Abstract
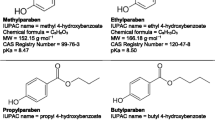
In this paper, ultraviolet radiation combined with hydrogen peroxide is presented to investigate the degradation of methyl, ethyl, and propyl parabens. The proposed kinetic model incorporates photochemical, chemical reactions, and reaction-rate constants to formulate the overall kinetic rate expressions, which are integrated into MATLAB. Thus, the chemical actinometry has been investigated to identify the incident ultraviolet-light intensity (1.59 × 10−6 Einstein.cm−1 s−1) and the optical path-length (5.83 cm) of the new reactor. Based on the optimal parameters of the previous study, an experimental campaign was performed at fixed hydrogen peroxide concentration to compare the half-life of the decomposition of these parabens and to determine the evolution of the pH and the pseudo-first-order rate constant. Accordingly, the study provides a comparison between numerical results and experimental data. The use of hydrogen peroxide at 25 µM resulted in 80% removal of propyl paraben (10 mg/L) within 90 min reaction time, while 50% of methyl and ethyl was oxidized. The performed model had a satisfactory predictive performance of paraben removal during the reaction, and the numerical pseudo-first-order rate constant was in agreement with the experimental value. Although the proposed model slightly overestimates the decrease of experimental pH, it provides insight into the evolution of hydroxide, superoxide, and hydroperoxyl ions. Finally, this paper proposes a discussion on the limitations of the study and suggests future work concerning the effects of water matrix, carbonate species, paraben mixture, and cytotoxicity of degradation by-products.







Similar content being viewed by others
References
Álvarez MA, Ruidíaz-Martínez M, Cruz-Quesada G, López-Ramóna V, Rivera-Utrilla J, Sánchez-Polo M, Mota AJ (2019) Removal of parabens from water by UV-driven advanced oxidation processes. Chem Eng J 379:122334. https://doi.org/10.1016/j.cej.2019.122334
Ameur H (2016) 3D hydrodynamics involving multiple eccentric impellers in unbaffled cylindrical tank. Chin J Chem Eng 24(5):572–580. https://doi.org/10.1016/j.cjche.2015.12.010
Andersen FA (2008) Final amended report on the safety assessment of methylparaben, ethylparaben, propylparaben, isopropylparaben, butylparaben, isobutylparaben, and benzylparaben as used in cosmetic products. Int J Toxicol 27(suppl 4):1–82. https://doi.org/10.1080/10915810802548359
Beck G (1969) Detection of charged intermediate of pulse radiolysis by electrical conductivity measurements. Int J Radiat Phys Chem. 1(3):361–371. https://doi.org/10.1016/0020-7055(69)90033-3
Beltran FJ (2003) Ozone reaction kinetics for water and wastewater systems. CRC Press, Boca Raton, p 384 (ISBN 020350917X, 9780203509173)
Beltran FJ, Gonzalez M, Gonzalez JF (1997) Industrial wastewater advanced oxidation. Part 1. UV radiation in the presence and absence of hydrogen peroxide. War Res. 31:2405–2414. https://doi.org/10.1016/S0043-1354(97)00077-8
Bielski HJB, Cabelli DE, Ravindra LA (1985) Reactivity of HO2/O2− in aqueous solution. J Phys Chem Ref Data 14(4):1041–1100. https://doi.org/10.1063/1.555739
Błędzka D, Gmurek M, Gryglik M, Olak M, Miller JS, Ledakowicz S (2010a) Photodegradaton and advanced oxidation of endocrine disruptors in aqueous solutions. Catal Today 151:125–130. https://doi.org/10.1016/j.cattod.2010.03.040
Błędzka D, Gryglik D, Olak M, Gębicki JL, Miller JS (2010b) Degradation of n-butylparaben and 4-tert-octylphenol in H2O2/UV system. Radiat Phys Chem 79(4):409–416. https://doi.org/10.1016/j.radphyschem.2009.11.012
Błędzka D, Gromadzinska J, Wasowicz W (2014) Parabens, From environmental studies to human health. Environ Int 67:27–42. https://doi.org/10.1016/j.envint.2014.02.007
Buxton GV (1988) Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (.OH/.O−) in aqueous solution. J Phys Chem Ref Data 17(2):513–886. https://doi.org/10.1063/1.555805
