Abstract
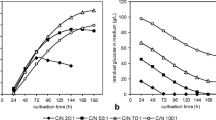
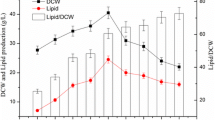
The oleaginous and red yeast Rhodotorula glutinis CCY 20-2-26 is known for its high-storage lipids accumulation in cells as well as for β-carotene biosynthesis. The work is focused on the study of lipogenesis and carotenoid pigments formation by the yeast grown in media with four different C/N ratios (20:1, 50:1, 70:1 and 100:1). Total fatty acids accumulation in cells reached the maximal value of 48% and yielded up to 8.9 g fatty acid/L media under C/N 70:1. On contrary, while the highest pigment accumulation in the yeast (1268 μg/g of cells) was found at the C/N ratio of 20:1, the maximal carotenoid yield (mainly β-carotene) of 12.7 mg/L was obtained under 50:1 ratio. Calculation of kinetic parameters of metabolites production revealed the metabolic correlations among glucose consumption, lipogenesis and carotenoid biosynthesis. It was demonstrated that glucose exhaustion resulted in reduction of growth and lipid accumulation in cells. Then, storage fatty acids, especially palmitic and oleic acids, begun to be degraded by β-oxidations and formed acetyl-CoA which was especially used for biosynthesis of β-carotene. This is the first study providing the new hypothesis about the metabolic connection between fatty acids and β-carotene metabolism in the red yeasts.








Similar content being viewed by others
References
Braunwald T, Schwemmlein L, Graeff-Hönninger S, French WT, Hernandez R, Holmes WE, Claupein W (2013) Effect of different C/N ratios on carotenoid and lipid production by Rhodotorula glutinis. Appl Microbiol Biotechnol 97:6581–6588. https://doi.org/10.1007/s00253-013-5005-8
Buzzini P, Goretti M, Branda E, Turchetti B (2010) Basidiomycetous yeast for production of carotenoids. In: Flickinger M (ed) Encyclopedia of industrial biotechnology: bioprocess, bioseparation, and cell technology. Wiley, New York, pp 469–481
Certik M, Shimizu S (2000) kinetic analysis of oil biosynthesis by an arachidonic acid-producing fungus, Mortierella alpina 1S-4. Appl Microbiol Biotechnol 54:224–230. https://doi.org/10.1007/s002530000361
Čertík M, Hanusová V, Breierová E, Márová I, Rapta P (2009) Biotechnological production and properties of carotenoid pigments. In: Hou CT, Shaw J-F (eds) Biocatalysis and agricultural biotechnology. Tailor and Francis-CRC Press, Boca Raton, pp 355–375
Deák T (2008) Handbook of food spoilage yeasts, 2nd edn. CRC Press, New York, pp 62–64
El-Banna AA, El-Razek AMA, El-Mahdy AR (2012) Some factors affecting the production of carotenoids by Rhodotorula glutinis var. glutinis. Food Nutr Sci 3:64–71. https://doi.org/10.4236/fns.2012.31011
Folch J, Lees M, Sloane-Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Gajdoš P, Nicaud JM, Rossignol T, Čertík M (2015) Single cell oil production on molasses by Yarrowia lipolytica strains overexpressing DGA2 in multicopy. Appl Microbiol Biotechnol 99:8065–8074. https://doi.org/10.1007/s00253-015-6733-8
Han M, Xu Z, Du Ch, Qian H, Zhang W (2016) Effects of nitrogen on the lipid and carotenoid accumulation of oleaginous yeast Sporidiobolus pararoseus. Bioprocess Biosyst Eng 39:1425–1433. https://doi.org/10.1007/s00449-016-1620-y
Harrison PJ, Bugg TDH (2014) Enzymology of the carotenoid cleavage dioxygenases: reaction mechanisms, inhibition and biochemical roles. Arch Biochem Biophys 544:105–111. https://doi.org/10.1016/j.abb.2013.10.005
Hernández-Almanza A, Montanez JC, Aguilar-González MA, Martínez-Ávila C, Rodríguez-Herrera R, Aguilar CN (2014) Rhodotorula glutinis as source of pigments and metabolites for food industry. Food Biosci 5:64–72. https://doi.org/10.1016/j.fbio.2013.11.007
Jaswir I, Noviendri D, Hasrini RF, Octavianti F (2011) Carotenoids: sources, medical properties and their application in food and nutraceutical industry. J Med Plants Res 5:7119–7131. https://doi.org/10.5897/JMPRx11.011
