Abstract
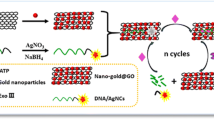
In the present work, electrochemical aptasensor for earlier detection of Hepatitis B virus infection was developed. First, the surface of a glassy carbon electrode was modified with gold nanoparticles functionalized reduced graphene oxide, then thiol-terminated aptamer specifically recognizes (HBsAg) was covalently linked to Nano-platform via strong gold—sulfur affinity. Subsequently, an electrochemical indicator Methylene Blue (MB) was intercalated into the aptamer structure through electrostatic interaction between MB and guanine bases. The function of the aptasensor relies on the specific binding between the aptamer and (HBsAg). In the absence of HBsAg, a strong electrochemical signal was observed. If the HBsAg was added, The MB- intercalated aptamer can be forced to release of MB from the sensing interface after HBsAg triggered structure switching of the aptamer. Hence, the electrochemical signal of cyclic voltammetry was linearly declined with the concentration of HBsAg over a range of 0.125–2.0 fg/mL with a limit of detection (at 3σ/slope) equivalent to 0.0014 fg/mL. To the best of our knowledge the developed aptasensor recount one of the lowest limit of detection for determination HBsAg recently reported in the literature. The selectivity of this electrochemical aptasensor was investigated, the peak current was slightly decreased even then the high concentration of interfering reagents was added. The aptasensor was applied for determining HBsAg in spiked human serum samples. The method was ultrasensitive, highly selective, good stability, and a wide analytical performance range.
















Similar content being viewed by others
References
Akkapinyo C, Khownarumit P, Waraho-Zhmayev D, Poo-arporn RP (2020) Development of a multiplex immunochromatographic strip test and ultrasensitive electrochemical immunosensor for hepatitis B virus screening. Anal Chim Acta 1095:162–171. https://doi.org/10.1016/j.aca.2019.10.016
Akter R, Rhee CK, Rahman MA (2015) A highly sensitive quartz crystal microbalance immunosensor based on magnetic bead-supported bienzymes catalyzed mass enhancement strategy. Biosens Bioelectron 66:539–546. https://doi.org/10.1016/j.bios.2014.12.007
Al-Sadeq DW, Taleb SA, Zaied RE, Fahad SM, Smatti MK, Rizeq BR, Al Thani AA, Yassine HM, Nasrallah GK (2019) Hepatitis B virus molecular epidemiology, host-virus interaction, coinfection, and laboratory diagnosis in the MENA region: an update. Pathogens. https://doi.org/10.3390/pathogens8020063
Argoubi W, Sánchez A, Parrado C, Raouafi N, Villalonga R (2018) Label-free electrochemical aptasensing platform based on mesoporous silica thin film for the detection of prostate specific antigen. Sens Actuators B Chem 255:309–315. https://doi.org/10.1016/j.snb.2017.08.045
Baghbaderani SS, Noorbakhsh A (2019) Novel chitosan-Nafion composite for fabrication of highly sensitive impedimetric and colorimetric As(III) aptasensor. Biosens Bioelectron 131(1):1–8. https://doi.org/10.1016/j.bios.2019.01.059
Bang GS, Cho S, Kim BG (2005) A novel electrochemical detection method for aptamer biosensors. Biosens Bioelectron 21(6):863–870. https://doi.org/10.1016/j.bios.2005.02.002
