and osmotic adjustment substances of sugar beet seedlings under salt (300 mM Na+) and non-salt stresses. The results showed that salt stress resulted in significantly reduced biomass, reduced photochemical activity of photosystem II (PSII), and evoked the production of reactive oxygen species (ROS). In contrast, the application of melatonin significantly increased antioxidant enzyme activities (SOD, POD, and CAT) under salt stress, reduced ROS accumulation (MDA and O2·−), and enhanced photosynthesis in seedlings. There was no significant difference in the above indicators of melatonin pretreatment under control condition (non-salinized). On day 1 of stress application, the concentration of sucrose decreased significantly, and the concentration of proline and H2O2 increased significantly under melatonin treatment. On day 7, soluble sugar and betaine concentrations increased significantly. Current research speculates that melatonin enhances cellular energy metabolism and may be involved in activating the antioxidant system to eliminate ROS. In conclusion, these results indicated that the application of 60 µM melatonin could act as a feasible way to alleviate the salt stress in sugar beet production.








Similar content being viewed by others
Abbreviations
- Car:
-
Carotenoids
- CAT:
-
Catalase
- Chl:
-
Chlorophyll
- Ci:
-
Intercellular CO2 concentration
- E :
-
Transpiration rate
- F v/F m :
-
Maximum quantum yield of PSII
- LRWC:
-
Leave relative water content
- MDA:
-
Malondialdehyde content
- g s :
-
Stomatal conductance
- Pn:
-
Net photosynthetic rate
- q L :
-
Estimates the fraction of open PSII centers
- POD:
-
Peroxidase
- q P :
-
Photochemical quenching
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- Y(II):
-
Effective quantum yield of PSII
- Y(NO):
-
Quantum yield of nonregulated non-photochemical energy dissipation
- Y(NPQ):
-
Quantum yield of regulated non-photochemical energy dissipation
References
Aebi H (1984) Catalase in vitro methods. Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahmad S et al (2019) Exogenous melatonin confers drought stress by promoting plant growth, photosynthetic capacity and antioxidant defense system of maize seedlings. PeerJ 7:e7793. https://doi.org/10.7717/peerj.7793
Alcázar Á, Jurado JM, Martín MJ, Pablos F, González AG (2005) Enzymatic-spectrophotometric determination of sucrose in coffee beans. Talanta 67:760–766. https://doi.org/10.1016/j.talanta.2005.04.005
Arnao MB, Hernández-Ruiz J (2015) Functions of melatonin in plants: a review. J Pineal Res 59:133–150. https://doi.org/10.1111/jpi.12253
Arnao MB, Hernández-Ruiz J (2017) Growth activity, rooting capacity, and tropism: three auxinic precepts fulfilled by melatonin. Acta Physiol Plant 39:127. https://doi.org/10.1007/s11738-017-2428-3
Arnao MB, Hernández-Ruiz J (2018) Melatonin: a new plant hormone and/or a plant master regulator? Trends Plant Sci. https://doi.org/10.1016/j.tplants.2018.10.010
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Ben Ahmed C, Ben Rouina B, Sensoy S, Boukhriss M, Ben Abdullah F (2010) Exogenous proline effects on photosynthetic performance and antioxidant defense system of young olive tree. J Agric Food Chem 58:4216–4222. https://doi.org/10.1021/jf9041479
Bor M, Ozdemir F, Turkan I (2003) The effect of salt stress on lipid peroxidation and antioxidants in leaves of sugar beet Beta vulgaris L. and wild beet Beta maritima L. Plant Sci 164:77–84. https://doi.org/10.1016/s0168-9452(02)00338-2
Bota J, Medrano H, Flexas J (2004) Is photosynthesis limited by decreased rubisco activity and RuBP content under progressive water stress? New Phytol 162:671–681. https://doi.org/10.1111/j.1469-8137.2004.01056.x
Chao L et al (2012) The mitigation effects of exogenous melatonin on salinity-induced stress in Malus hupehensis. J Pineal Res 53:298–306. https://doi.org/10.1111/j.1600-079X.2012.00999.x
Chen Z et al (2007) Root plasma membrane transporters controlling K+/Na+ homeostasis in salt-stressed barley. Plant Physiol 145:1714–1725. https://doi.org/10.1104/pp.107.110262
Chen YE et al (2018) Exogenous melatonin enhances salt stress tolerance in maize seedlings by improving antioxidant and photosynthetic capacity. Physiol Plant 164:349–363. https://doi.org/10.1111/ppl.12737