Chinh VD, Broggi A, Di Palma L, Scarsella M, Speranza G, Vilardi G, Pham NT (2018) XPS spectra analysis of Ti2+, Ti3+ ions and dye photodegradation evaluation of titania–silica mixed oxide nanoparticles. J Elec Mater 47:2215–2224. https://doi.org/10.1007/s11664-017-6036-1
Christensen HS, Sehested K, Corftizan H (1982) Reaction of hydroxyl radicals with hydrogen peroxide at ambient temperatures. J Phys Chem 86:15–88. https://doi.org/10.1021/j100206a023
Crittenden JC, Hu S, Hand DW, Green SA (1999) A kinetic model for H2O2/UV process in a completely mixed batch reactor. Wat Res. 33:2315–2328. https://doi.org/10.1016/S0043-1354(98)00448-5
Darbre PD, Aljarrah A, Miller WR, Coldham NG, Sauer MJ, Pope GS (2004) Concentrations of parabens in human breast tumours. J Appl Toxicol 24:5–13. https://doi.org/10.1002/jat.958
Dickson AG (1984) pH Scales and proton-transfer reactions in saline media such as sea-water. Geochim Cosmochim Acta 48(11):2299–2308. https://doi.org/10.1016/0016-7037(84)90225-4
Domínguez JR, Muñoz MJ, Palo P, González T, Peres JA (2014) Cuerda-Correa EM (2014) Fenton advanced oxidation of emerging pollutants: parabens. Int J Energy Environ Eng 5:89. https://doi.org/10.1007/s40095-014-0089-1
Draganic ZD, Negron-Mendoza A, Sehested K, Vujosevic SI, Navarro-Gonzales R, Albarran-Sanchez MG, Draganic IG (1991) Radiolysis of aqueous solutions of ammonium bicarbonate over a large dose range. Int J Radiat Appl Instrum. 38(3):317–321. https://doi.org/10.1016/1359-0197(91)90100-G
El Seoud OA, Baader WJ, Bastos EL (2017) Encyclopedia of Physical Organic Chemistry, First Edition. Edited by Zerong Wang. John Wiley & Sons, Inc. ISBN 978-1-118-46858-6. https://doi.org/10.1002/9781118468586
Ferreira AMC, Möder M, Fernández Laespada ME (2011) GC–MS determination of parabens, triclosan and methyl triclosan in water by in situ derivatisation and stir-bar sorptive extraction. Anal Bioanal Chem 399:945–953. https://doi.org/10.1007/s00216-010-4339-7
Glaze WH, Kang JW, Chapin DH (1997) The chemistry of water treatment processes involving ozone, hydrogen peroxide and ultraviolet radiation. Ozone Sci Eng 9:335–352. https://doi.org/10.1080/01919518708552148
Gmurek M, Rossi AF, Martins RC, Quinta-Ferreira RM, Ledakowicz S (2015) Photodegradation of single and mixture of parabens—Kinetic, by-products identification and cost-efficiency analysis. Chem Eng J 276:303–314. https://doi.org/10.1016/j.cej.2015.04.093
Goldstein S, Aschengrau D, Diamant Y, Rabani J (2007) Photolysis of aqueous H2O2: quantum yield and applications for polychromatic UV actinometry in photoreactors. Environ Sci Technol 41:7486–7490. https://doi.org/10.1021/es071379t
González-Mariño I, Quintana JB, Rodríguez I, Cela R (2011) Evaluation of the occurrence and biodegradation of parabens and halogenated by-products in wastewater by accurate mass liquid chromatography quadrupole time-of-flight mass spectrometry (LC–QTOF-MS). Water Res 45:6770–6780. https://doi.org/10.1016/j.watres.2011.10.027
Gurol MD, Akata A (1996) Kinetics of Ozone Photolysis in Aqueous Solution. AIChE J 42(11):3283–3292. https://doi.org/10.1002/aic.690421128
Handa O, Kokura S, Adachi S, Takagi T, Naito Y, Tanigawa T, Yoshida N, Yoshikawa T (2006) Methylparaben potentiates UV-induced damage of skin keratinocytes. Toxicology 227:62–72. https://doi.org/10.1016/j.tox.2006.07.018
Hofmann AF, Meysman FJR, Soetaert K, Middelburg JJ (2008) A step-by-step procedure for pH model construction in aquatic systems. Biogeosciences 5:227–251. https://doi.org/10.5194/bg-5-227-2008
Honda M, Robinson M, Kannan K (2018) Parabens in human urine from several Asian countries, Greece, and the United States. Chemosphere 201:13–19. https://doi.org/10.1016/j.chemosphere.2018.02.165
Inamuddin, Asiri AM, Mohammad A (2018) Organic Pollutants in Wastewater I: Methods of Analysis, Removal and Treatment. Materials Research Foundations, Volume 29, 362 pages. https://doi.org/10.21741/9781945291630