Beheshti-Marnani A, Hatefi-Mehrjardi A, Es’haghi Z (2019) A sensitive biosensing method for detecting of ultra-trace amounts of AFB1 based on “Aptamer/reduced graphene oxide” nano-bio interaction. Colloids Surf B 175:98–105. https://doi.org/10.1016/j.colsurfb.2018.11.087
Cabral DGA, Lima ECS, Moura P, Dutra RF (2016) A label-free electrochemical immunosensor for hepatitis B based on hyaluronic acid-carbon nanotube hybrid film. Talanta 148:209–215. https://doi.org/10.1016/j.talanta.2015.10.083
Choi YH, Lee GY, Ko H, Chang YW, Kang MJ, Pyun JC (2014) Development of SPR biosensor for the detection of human hepatitis B virus using plasma-treated parylene-N film. Biosens Bioelectron 56:286–294. https://doi.org/10.1016/j.bios.2014.01.035
Crulhas BP, Basso CR, Parra JPRLL, Castro GR, Pedrosa VA (2019) Reduced graphene oxide decorated with AuNPs as a new aptamer-based biosensor for the detection of androgen receptor from Prostate Cells. J Sens. https://doi.org/10.1155/2019/5805609
Danzer K, Currie LA (1998) Guideline for calibration in analytical chemistry—Part 1. Fundamentals and single component calibration. Pure Appl Chem 70(4):993–1014. https://doi.org/10.1351/pac199870040993
Deng B, Lin Y, Wang C, Li F, Wang Z, Zhang H, Li XF, Le XC (2014) Aptamer binding assays for proteins: the thrombin example-A review. Anal Chim Acta 837:1–15. https://doi.org/10.1016/j.aca.2014.04.055
Dinshaw IJ, Muniandy S, Teh SJ, Ibrahim F, Leo BF, Thong KL (2017) Development of an aptasensor using reduced graphene oxide chitosan complex to detect Salmonella. J Electroanal Chem 806(May):88–96. https://doi.org/10.1016/j.jelechem.2017.10.054
Ehsani M, Chaichi MJ, Nezammeddin Hosseini S (2017) Comparison of CuO nanoparticle and CuO/MWCNT nanocomposite for amplification of chemiluminescence immunoassay for detection of the hepatitis B surface antigen in biological samples. Sens Actuators B Chem 247:319–328. https://doi.org/10.1016/j.snb.2017.02.019
Ensafi AA, Akbarian F, Heydari-Soureshjani E, Rezaei B (2018) A novel aptasensor based on 3D-reduced graphene oxide modified gold nanoparticles for determination of arsenite. Biosen Bioelectron 122:25–31. https://doi.org/10.1016/j.bios.2018.09.034
Fraser LA, Kinghorn AB, Tang MSL, Cheung YW, Lim B, Liang S, Dirkzwager RM, Tanner JA, Miller AOA (2015) Oligonucleotide functionalised microbeads: indispensable tools for high-throughput aptamer selection. Molecules 20(12):21298–21312. https://doi.org/10.3390/molecules201219766
Gan L, Li B, Chen Y, Yu B, Chen Z (2018) Green synthesis of reduced graphene oxide using bagasse and its application in dye removal: a waste-to resource supply chain. Chemosphere. https://doi.org/10.1016/j.chemosphere.2018.11.181
Georgitsopoulou S, Petrai O, Georgakilas V (2019) Highly conductive functionalized reduced graphene oxide. Surf Interfaces 16:152–156. https://doi.org/10.1016/j.surfin.2019.05.010
Govindhan M, Amiri M, Chen A (2015) Au nanoparticle/graphene nanocomposite as a platform for the sensitive detection of NADH in human urine. Biosens Bioelectron 66:474–480. https://doi.org/10.1016/j.bios.2014.12.012
Gurunathan S, Han JW, Kim ES, Park JH, Kim JH (2015) Reduction of graphene oxide by resveratrol: a novel and simple biological method for the synthesis of an effective anticancer nanotherapeutic molecule. Int J Nanomed 10:2951–2969. https://doi.org/10.2147/IJN.S79879