Crawford T, Lehotai N, Strand A (2018) The role of retrograde signals during plant stress responses. J Exp Bot 69:2783–2795. https://doi.org/10.1093/jxb/erx481
Cristiano G, Camposeo S, Fracchiolla M, Vivaldi G, De Lucia B, Cazzato E (2016) Salinity differentially affects growth and ecophysiology of two mastic tree (Pistacia lentiscus L.) accessions. Forests 7:156. https://doi.org/10.3390/f7080156
Cuin TA, Shabala S (2010) Compatible solutes reduce ROS-induced potassium efflux in Arabidopsis roots. Plant Cell Environ 30:875–885. https://doi.org/10.1111/j.1365-3040.2007.01674.x
De Block M, Verduyn C, De Brouwer D, Cornelissen M (2005) Poly(ADP-ribose) polymerase in plants affects energy homeostasis, cell death and stress tolerance. Plant J 41:95–106. https://doi.org/10.1111/j.1365-313X.2004.02277.x
Egea I et al (2018) The SlCBL10 calcineurin B-like protein ensures plant growth under salt stress by regulating Na+ and Ca2+ homeostasis. Plant Physiol 176:1676–1693. https://doi.org/10.1104/pp.17.01605
FAO (2009) FAO land and plant nutrition management service. http://www.fao.org/ag/agl/agll/spush. Accessed 10 Oct 2019
Farouk S, Al-Amri S (2019a) Ameliorative roles of melatonin and/or zeolite on chromium-induced leaf senescence in marjoram plants by activating antioxidant defense, osmolyte accumulation, and ultrastructural modification. Ind Crops Prod 142:111823. https://doi.org/10.1016/j.indcrop.2019.111823
Farouk S, Al-Amri SM (2019b) Exogenous melatonin-mediated modulation of arsenic tolerance with improved accretion of secondary metabolite production, activating antioxidant capacity and improved chloroplast ultrastructure in rosemary herb. Ecotoxicol Environ Saf 180:333–347. https://doi.org/10.1016/j.ecoenv.2019.05.021
Farouk S, Al-Amri SM (2019c) Exogenous zinc forms counteract NaCl-induced damage by regulating the antioxidant system, osmotic adjustment substances, and ions in canola (Brassica napus L. cv. Pactol) plants. J Soil Sci Plant Nutr 19:887–899. https://doi.org/10.1007/s42729-019-00087-y
Farouk S, Arafa SA (2018) Mitigation of salinity stress in canola plants by sodium nitroprusside application Span. J Agric Res 16:0802. https://doi.org/10.5424/sjar/2018163-13252
Fu Y et al (2014) Bioaccumulation, subcellular, and molecular localization and damage to physiology and ultrastructure in Nymphoides peltata (Gmel.) O. Kuntze exposed to yttrium. Environ Sci Pollut Res 21:2935–2942. https://doi.org/10.1007/s11356-013-2246-0
Gao WY, Feng Z, Bai QQ, He JJ, Wang YJ (2019) Melatonin-mediated regulation of growth and antioxidant capacity in salt-tolerant naked oat under salt stress. Int J Mol Sci. https://doi.org/10.3390/ijms20051176
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta (BBA)-Gen Subj 990:87–92. https://doi.org/10.3390/ijms20051176
Gorham J (1984) Separation of plant betaines and their sulphur analogues by cation-exchange high-performance liquid chromatography. J Chromatogr 287:345–351. https://doi.org/10.1016/S0021-9673(01)87710-4
Helaly M, Farouk S, Arafa SA, Amhimmid NB (2018) Inducing salinity tolerance of rosemary (Rosmarinus officinalis L.) plants by chitosan or zeolite application. Asian J Adv Agric Res 5:1–20. https://doi.org/10.9734/AJAAR/2018/40051
Hossain MS, Elsayed AI, Moore M, Dietz KJ (2017) Redox and reactive oxygen species network in acclimation for salinity tolerance in sugar beet. J Exp Bot 68:1283–1298. https://doi.org/10.1093/jxb/erx019
Jaleel CA et al (2009) Antioxidant defense responses: physiological plasticity in higher plants under abiotic constraints. Acta Physiol Plant 31:427–436. https://doi.org/10.1007/s11738-009-0275-6
Jamsheer KM, Laxmi A (2015) Expression of Arabidopsis FCS-Like Zinc finger genes is differentially regulated by sugars, cellular energy level, and abiotic stress. Front Plant Sci. https://doi.org/10.3389/fpls.2015.00746
Ke Q et al (2016) Transgenic poplar expressing codA exhibits enhanced growth and abiotic stress tolerance. Plant Physiol Biochem 100:75–84. https://doi.org/10.1016/j.plaphy.2016.01.004
Kitajima M, Butler W (1975) Quenching of chlorophyll fluorescence and primary photochemistry in chloroplasts by dibromothymoquinone. Biochim Biophys Acta (BBA)-Bioenerg 376:105–115. https://doi.org/10.1016/0005-2728(75)90209-1
