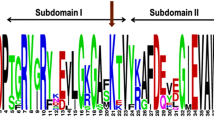
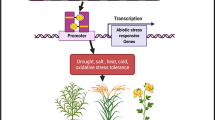
Abstract
Plants respond to abiotic stresses such as salinity, extreme temperature and drought by the activation of complex intracellular signaling cascades that regulate acclimatory biochemical and physiological changes. Protein kinases are major signal transduction factors that play a central role in mediating acclimation to environmental changes in eukaryotic organisms. It is well known that changes in abiotic conditions such as the concentration of ions, temperature and humidity lead to modulation of polyamine contents in plants. However, little is known about the relevant part these polyamines play in abiotic stress responses. Here, we address a specific role of spermidine during high salt stress by studying its interaction with OSPDK, a sucrose nonfermenting 1-related protein kinase2 (SnRK2)-type serine/threonine protein kinase SAPK4 homolog in indica rice. In this report, we demonstrate that spermidine mediates in vitro phosphorylation of OSBZ8, a bZIP class of ABRE-binding transcription factor, by OSPDK. Our results give a first-hand indication of the pivotal role played by polyamines in abiotic stress cell signaling in plants.










Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- ABRE:
-
Abscisic acid responsive element
- bZIP:
-
Basic leucine zipper
- EMSA:
-
Electrophoretic mobility shift assay
- GST:
-
Glutathione S-transferase
- MBP:
-
Myelin basic protein
- PA:
-
Polyamine
- Put:
-
Putrescine
- SNF-1:
-
Sucrose non-fermenting protein 1
- SnRK2:
-
SNF1-related protein kinase 2
- Spd:
-
Spermidine
- Spm:
-
Spermine
References
Alcazar R, Marco F, Cuevas JC, Patron M, Ferrando A, Carrasco P (2006) Involvement of polyamines in plant response to abiotic stress. Biotech Lett 28:1867–1876
Alcázar R, Altabella T, Marco F, Bortolotti C, Reymond M, Koncz C, Carrasco P, Tiburcio A (2010) Polyamines: molecules with regulatory functions in plant abiotic stress tolerance. Planta 231:1237–1249
Alcázar R, Cuevas JC, Planas J, Zarza X, Bortolotti C, Carrasco P, Salinas J, Tiburcio AF, Altabella T (2011) Integration of polyamines in the cold acclimation response. Plant Sci 180(1):31–38
Basu R, Ghosh B (1991) Polyamines in various rice genotypes with respect to NaCl salinity. Physiol Plant 82:575–581
Batistic O, Kudla J (2004) Integration and channeling of calcium signaling through the CBL calcium sensor/CIPK protein kinase network. Planta 219:915–924
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein using the principle of protein–dye binding. Anal Biochem 72:248–254
Bueno M, Matilla A (1992) Abscisic acid increases the content of free polyamines and delays mitotic activity induced by spermine in isolated embryonic axes of chickpea seeds. Physiol Plant 85:531–536
Celano P, Baylin SB, Casero RA Jr (1989) PAs differentially modulate the transcription of growth-associated genes in human colon carcinoma cells. J Biol Chem 264:8922–8927
Chomczynski P, Sacchi N (1987) Single step method of RNA isolation by acid guanidium thiocyanate–phenol–chloroform extraction. Anal Biochem 162:156–159
Cochet C, Chambaz EM (1983) PA-mediated protein phosphorylations: a possible target for intracellular PA action. Mol Cell Endocrinol 30(3):247–266
Dasgupta M (1994) Characterization of a calcium dependent protein kinase from Arachis hypogea (ground nut) seeds. Plant Physiol 104:961–969
Datta N, Schell MB, Roux SJ (1987) Spermine stimulation of a nuclear NII kinase from pea plumules and its role in the phosphorylation of a nuclear polypeptide. Plant Physiol 84:1397–1401
Fujii H, Verslues PE, Zhu JK (2007) Identification of two protein kinases required for abscisic acid regulation of seed germination, root growth and gene expression in Arabidopsis. Plant Cell 19(2):485–494
Furihata T, Maruyama K, Fujita Y, Umezawa T, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K (2006) Abscisic acid dependent multisite phosphorylation regulates the activity of a transcription activator AREB1. Proc Natl Acad Sci USA 103(6):1988–1993