Haghshenas E, Madrakian T, Afkhami A, Saify Nabiabad H (2019) An electrochemical ceruloplasmin aptasensor using a glassy carbon electrode modified by diazonium-functionalized multiwalled carbon nanotubes. J Iran Chem Soc 16(3):593–602. https://doi.org/10.1007/s13738-018-1533-6
Hayat A, Marty JL (2014) Aptamer based electrochemical sensors for emerging environmental pollutants. Front Chem 2(JUN):1–9. https://doi.org/10.3389/fchem.2014.00041
Ho JKT, Jeevan-Raj B, Netter HJ (2020) Hepatitis B virus (HBV) subviral particles as protective vaccines and vaccine platforms. Viruses 12(2):1–26. https://doi.org/10.3390/v12020126
Hong P, Li W, Li J (2012) Applications of aptasensors in clinical diagnostics. Sensors 12(2):1181–1193. https://doi.org/10.3390/s120201181
Hu D, Hu Y, Zhan T, Zheng Y, Ran P, Liu X, Guo Z, Wei W, Wang S (2020) Coenzyme A-aptamer-facilitated label-free electrochemical stripping strategy for sensitive detection of histone acetyltransferase activity. Biosens Bioelectron 150:111934. https://doi.org/10.1016/j.bios.2019.111934
Javed SI, Hussain Z (2015) Covalently functionalized graphene oxide - characterization and its electrochemical performance. Int J Electrochem Sci 10(11):9475–9487
Jeong HK, Jin MH, So KP, Lim SC, Lee YH (2009) Tailoring the characteristics of graphite oxides by different oxidation times. J Phys D: Appl Phys. https://doi.org/10.1088/0022-3727/42/6/065418
Joshi SR, Sharma A, Kim GH, Jang J (2020) Low cost synthesis of reduced graphene oxide using biopolymer for influenza virus sensor. Mater Sci Eng C 108:110465. https://doi.org/10.1016/j.msec.2019.110465
Kim SH (2017) ELISA for quantitative determination of hepatitis b virus surface antigen. Immune Netw 17(6):451–459. https://doi.org/10.4110/in.2017.17.6.451
Ku TH, Zhang T, Luo H, Yen TM, Chen PW, Han Y, Lo YH (2015) Nucleic acid aptamers: an emerging tool for biotechnology and biomedical sensing. Sensors 15(7):16281–16313. https://doi.org/10.3390/s150716281
Latthaphasavang V, Vanhems P, Ngo-Giang-Huong N, Sibounlang P, Paboriboune P, Malato L, Keoluangkhot V, Thammasack S, Salvadori N, Khamduang W, Steenkeste N, Trépo C, Dény P, Jourdain G (2019) Perinatal hepatitis B virus transmission in Lao PDR: a prospective cohort study. PLoS ONE 14(4):1–14. https://doi.org/10.1371/journal.pone.0215011
Lavanchy D (2005) Worldwide epidemiology of HBV infection, disease burden, and vaccine prevention. J Clin Virol. https://doi.org/10.1016/S1386-6532(05)00384-7
Li Y, Tian L, Liu L et al (2016a) An ultrasensitive sandwich-type electrochemical immunosensor based on δ-MnO2and palladium nanoparticles covered natural halloysite nanotubes for the detection of hepatitis B surface antigen. New J Chem 40(1):558–563. https://doi.org/10.1039/c5nj01251a
Li X, Wu X, Shi P, Ye ZG (2016b) Lead-Free piezoelectric diaphragm biosensors based on micro-machining technology and chemical solution deposition. Sensors (Switzerland) 16(1):1–10. https://doi.org/10.3390/s16010069
Liu YY, Liang XS (2018) Progression and status of antiviral monitoring in patients with chronic hepatitis B: from HBsAg to HBV RNA. World Hepatol 10(9):603–611. https://doi.org/10.4254/WJH.V10.I9.603