Kolár J, Machácková I (2010) Melatonin in higher plants: occurrence and possible functions. J Pineal Res 39:333–341. https://doi.org/10.1111/j.1600-079X.2005.00276.x
Lawlor D (1995) The effects of water deficit on photosynthesis. Environ Plant Metab 5:129–160
Li H et al (2017a) Exogenous melatonin confers salt stress tolerance to watermelon by improving photosynthesis and redox homeostasis. Front Plant Sci 8:295. https://doi.org/10.1111/j.1600-079X.2005.00276.x
Li H et al (2017b) Exogenous melatonin confers salt stress tolerance to watermelon by improving photosynthesis and redox homeostasis. Front Plant Sci. https://doi.org/10.3389/fpls.2017.00295
Li Q, Lv LR, Teng YJ, Si LB, Ma T, Yang YL (2018) Apoplastic hydrogen peroxide and superoxide anion exhibited different regulatory functions in salt-induced oxidative stress in wheat leaves. Biol Plant 62:750–762. https://doi.org/10.1007/s10535-018-0808-1
Li JP, Liu J, Zhu TT, Zhao C, Li LY, Chen M (2019) The role of melatonin in salt stress responses. Int J Mol Sci. https://doi.org/10.3390/ijms20071735
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents analysis (Peach). Biochem Soc Transc 11:591–592
Liu F, Pang SJ (2010) Stress tolerance and antioxidant enzymatic activities in the metabolisms of the reactive oxygen species in two intertidal red algae Grateloupia turuturu and Palmaria palmata. J Exp Mar Biol Ecol 382:82–87. https://doi.org/10.1016/j.jembe.2009.11.005
Liu D, Liu M, Liu X-L, Cheng X-G, Liang Z-W (2018) Silicon priming created an enhanced tolerance in alfalfa (Medicago sativa L.) seedlings in response to high alkaline stress. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00716
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Neto ADDA, Prisco JT, Enéas-Filho J, Medeiros JVR, Gomes-Filho E (2005) Hydrogen peroxide pre-treatment induces saltstress acclimation in maize plants. J Plant Physiol 162:1114–1122. https://doi.org/10.1016/j.jplph.2005.01.007
Nishimura N, Zhang J, Abo M, Okubo A, Yamazaki S (2001) Application of capillary electrophoresis to the simultaneous determination of betaines in plants. Anal Sci 17:103–106. https://doi.org/10.2116/analsci.17.103
Pape C, Lüning K (2006) Quantification of melatonin in phototrophic organisms. J Pineal Res 41:157–165. https://doi.org/10.1111/j.1600-079X.2006.00348.x
Pekal A, Pyrzynska K (2014) Evaluation of aluminium complexation reaction for flavonoid content assay. Food Anal Methods 7:1776–1782. https://doi.org/10.1007/s12161-014-9814-x
Pfündel E, Klughammer C, Schreiber U (2008) Monitoring the effects of reduced PS II antenna size on quantum yields of photosystems I and II using the Dual-PAM-100 measuring system. PAM Appl Notes 1:21–24
Rady MO, Semida WM, El-Mageed TAA, Hemida KA, Rady MM (2018) Up-regulation of antioxidative defense systems by glycine betaine foliar application in onion plants confer tolerance to salinity stress. Sci Hortic 240:614–622. https://doi.org/10.1016/j.scienta.2018.06.069
Reiter RJ, Tan DX, Galano A (2014) Melatonin: exceeding expectations. Physiology 29:325–333. https://doi.org/10.1152/physiol.00011.2014
Ren B, Zhang J, Dong S, Liu P, Zhao B (2018) Exogenous 6-benzyladenine improves antioxidative system and carbon metabolism of summer maize waterlogged in the field. J Agro Crop Sci 204:175–184. https://doi.org/10.1111/jac.12253
Sharkey TD, Bernacchi CJ, Farquhar GD, Singsaas EL (2007) Fitting photosynthetic carbon dioxide response curves for C3 leaves. Plant Cell Environ 30:1035–1040. https://doi.org/10.1111/j.1365-3040.2007.01710.x
Shi H, Ye T, Chan Z (2013) Exogenous application of hydrogen sulfide donor sodium hydrosulfide enhanced multiple abiotic stress tolerance in bermudagrass (Cynodon dactylon (L). Pers.). Plant Physiol Biochem 71:226–234. https://doi.org/10.1016/j.plaphy.2013.07.021
Siddiqui MH et al (2019) Exogenous melatonin counteracts NaCl-induced damage by regulating the antioxidant system, proline and carbohydrates metabolism in tomato seedlings. Int J Mol Sci. https://doi.org/10.3390/ijms20020353
Smart RE, Bingham GE (1974) Rapid estimates of relative water content. Plant Physiol 53:258–260. https://doi.org/10.1104/pp.53.2.258
Smirnoff N, Arnaud D (2019) Hydrogen peroxide metabolism and functions in plants. New Phytol 221:1197–1214. https://doi.org/10.1111/nph.15488