Johnson MB (2007) Modelling the advanced oxidation of the pharmaceutical compound metronidazole in single- and multilamp tubular photoreactors. Theses and dissertations. Ryerson University. https://digital.library.ryerson.ca/islandora/object/RULA%3A763/datastream/OBJ/view
Kasprzyk-Hordern B, Dinsdale MR, Guwy JA (2008) Multiresidue methods for the analysis of pharmaceuticals, personal care products and illicit drugs in surface water and wastewater by solid-phase extraction and ultra performance liquid chromatography–electrospray tandem mass spectrometry. Anal Bioanal Chem 391:1293–1308. https://doi.org/10.1016/j.chemosphere.2018.02.165
Koppenol WH, Butler J, Van Leeuwen JWL (1978) The Haber-Weiss cycle. Photochem Photobiol 28:655–660. https://doi.org/10.1111/j.1751-1097.1978.tb06989.x
Krystynik P, Kluson P, Hejda S, Buzek D, Masin P, Tito DN (2014) Semi-pilot scale environment friendly photocatalytic degradation of 4-chlorophenol with singlet oxygen species—Direct comparison with H2O2/UV-C reaction system. Appl Catal B 160–161:506–513. https://doi.org/10.1016/j.apcatb.2014.05.051
Lin Y, Ferronato C, Deng N, Chovelon JM (2011) Study of benzylparaben photocatalytic degradation by TiO2. Appl Catal B Environ 104:353–360. https://doi.org/10.1016/j.apcatb.2011.03.006
Madsen T, Boyd HB, Nylén D, Pedersen AR, Petersen GI, Simonsen F (2001) Environmental and health assessment of substances in household detergents and cosmetic detergent products. Environmental project 615: 1–240. https://www2.mst.dk/udgiv/Publications/2001/87-7944-596-9/pdf/87-7944-597-7.pdf
Marco Stoller M, Azizova G, Mammadova A, Vilardi G, Di Palma G, Chianese A (2016) Treatment of Olive Oil Processing Wastewater by Ultrafiltration, Nanofiltration, Reverse Osmosis and Biofiltration. Chem Eng Trans 47:409–414. https://doi.org/10.3303/CET1647069
Montanaro D, Bavasso I, Di Palma L, Petrucci E (2017) Photolysis of in-situ electrogenerated hydrogen peroxide for the degradation of emerging pollutants. Chem Eng Trans 57:643–648. https://doi.org/10.3303/CET1757108
Nguyen VD, Pierens X, Benhabib K (2019) Experimental and numerical study of methylparaben decomposition in aqueous solution using the UV/H2O2 process. J Environ Sci Health B 54(5):357–365. https://doi.org/10.1080/03601234.2019.1571365
Oishi S (2002) Effects of propyl paraben on the male reproductive system. Food Chem Toxicol 40:1807–1813. https://doi.org/10.1016/S0278-6915(02)00204-1
Peng X, Yu Y, Tang C, Tan J, Huang Q, Wang Z (2008) Occurrence of steroid estrogens, endocrinedisrupting phenols, and acid pharmaceutical residues in urban riverine water of the Pearl River Delta, South China. Sci Total Environ 397:158–166. https://doi.org/10.1016/j.scitotenv.2008.02.059
Perry RH, Green DW, Maloney JD (2015) Chemical Engineers’ Handbook 7th edn. McGraw-Hill, New York. https://chembugs.files.wordpress.com/2015/12/perrys-chemical-engineering-handbook1.pdf
Petala A, Frontistis Z, Antonopoulou M, Konstantinou I, Kondarides DI, Mantzavinos D (2015) Kinetics of ethyl paraben degradation by simulated solar radiation in the presence of N-doped TiO2 catalysts. Water Res 81:157–166. https://doi.org/10.1016/j.watres.2015.05.056
Rastogi SC, Schouten A, De Kruijf N, Weijland JW (1995) Contents of methyl-, ethyl-, propyl-, butyl- and benzyl paraben in cosmetic products. Contact Dermatitis 32:28–30. https://doi.org/10.1111/j.1600-0536.1995.tb00836.x
Rizzo L, Meric S, Kassinos D, Guida M, Russo F, Belgiorno V (2009) Degradation of diclofenac by TiO2 photocatalysis: UV absorbance kinetics and process evaluation through a set of toxicity bioassays. Water Res 43(4):979–988. https://doi.org/10.1016/j.watres.2008.11.040
Rojas MR, Pérez F, Whitley D, Arnold RG, Sáez AE (2010) Modeling of advanced oxidation of trace organic contaminants by hydrogen peroxide photolysis and Fenton’s Reaction. Ind Eng Chem Res 49:11331–11343. https://doi.org/10.1021/ie101592p