Mansouri R, Azadbakht A (2019) Aptamer-based approach as potential tools for construction the electrochemical aptasensor. J Inorg Organomet Polym Mater 29(2):517–527. https://doi.org/10.1007/s10904-018-1024-3
Mast EE, Alter MJ, Margolis HS (1999) Strategies to prevent and control hepatitis B and C virus infections: a global perspective. Vaccine 17(13–14):1730–1733. https://doi.org/10.1016/S0264-410X(98)00415-0
McMahon BJ (2009) The influence of hepatitis B virus genotype and subgenotype on the natural history of chronic hepatitis B. Hepatol Int 3(2):334–342. https://doi.org/10.1007/s12072-008-9112-z
Mirian M, Khanahmad H, Darzi L, Salehi M, Sadeghi-Aliabadi H (2017) Oligonucleotide aptamers: potential novel molecules against viral hepatitis. Res Pharm Sci 12(2):88–98. https://doi.org/10.4103/1735-5362.202447
Mohanraj J, Durgalakshmi D, Rakkesh RA, Balakumar S, Rajendran S, Karimi-Maleh H (2020) Facile synthesis of paper based graphene electrodes for point of care devices: a double stranded DNA (dsDNA) biosensor. J Colloid Interface Sci 566:463–472. https://doi.org/10.1016/j.jcis.2020.01.089
Niu X, Li X, Chen W, Li X, Weng W, Yin C, Dong R, Sun W, Li G (2018) Three-dimensional reduced graphene oxide aerogel modified electrode for the sensitive quercetin sensing and its application. Mater Sci Eng C. https://doi.org/10.1016/j.msec.2018.04.015
Otari SV, Kumar M, Anwar MZ, Thorat ND, Patel SKS, Lee D, Lee JH, Lee JK, Kang YC, Zhang L (2017) Rapid synthesis and decoration of reduced graphene oxide with gold nanoparticles by thermostable peptides for memory device and photothermal applications. Sci Rep. https://doi.org/10.1038/s41598-017-10777-1
Peiffer KH, Kuhnhenn L, Jiang B, Mondorf A, Vermehren J, Knop V, Susser S, Walter D, Dietz J, Carra G, Finkelmeier F, Zeuzem S, Sarrazin C, Hildt E (2018) Divergent preS sequences in virion-associated hepatitis B virus genomes and subviral HBV surface antigen particles from HBV e antigen-negative patients. J Infect Dis 218(1):114–123. https://doi.org/10.1093/infdis/jiy119
Poorali MS, Bagheri-Mohagheghi MM (2016) Comparison of chemical and physical reduction methods to prepare layered graphene by graphene oxide: optimization of the structural properties and tuning of energy band gap. J Mater Sci: Mater Electron 27(1):260–271. https://doi.org/10.1007/s10854-015-3749-x
Rabchinskii MK, Dideikin AT, Kirilenko DA, Baidakova MV, Shnitov VV, Roth F, Konyakhin SV, Besedina NA, Pavlov SI, Kuricyn RA, Lebedeva NM, Brunkov PN, Vul’ AY (2018) Facile reduction of graphene oxide suspensions and films using glass wafers. Sci Rep. https://doi.org/10.1038/s41598-018-32488-x
Razavipanah I, Rounaghi GH, Deiminiat B, Damirchi S, Abnous K, Izadyar M, Khavani M (2019) A new electrochemical aptasensor based on MWCNT-SiO 2 @Au core-shell nanocomposite for ultrasensitive detection of bisphenol A. Microchem J 146:1054–1063. https://doi.org/10.1016/j.microc.2019.02.018
Rezaei B, Jamei HR, Ensafi AA (2018) An ultrasensitive and selective electrochemical aptasensor based on rGO-MWCNTs/Chitosan/carbon quantum dot for the detection of lysozyme. Biosens Bioelectron 115(April):37–44. https://doi.org/10.1016/j.bios.2018.05.012
Rozenblum GT, Pollitzer IG, Radrizzani M (2019) Challenges in electrochemical aptasensors and current sensing architectures using flat gold surfaces. Chemosensors. https://doi.org/10.3390/chemosensors7040057