Garnier J, Osguthorpe DJ, Robson B (1978) Analysis of the accuracy and implications of simple methods for predicting the secondary structure of globular proteins. J Mol Biol 120(1):97–120
Gill SS, Tuteja N (2010) Polyamines and abiotic stress tolerance in plants. Plant Signal Behav 5(1):26–33
Gupta S, Chattopadhyay MK, Chatterjee P, Ghosh B, Sengupta DN (1998) Expression of abscisic acid-responsive element-binding protein in salt-tolerant indica rice (Oryza sativa L. cv. Pokkali). Plant Mol Biol 37:629–637
Gupta K, Gupta B, Ghosh B, Sengupta DN (2012) Spermidine and abscisic acid-mediated phosphorylation of a cytoplasmic protein from rice root in response to salinity stress. Acta Physiol Plant 34:29–40. doi:10.1007/s11738-011-0802-0
Hussain SS, Ali M, Ahmad M, Siddique KH (2011) Polyamines: natural and engineered abiotic and biotic stress tolerance in plants. Biotechnol Adv 29(3):300–311
Igarashi K, Kashiwagi K (2000) Polyamines: mysterious modulators of cellular functions. Biochem Biophys Res Commun 271:559–564
Jakoby M, Weisshaar B, Droge-Laser W, Vicente-Carbajosa J, Tiedemann J, Kroj T, Parcy F, bZIP Research Group (2002) bZIP transcription factors in Arabidopsis. Trends Plant Sci 7:106–111
Janicka-Russak M, Kabała K, Młodzińska E, Kłobus G (2010) The role of polyamines in the regulation of the plasma membrane and the tonoplast proton pumps under salt stress. J Plant Physiol 167(4):261–269
Jiménez-Bremont JF, Ruiz OA, Rodríguez-Kessler M (2007) Modulation of spermidine and spermine levels in maize seedlings subjected to long-term salt stress. Plant Physiol Biochem 45(10–11):812–821
Kelner A, Pękala I, Kaczanowski S, Muszyńska G, Hardie DG, Dobrowolska G (2004) Biochemical characterization of the tobacco 42-kD protein kinase activated by osmotic stress. Plant Physiol 136:3255–3265
Kobayashi Y, Yamamoto S, Minami H, Kagaya Y, Hattori T (2004) Differential activation of the rice sucrose nonfermenting 1-related protein kinase 2 family by hyperosmotic stress and abscisic acid. Plant Cell 16:1163–1177
Koenig H, Goldstone A, Lu CY (1983) PAs regulate calcium fluxes in a rapid membrane response. Nature 305:530–534
Kovács Z, Simon-Sarkadi L, Szucs A, Kocsy G (2010) Differential effects of cold, osmotic stress and abscisic acid on polyamine accumulation in wheat. Amino Acids 38:623–631
Krishnamurthy R, Bhagwat KA (1989) Polyamines as modulator of salt tolerance in rice cultivars. Plant Physiol 91:500–504
Kuehn GD, Affolter HU, Atmar VJ, Seebeck T, Gubler U, Braun R (1979) Polyamine-mediated phosphorylation of a nucleolar protein from Physarum polycephalum that stimulates ribosomal-RNA synthesis. Proc Natl Acad Sci 76:2541–2545
Lee TM, Lur HS, Chu C (1997) Role of abscisic acid in chilling tolerance of rice (Oryza sativa L.) seedlings. 2. Modulation of free polyamine levels. Plant Sci 126:1–10
Liu J, Jiang MY, Zhou YF, Liu YL (2005) Production of polyamines is enhanced by endogenous abscisic acid in maize seedlings subjected to salt stress. J Integr Plant Biol 47:1326–1334
Liu J-H, Kitashiba H, Wang J, Ban Y, Moriguchi T (2007) Polyamines and their ability to provide environmental stress tolerance to plants. Plant Biotechnol 24:117–126
Marcotte WR Jr, Russell SH, Quatrano RS (1989) Abscisic acid-responsive sequences from the Em gene of wheat. Plant Cell 1:969–976
Mikoajczyk M, Awotunde OS, Muszyska G, Klessig DF, Dobrowolska G (2000) Osmotic stress induces rapid activation of a salicylic acid-induced protein kinase and a homolog of protein kinase ASK1 in tobacco cells. Plant Cell 12:165–178
Mukherjee K, Roy Choudhury A, Gupta B, Gupta S, Sengupta DN (2006) An ABRE-binding factor, OSBZ8, is highly expressed in salt tolerant cultivars than in salt sensitive cultivars of indica rice. BMC Plant Biol 6:18
Mundy J, Yamaguchi-Shinozaki K, Chua N-H (1990) Nuclear proteins bind conserved elements in the abscisic acid-responsive promoter of a rice rab gene. Proc Natl Acad Sci USA 87:1406–1410