Klempova T, Basil E, Kubatova A, Certik M (2013) Biosynthesis of gamma-linolenic acid and beta-carotene by Zygomycetes fungi. Biotechnol J 8:794–800. https://doi.org/10.1002/biot.201200099
Li Y, Zhao ZK, Bai F (2007) High-density cultivation of oleaginous yeast Rhodosporidium toruloides y4 in fed-batch culture. Enzyme Microb Technol 41:312–317. https://doi.org/10.1016/j.enzmictec.2007.02.008
Lin J, Shen H, Tan H, Zhao X, Wu S, Hu C, Zhao ZK (2011) Lipid production by Lipomyces starkeyi cells in glucose solution without auxiliary nutrients. J Biotechnol 152:184–188. https://doi.org/10.1016/j.jbiotec.2011.02.010
Marova I, Breierova E, Koci R, Friedl Z, Slovak B, Pokorna J (2004) Influence of exogenous stress factors on production of carotenoids by some strains of carotenogenic yeasts. Ann Microbiol 54:73–85
Mata-Gómez LC, Montañez JC, Méndez-Zavala A, Aguilar CN (2014) Biotechnological production of carotenoids by yeasts: an overview. Microb Cell Fact 13:1–11. https://doi.org/10.1186/1475-2859-13-12
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Ratledge C (2004) Fatty acids biosynthesis in microorganisms being used for single cell oil production. Biochimie 86:807–815. https://doi.org/10.1016/j.biochi.2004.09.017
Rodrigues F, Ludovico P, Lea͂o C (2006) Sugar metabolism in yeasts: an overview of aerobic and anaerobic glucose catabolism. In: Gábor P, Carlos R (eds) Biodiversity and ecology of yeasts. The yeast handbook. Springer, Berlin, pp 101–121
Salar RK, Certik M, Brezova V, Brlejova M, Hanusova V, Breierová E (2013) Stress influenced increase in phenolic content and radical scavenging capacity of Rhodotorula glutinis CCY 20-2-26. 3. Biotech 3:53–60. https://doi.org/10.1007/s13205-012-0069-1
Schmidt I, Schewe H, Gassel S, Jin Ch, Buckingham J, Hümbelin M, Sandmann G, Schrader J (2011) Biotechnological production of astaxanthin with Phaffia rhodozyma/Xanthophyllomyces dendrorhous. Appl Microbiol Biotechnol 89:555–571. https://doi.org/10.1007/s00253-010-2976-6
Somashekar D, Joseph R (2000) Inverse relationship between carotenoid and lipid formation in Rhodotorula gracilis according to the C/N ratio of the growth medium. World J Microbiol Biotechnol 16:491–493. https://doi.org/10.1023/A:1008917612616
Stahl W, Sies H (2012) Photoprotection by dietary carotenoids: concept, mechanisms, evidence and future development. Mol Nutr Food Res 56:287–295. https://doi.org/10.1002/mnfr.201100232
Tang X, Chen WN (2014) Investigation of fatty acid accumulation in the engineered Saccharomyces cerevisiae under nitrogen limited culture condition. Bioresour Technol 162:200–206. https://doi.org/10.1016/j.biortech.2014.03.061
Tkáčová J, Čaplová J, Klempová T, Čertík M (2017) Correlation between lipid and carotenoid synthesis in torularhodin-producing Rhodotorula glutinis. Ann Microbiol 67:541–551. https://doi.org/10.1007/s13213-017-1284-0
Van Roermund CWT, Hettema EH, Kal AJ, van den Berg M, Tabak HF, Wanders RJA (1998) Peroxisomal β-oxidation of polyunsaturated fatty acids in Saccharomyces cerevisiae: isocitrate dehydrogenase provides NADPH for reduction of double bonds at even position. EMBO J 17:677–687. https://doi.org/10.1093/emboj/17.3.677
Viega A, Arrabaça DJ, Loureiro-Dias MC (2000) Cyanide-resistant respiration is frequent, but confined to yeasts incapable of aerobic fermentation. FEMS Microbiol Lett 190:93–97. https://doi.org/10.1111/j.1574-6968.2000.tb09268.x
Zhu LY, Zong MH, Wu H (2008) Efficient lipid production with Trichosporon fermentans and its use for biodiesel preparation. Bioresour Technol 99:7881–7885. https://doi.org/10.1016/j.biortech.2008.02.033
Acknowledgements
Authors acknowledge to Grant VEGA 1/0574/15 from Ministry of Education, Science, Research and Sport of the Slovak Republic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tkáčová, J., Klempová, T. & Čertík, M. Kinetic study of growth, lipid and carotenoid formation in β-carotene producing Rhodotorula glutinis . Chem. Pap. 72, 1193–1203 (2018). https://doi.org/10.1007/s11696-017-0368-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0368-4