Abstract
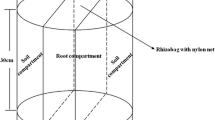
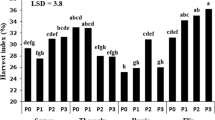
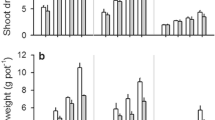
Plant available phosphorus (P) is limited in the vast rice-fallow areas where the soil is mainly acidic. Screening of fifty lentil genotypes in soil cylinder filled with solid-phase P-buffer identified genotypes with high P-acquisition ability even though desorbed P was 100 μmol/g only. Genotypes with high P-acquisition ability were identified based on their almost equal P-acquisition ability both in P-deficient and sufficient conditions. Root dry weight and root surface area showed a significant positive correlation with shoot phosphate content in P-deficiency condition, but not in P-sufficiency condition. Identified top three tolerant genotypes, namely IC560183, WBL77, and BM3 showed a 30% reduced seed yield in P-deficient field as compared to 60% or more reduction by intolerant genotypes. In P-Deficiency condition, 4–6 times high intercellular acid phosphatase activity in roots of the tolerant genotypes may be one of the factors for high P-acquisition. Higher external secretion of acid phosphatase in tolerant genotypes was also confirmed by blue colour through 5-bromo-4-chloro-3-indolyl-phosphate (BCIP) staining. P-deficiency tolerant lentil genotypes will be useful in expansion and enhancing the productivity of lentil in vast rice-fallow areas, mostly where plant-available P is low.



Similar content being viewed by others
Availability of data and materials
All data are available (already attached as the supplementary files).
References
Bhadouria J, Singh AP, Mehra P, Verma L, Srivastawa R, Parida SK, Giri J (2017) Identification of purple acid phosphatases in Chickpea and potential roles of CaPAP7 in seed phytate accumulation. Sci Rep 7:1–12
Chiou TJ, Lin SI (2011) Signaling network in sensing phosphate availability in plants. Annu Rev Plant Biol 62:185–206
De Groot C, Marcelis LFM, Van den Boogaard R, Lambers H (2001) Growth and dry mass partitioning in tomato as affected by phosphorus nutrition and light. Plant Cell Environ 24:1309–1317
Gahoonia TS, Nielsen NE (2004) Barley genotypes with long root hairs sustain high grain yields in low-P field. Plant Soil 262:55–62
Gahoonia TS, Ali O, Sarker A, Nielsen NE, Rahman MM (2006) Genetic variation in root traits and nutrient acquisition of lentil genotypes. J Plant Nutr 29:643–655
Gamuyao R, Chin JH, Pariasca-Tanaka J, Pesaresi P, Catausan S, Dalid C, Heuer S (2012) The protein kinase Pstol1 from traditional rice confers tolerance of phosphorus deficiency. Nature 488:535–539
Gerke J, Beissner L, Romer W (2000) The quantitative effect of chemical phosphate mobilization by carboxylate anions on P uptake by a single root. I. The basic concept and determination of soil parameters. J Plant Nutr Soil Sci 163:207–212
Gong X, Wheeler R, Bovill WD, McDonald GK (2016) QTL mapping of grain yield and phosphorus efficiency in barley in a Mediterranean-like environment. Theor Appl Genet 129:1657–1672
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci 11:610–617
Hernández G, Ramírez M, Valdés-López O, Tesfaye M, Graham MA, Czechowski T, Wu HC (2007) Phosphorus stress in common bean: root transcript and metabolic responses. Plant Physiol 144:752–767
Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237:173–195
Johnson N, Johnson CR, Thavarajah P, Kumar S, Thavarajah D (2020) The roles and potential of lentil prebiotic carbohydrates in human and plant health. Plants People Planet 2(4):310–319
Jones DL (1998) Organic acids in the rhizosphere-a critical review. Plant Soil 205:25–44
Kuiper D, Kuiper PJC, Lambers H, Schuit JT, Staal M (1989) Cytokinin contents in relation to mineral nutrition and benzyladenine addition in Plantago major ssp. pleiosperma. Physiol Plant 75:511–517