Spiro RG (1966) Analysis of sugars found in glycoproteins. Methods Enzymol 8:3–26. https://doi.org/10.1016/0076-6879(66)08005-4
Stewart RR, Bewley JD (1980) Lipid peroxidation associated with accelerated aging of soybean axes. Plant Physiol 65:245–248. https://doi.org/10.1104/pp.65.2.245
Sui N, Wang Y, Liu SS, Yang Z, Wang F, Wan SB (2018) Transcriptomic and physiological evidence for the relationship between unsaturated fatty acid and salt stress in peanut. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00007
Tahjib-Ul-Arif M, Afrin S, Polash MAS, Akter T, Ray SR, Hossain MT, Hossain MA (2019) Role of exogenous signaling molecules in alleviating salt-induced oxidative stress in rice (Oryza sativa L.): a comparative study. Acta Physiol Plant. https://doi.org/10.1007/s11738-019-2861-6
Van Tassel DL, Roberts N, Lewy A, O’Neill SD (2010) Melatonin in plant organs. J Pineal Res 31:8–15. https://doi.org/10.1034/j.1600-079X.2001.310102.x
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151(59):66. https://doi.org/10.1016/S0168-9452(99)00197-1
Vicente O, Al Hassan M, Boscaiu M (2016) Contribution of osmolyte accumulation to abiotic stress tolerance in wild plants adapted to different stressful environments. Osmolytes and plants acclimation to changing environment: emerging omics technologies. Springer, pp 13–25. https://doi.org/10.1007/978-81-322-2616-1_2
Wang L, Liu J, Wang W, Sun Y (2016) Exogenous melatonin improves growth and photosynthetic capacity of cucumber under salinity-induced stress. Photosynthetica 54:19–27. https://doi.org/10.1007/s11099-015-0140-3
Wang Y, Reiter RJ, Chan Z (2018) Phytomelatonin: a universal abiotic stress regulator. J Exp Bot 69:963. https://doi.org/10.1093/jxb/erx473
Wei W et al (2015) Melatonin enhances plant growth and abiotic stress tolerance in soybean plants. J Exp Bot 66:695–707. https://doi.org/10.1093/jxb/eru392
Yang C, Chong J, Li C, Kim C, Shi D, Wang D (2007) Osmotic adjustment and ion balance traits of an alkali resistant halophyte Kochia sieversiana during adaptation to salt and alkali conditions. Plant Soil 294:263–276. https://doi.org/10.1007/s11104-007-9251-3
You J, Chan ZL (2015) ROS regulation during abiotic stress responses in crop plants. Front Plant Sci. https://doi.org/10.3389/fpls.2015.01092
Zhang JR et al (2017a) Melatonin alleviates aluminium toxicity through modulating antioxidative enzymes and enhancing organic acid anion exudation in soybean. Funct Plant Biol 44:961–968. https://doi.org/10.1071/fp17003
Zhang N et al (2017b) Proteomic analysis reveals a role of melatonin in promoting cucumber seed germination under high salinity by regulating energy production. Sci Rep 7:503. https://doi.org/10.1038/s41598-017-00566-1
Zhang W, Liu SH, Li CC, Zhang PY, Zhang PY (2019) Transcriptome sequencing of Antarctic moss under salt stress emphasizes the important roles of the ROS-scavenging system. Gene 696:122–134. https://doi.org/10.1016/j.gene.2019.02.037
Zhao Y, Yu H, Zhou J-M, Smith SM, Li J (2020) Malate circulation: linking chloroplast metabolism to mitochondrial ROS. Trends Plant Sci. https://doi.org/10.1016/j.tplants.2020.01.010
Zhou XT, Zhao HL, Cao K, Hu LP, Du TH, Baluska F, Zou ZR (2016) Beneficial roles of melatonin on redox regulation of photosynthetic electron transport and synthesis of D1 protein in tomato seedlings under salt stress. Front Plant Sci. https://doi.org/10.3389/fpls.2016.01823
Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6:66–71. https://doi.org/10.1016/S1360-1385(00)01838-0
Zou CL et al (2019) Photosynthetic capacity, osmotic adjustment and antioxidant system in sugar beet (Beta vulgaris L.) in response to alkaline stress. Photosynthetica 57:350–360. https://doi.org/10.32615/ps.2019.010
Acknowledgements
This work was supported by the National Natural Science Foundation of China (32071973 and 31671622). Special thanks to Fuxin Zhao for his help and encouragement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Communicated by Z.-L. Zhang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, L., Wang, Z., Gai, Z. et al. Exogenous application of melatonin improves salt tolerance of sugar beet (Beta vulgaris L.) seedlings. Acta Physiol Plant 44, 57 (2022). https://doi.org/10.1007/s11738-022-03389-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-022-03389-4