Saberian-Borujeni M, Johari-Ahar M, Hamzeiy H, Barar J, Omidi Y (2014) Nanoscaled aptasensors for multi-analyte sensing. BioImpacts 4(4):205–215. https://doi.org/10.15171/bi.2014.015
Samie HA, Arvand M (2020) Bioelectrochemistry label-free electrochemical aptasensor for progesterone detection in biological fluids. Bioelectrochemistry 133:107489. https://doi.org/10.1016/j.bioelechem.2020.107489
Seeger C, Mason WS (2015) Molecular biology of hepatitis B virus infection. Virology 479–480:672–686. https://doi.org/10.1016/j.virol.2015.02.031
Shrivastava A, Gupta V (2011) Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chron Young Sci 2(1):21. https://doi.org/10.4103/2229-5186.79345
Tian L, Liu L, Li Y, Feng X, Wei Q, Cao W (2016) A novel label-free electrochemical immunosensor for the detection of hepatitis B surface antigen. Anal Methods 8(40):7380–7386. https://doi.org/10.1039/c6ay01959e
Tiwari YK, Pundir S, Saraf G, Pawan K, Dashora D, Pokra M, Patidar V, Rana J, Mishra RK (2017) A Comparison of rapid card test with enzyme-linked immunosorbent assay for the detection of hepatitis B surface antigen [HBsAg] in tertiary care hospital. Result Rev J Microbiol Virol 7(3):27–31
Torabi R, Bagherzadeh K, Ghourchian H, Amanlou M (2016) An investigation on the interaction modes of a single-strand DNA aptamer and RBP4 protein: a molecular dynamic simulations approach. Org Biomol Chem 14(34):8141–8153. https://doi.org/10.1039/c6ob01094f
Torresi J, Tran BM, Christiansen D, Earnest-Silveira L, Schwab RHM, Vincan E (2019) HBV-related hepatocarcinogenesis: the role of signalling pathways and innovative ex vivo research models. BMC Cancer 19(1):707. https://doi.org/10.1186/s12885-019-5916-6
Trépo C, Chan HLY, Lok A (2014) Hepatitis B virus infection. Lancet 384(9959):2053–2063. https://doi.org/10.1016/S0140-6736(14)60220-8
Tsai HY, Chan JR, Li YC, Cheng FC, Fuh CB (2010) Determination of hepatitis B surface antigen using magnetic immunoassays in a thin channel. Biosens Bioelectron 25(12):2701–2705. https://doi.org/10.1016/j.bios.2010.04.035
Turcheniuk K, Boukherroub R, Szunerits S (2015) Gold-graphene nanocomposites for sensing and biomedical applications. J Mater Chem B 3(21):4301–4324. https://doi.org/10.1039/c5tb00511f
Tyner T, Francis J (2017) Potassium Ferricyanide. ACS Reagent Chem. https://doi.org/10.1021/acsreagents.4287.20160601
Velasco-Garcia M, Missailidis S, elasco-Garcia M, Missailidis S (2009) New trends in aptamer-based electrochemical biosensors. Gene Ther Mol Biol 13:1–10
Wang S, Li L, Jin H et al (2013) Electrochemical detection of hepatitis B and papilloma virus DNAs using SWCNT array coated with gold nanoparticles. BiosensBioelectron 41(1):205–210. https://doi.org/10.1016/j.bios.2012.08.021
Wiederoder MS, Misri I, DeVoe DL (2016) Impedimetric immunosensing in a porous volumetric microfluidic detector. Sens Actuators B: Chem 234:493–497. https://doi.org/10.1016/j.snb.2016.05.015
Wu Y, Guo W, Peng W, Zhao Q, Piao J, Zhang B, Wu X, Wang H, Gong X, Chang J (2017) Enhanced fluorescence ELISA based on HAT triggering fluorescence “Turn-on” with enzyme-antibody dual labeled AuNP probes for ultrasensitive detection of AFP and HBsAg. ACS Appl Mater Interfaces 9(11):9369–9377. https://doi.org/10.1021/acsami.6b16236