Kumar A, Pandey A, Gowda MVC (2015) Evaluation of groundnut genotypes for tolerance to phosphorus deficiency. J Plant Nutr 38:687–699
Kumar R, Mishra JS, Upadhyay PK, Hans H (2019) Rice fallows in the Eastern India: problems and prospects. Indian J Agric Sci 89:567–577
López-Bucio J, Hernández-Abreu E, Sánchez-Calderón L, Nieto-Jacobo MF, Simpson J, Herrera-Estrella L (2002) Phosphate availability alters architecture and causes changes in hormone sensitivity in the Arabidopsis root system. Plant Physiol 129:244–256
Lynch J, Epstein E, Läuchli A, Weight GI (1990) An automated greenhouse sand culture system suitable for studies of P nutrition. Plant, Cell Environ 13:547–554
Mukherjee A, Sarkar S, Chakraborty AS, Yelne R, Kavishetty V, Biswas T, Bhattacharyya S (2014) Phosphate acquisition efficiency and phosphate starvation tolerance locus (PSTOL1) in rice. J Genet 93:683–688
Nestler J, Wissuwa M (2016) Superior root hair formation confers root efficiency in some, but not all, rice genotypes upon P deficiency. Front Plant Sci 7:1935. https://doi.org/10.3389/fpls.2016
Sarkar S, Yelne R, Chatterjee M, Das P, Debnath S, Chakraborty A, Bhattacharyya S (2011) Screening for phosphorus (P) tolerance and validation of Pup-1 linked markers in indica rice. Indian J Genet Plant Breed 71:209–213
Su J, Xiao Y, Li M, Liu Q, Li B, Tong Y, Li Z (2006) Mapping QTLs for phosphorus-deficiency tolerance at wheat seedling stage. Plant Soil 281:25–36
Sulieman S, Ha CV, Schulze J, Tran LSP (2013) Growth and nodulation of symbiotic Medicago truncatula at different levels of phosphorus availability. J Exp Bot 64:2701–2712
Tadano T, Sakai H (1991) Secretion of acid phosphatase by the roots of several crop species under phosphorus-deficient conditions. Soil Sci Plant Nutr 37:129–140
Tian J, Wang C, Zhang Q, He X, Whelan J, Shou H (2012) Overexpression of OsPAP10a, a root-associated acid phosphatase, increased extracellular organic phosphorus utilization in rice. J Integr Plant Biol 54:631–639
Tomscha JL, Trull MC, Deikman J, Lynch JP, Guiltinan MJ (2004) Phosphatase under-producer mutants have altered phosphorus relations. Plant Physiol 135:334–345
Williamson LC, Ribrioux SP, Fitter AH, Leyser HO (2001) Phosphate availability regulates root system architecture in Arabidopsis. Plant Physiol 126:875–882
Wissuwa M, Ae N (2001) Genotypic variation for tolerance to phosphorus deficiency in rice and the potential for its exploitation in rice improvement. Plant Breed 120:43–48
Xiao K, Zhang JH, Harrison M, Wang ZY (2006) Ectopic expression of a phytase gene from Medicago truncatula barrel medic enhances phosphorus absorption in plants. J Integr Plant Biol 48:35–43
Zhang Q, Wang C, Tian J, Li K, Shou H (2011) Identification of rice purple acid phosphatases related to phosphate starvation signalling. Plant Biol 13:7–15
Acknowledgements
Authors acknowledge ICARDA for the fund support; NBPGR for sharing the germplasm; AR acknowledges the UGC-National Fellowship for OBC, Government of India.
Funding
The International Center for Agriculture Research in the Dry Areas (ICARDA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Conflict of interest none.
Ethical declaration
Not applicable.
Consent for the publication
On behalf of all the authors, corresponding author gives the consent for publication of the document.
Additional information
Communicated by S. Srivastava.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ganguly, S., Roy, A., Murmu, S.K. et al. Variation in P-acquisition ability and acid phosphatase activity at the early vegetative stage of lentil and their validation on P-deficiency field. Acta Physiol Plant 43, 109 (2021). https://doi.org/10.1007/s11738-021-03280-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-021-03280-8