Nakagawa H, Ohmiya K, Hattori T (1996) A rice bZIP protein, designated OSBZ8, is rapidly induced by abscisic acid. Plant J 9(2):217–227
Nieves N, Martinez ME, Castillo R, Blanco MA, Gonzalez-Olmedo JL (2001) Effect of abscisic acid and jasmonic acid on partial desiccation of encapsulated somatic embryos of sugarcane. Plant Cell Tissue Organ Cult 65:15–21
Ohmura Y, Uchida T, Teraoka H, Tsukada K (1987) Detection in situ of active polypeptides of mammalian and T4 DNA kinases after sodium dodecyl sulfate/polyacrylamide gel electrophoresis. Eur J Biochem 162:15–18
Quinet M, Ndayiragije A, Lefe`vre I, Lambillotte B, Dupont-Gillain CC, Stanley L (2010) Putrescine differently influences the effect of salt stress on polyamine metabolism and ethylene synthesis in rice cultivars differing in salt resistance. J Exp Bot 61(10):2719–2733
Roy P, Niyogi K, Sengupta DN, Ghosh B (2005) Spermidine treatment to rice seedlings recovers salinity stress induced damage of Plasma membrane and PM-bound H+-ATPase in salt-tolerant and salt sensitive rice cultivars. Plant Sci 168:583–591
RoyChoudhury A, Gupta B, Sengupta DN (2008) Trans-acting factor designated OSBZ8 interacts with both typical abscisic acid responsive elements as well as abscisic acid responsive element like sequences in the vegetative tissues of indica rice cultivars. Plant Cell Rep 27:779–794
Roychoudhury A, Basu S, Sengupta DN (2011) Amelioration of salinity stress by exogenously applied spermidine or spermine in three varieties of indica rice differing in their level of salt tolerance. J Plant Physiol 168:317–328
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, vol 1, 2 and 3, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schuber F (1989) Influence of PAs on membrane functions. Biochem J 260:1–10
Serafini-Fracassini D, Del Duca S, Beninati S (1995) Plant transglutaminases. Phytochemistry 40:355–365
Shinozaki K, Yamaguchi-Shinozaki K (1997) Gene expression and signal transduction in water-stress response. Plant Physiol 115:327–334
Vinocur B, Altman A (2005) Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Curr Opin Biotechnol 16:1–10
Vlad F, Benjamin ET, Philippe P, Jeffrey L, Sylvain M (2008) A versatile strategy to define the phosphorylation preferences of plant protein kinases and screen for putative substrates. Plant J 55:104–117
Zhang L, Lu YT (2003) Calmodulin-binding protein kinases in plants. Trends Plant Sci 8:123–127
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Acknowledgments
BG gratefully acknowledges the award of the JRF-ship and SRF-ship from Council of Scientific and Industrial Research [F. No. 9/15(263)/2002-EMR-1]. Both BG and KG acknowledge the support of technical facilities available at Presidency University and Bethune College (Govt. of West Bengal, India), respectively. Financial assistance from UGC (Govt. of India) [F. PSW-071/09-10(ERO) and F. PSW-042/10-11(ERO)] to BG and KG, respectively, are also gratefully acknowledged.
Authors’ contribution
BG performed the cloning, bacterial over-expression and purification of full length OSBZ8 and OSPDK cDNAs, SQ RT-PCR of OSPDK, all gel shift experiments, and also participated in activity and in vitro phosphoryation assays. KG designed and performed all the phosphorylation assays, performed necessary bioinformatics and statistical analysis and assisted BG in cloning and expression analysis. DNSG assisted in design of experiment, coordinated the study and procured research fund. BG and KG, both contributed equally in drafting the manuscript in its final format. All authors read and approved the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by R. Aroca.
B. Gupta and K. Gupta contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gupta, B., Gupta, K. & Sengupta, D.N. Spermidine-mediated in vitro phosphorylation of transcriptional regulator OSBZ8 by SNF1-type serine/threonine protein kinase SAPK4 homolog in indica rice. Acta Physiol Plant 34, 1321–1336 (2012). https://doi.org/10.1007/s11738-012-0929-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-012-0929-7