Rosales E, Buftia G, Pazos M, Lazar G, Sanromán MA (2018) Highly active based iron-carbonaceous cathodes for heterogeneous electro-Fenton process: application to degradation of parabens. Process Saf Environ Prot 117:363–371. https://doi.org/10.1016/j.psep.2018.05.014
Sanchez M, Rivero MJ, Ortiz I (2011) Kinetics of dodecylbenzenesulphonate mineralisation by TiO2 photocatalysis. Appl Catal B 101:515–521. https://doi.org/10.1016/j.apcatb.2010.10.023
Sanchez-Martin J, Beltran-Heredia J, Dominguez JR (2013) Advanced photochemical degradation of emerging pollutants. J Water Air Soil Pollut 224:1483–1495. https://doi.org/10.1007/s11270-013-1483-7
Sehested K, Rasmussen OL, Fricke H (1968) Rate constants of OH with HO2, O2− and H2O2+ from hydrogen peroxide formation in pulse-irradiated oxygenated water. J Phys Chem 72(2):626–631. https://doi.org/10.1021/j100848a040
Song W, Ravindran V, Pirbazari M (2008) Process optimization using a kinetic model for the ultraviolet radiation-hydrogen peroxide decomposition of natural and synthetic organic compounds in groundwater. Chem Eng Sci 63:3249–3270. https://doi.org/10.1016/j.ces.2008.03.024
Soni MG, Carabin IG (2005) Assessment of esters of p-hydroxybenzoic acid (parabens). Chem. Toxicol. 43:985–1015. https://doi.org/10.1016/j.fct.2005.01.020
Tay KS, Rahman NA, Bin Abas MR (2010) Ozonation of parabens in aqueous solution: kinetics and mechanism of degradation. Chemosphere 81:1446–1453. https://doi.org/10.1016/j.chemosphere.2010.09.004
Ulliman S, Miklos D, Huebner U, Drewes J, Linden K (2018) Improving UV/H2O2 performance following tertiary treatment of municipal wastewater. Environ Sci 4:1321–1330. https://doi.org/10.1039/C8EW00233A
Vilardi G (2019) Mathematical modelling of simultaneous nitrate and dissolved oxygen reduction by Cu-nZVI using a bi-component shrinking core model. Powder Technol 343:613–618. https://doi.org/10.1016/j.powtec.2018.11.082
Vilardi G, Ochando-Pulido JM, Stoller M, Verdone N, Di Palma L (2018a) Fenton oxidation and chromium recovery from tannery wastewater by means of iron-based coated biomass as heterogeneous catalyst in fixed-bed columns. Chem Eng J 351:1–11. https://doi.org/10.1016/j.cej.2018.06.095
Vilardi G, Rodriguez-Rodriguez J, Ochando-Pulido JM, Verdone N, Martinez-Ferez A, Di Palma L (2018b) Large Laboratory-Plant application for the treatment of a Tannery wastewater by Fenton oxidation: Fe(II) and nZVI catalysts comparison and kinetic modelling. Process Saf Environ Prot 117:629–638. https://doi.org/10.1016/j.psep.2018.06.007
Weinstein J, Benon HJ, Bielski HJ (1979) Kinetics of the interaction of HO2 and O2− radicals with hydrogen peroxide The Haber–Weiss reaction. J Am Chem Soc 101(1):58–62. https://doi.org/10.1021/ja00495a010
Xiao Y, Zhang L, Yue J, Webster RD, Lim T (2015) Kinetic modeling and energy efficiency of UV/H2O2 treatment of iodinated trihalomethanes. Water Res 75:259–269. https://doi.org/10.1016/j.watres.2015.02.044
Yamaguchi Y, Shimodo T, Chikamori N, Usuki S, Kanai Y, Endo T, Katsumata K, Terashima C, Ikekita M, Fujishima A, Suzuki T, Sakai H, Nakata K (2016) Sporicidal performance induced by photocatalytic production of organic peroxide under visible light irradiation. Scientific Reports 6:33715. https://doi.org/10.1038/srep33715
Yue PL (1997) Oxidation reactors for water and wastewater treatment. Wat. Sci. Tech 35(4):189–196. https://doi.org/10.1016/S0273-1223(97)00025-5
Acknowledgements
We thank Dr. Thanh-Binh Nguyen (Institut de Chimie des Substances Naturelles, CNRS, Université Paris-Saclay, France) for reviewing of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pierens, X., Nguyen, VD., Lauzier, T. et al. Chemical actinometry and paraben decomposition in aqueous solution utilizing ultraviolet radiation combined with hydrogen peroxide. Chem. Pap. 74, 4283–4294 (2020). https://doi.org/10.1007/s11696-020-01237-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01237-8