Wu CC, Chen YS, Cao L, Chen XW, Lu MJ (2018) Hepatitis B virus infection: defective surface antigen expression and pathogenesis. World J Gastroenterol 24(31):3488–3499. https://doi.org/10.3748/wjg.v24.i31.3488
Xi Z, Huang R, Li Z, He N, Wang T, Su E, Deng Y (2015) Selection of HBsAg-specific DNA aptamers based on carboxylated magnetic nanoparticles and their application in the rapid and simple detection of hepatitis b virus infection. ACS Appl Mater Interfaces 7(21):11215–11223. https://doi.org/10.1021/acsami.5b01180
Xi Z, Gong Q, Wang C, Zheng B (2018) Highly sensitive chemiluminescent aptasensor for detecting HBV infection based on rapid magnetic separation and double-functionalized gold nanoparticles. Sci Rep 8(1):1–7. https://doi.org/10.1038/s41598-018-27792-5
Xia N, Wang X, Zhou B, Wu Y, Mao W, Liu L (2016) Electrochemical detection of amyloid-β oligomers based on the signal amplification of a network of silver nanoparticles. ACS Appl Mater Interfaces 8(30):19303–19311. https://doi.org/10.1021/acsami.6b05423
Xiang D, Shigdar S, Qiao G, Wang T, Kouzani AZ, Zhou SF, Kong L, Li Y, Pu C, Duan W (2015) Nucleic acid aptamer-guided cancer therapeutics and diagnostics: the next generation of cancer medicine. Theranostics 5(1):23–42. https://doi.org/10.7150/thno.10202
Yao CY, Fu WL (2014) Biosensors for hepatitis B virus detection. World J Gastroenterol 20(35):12485–12492. https://doi.org/10.3748/wjg.v20.i35.12485
Yildiz UH, Inci F, Wang SQ, Toy M, Tekin HC, Javaid A, Lau DTY, Demirci U (2015) Recent advances in micro/nanotechnologies for global control of hepatitis B infection. Biotechnol Adv 33(1):178–190. https://doi.org/10.1016/j.biotechadv.2014.11.003
Yuen MF, Chen DS, Dusheiko GM, Janssen HLA, Lau DTY, Locarnini SA, Peters MG, Lai CL (2018) Hepatitis B virus infection. Nature Rev Dis Prim. https://doi.org/10.1038/nrdp.2018.35
Zejli H, Goud KY, Marty JL (2019) An electrochemical aptasensor based on polythiophene-3-carboxylic acid assisted methylene blue for aflatoxin B1 detection. Sens Bio-Sens Res 25:100290. https://doi.org/10.1016/j.sbsr.2019.100290
Zhang P, Lu H, Chen J, Han H, Ma W (2014) Simple and sensitive detection of HBsAg by using a quantum dots nanobeads based dot-blot immunoassay. Theranostics 4(3):307–315. https://doi.org/10.7150/thno.8007
Zhu C, Guo S, Fang Y, Dong S (2010) Reducing sugar: New functional molecules for the green synthesis of graphene nanosheets. ACS Nano 4(4):2429–2437. https://doi.org/10.1021/nn1002387
Zhu C, Yang G, Li H, Du D, Lin Y (2015) Electrochemical sensors and biosensors based on nanomaterials and nanostructures. Anal Chem 87(1):230–249. https://doi.org/10.1021/ac5039863
Zhu C, Liu M, Li X, Zhang X, Chen J (2018) A new electrochemical aptasensor for sensitive assay of a protein based on the dual-signaling electrochemical ratiometric method and DNA walker strategy. Chem Commun 54(73):10359–10362. https://doi.org/10.1039/C8CC05829F
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohsin, D.H., Mashkour, M.S. & Fatemi, F. Design of aptamer-based sensing platform using gold nanoparticles functionalized reduced graphene oxide for ultrasensitive detection of Hepatitis B virus. Chem. Pap. 75, 279–295 (2021). https://doi.org/10.1007/s11696-020-01292-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01292-1