Abstract
Background
Bariatric surgery promotes expressive weight loss, improving the metabolic and inflammatory profiles. The behavior of these indicators in bariatric patients with weight recidivism is unknown. We aimed to investigate both profiles in bariatric patients with high ratio of weight regain (RWR), comparing them with nonsurgical patients with obesity.
Methods
Forty patients with obesity subjected to Roux-en-Y gastric bypass (RYGB) with high RWR composed the bariatric group, and 40 controls matched for BMI, age, and gender were recruited as nonsurgical group. Between-group comparisons were performed for clinical history, physical examination, biochemical, metabolic, and inflammatory profiles.
Results
Bariatric group was composed of a group with an excess weight loss of 85.9 ± 16.8%, a RWR of 56.5 ± 19.7%, and a time since surgery of 10.7 ± 4.3 years. We noticed a lower proportion of patients with type 2 diabetes mellitus and dyslipidemia (P ≤ 0.05) and lower neck and waist circumferences (P ≤ 0.05) in this group. No differences between groups were observed concerning hip circumference, blood pressure, heart rate, total cholesterol, LDL-c, acid uric, creatinine, ALT, ASP, interferon-γ (INF-γ), interferon gamma–induced protein-10 (IP-10), monocyte chemoattractant protein-1 (MCP-1), TNF-α, interleukin-1β (IL-1 β), interleukin-17 (IL-17), and interleukin-10 (IL-10). Of note, fasting glucose; HbA1c; triglycerides; and, surprisingly, IL-6 levels were lower (P ≤ 0.05) in the bariatric group than nonsurgical one while HDL-c level was higher (P < 0.001).
Conclusion
Expressive post-bariatric weight loss, even in patients with high RWR, suggests a possible metabolic benefit/protection in the long term. Probably decreased circulating levels of IL-6 are involved in it.
Trial Registration
NCT04193397.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The current obesity epidemic is a tremendous public health concern worldwide [1]. Obesity is considered a severe chronic disease related to genetic, environmental, and behavioral factors [2]. It is associated with an increased risk of developing cardiovascular disease, type 2 diabetes mellitus (T2DM), obstructive sleep apnea, nonalcoholic fatty liver disease, dyslipidemia, and cancer [3, 4].
Bariatric surgery is the most effective way to treat severe obesity. However, it should be indicated only when other nonsurgical approaches, such as diet, physical activity, and pharmacological therapies, are ineffective [2, 5]. As its name already denotes, this type of surgery typically results in impressive weight loss, improving and or remitting obesity-related comorbidities, with increased life expectancy and quality [6,7,8].
Substantial weight loss is commonly observed during the first 2 years after surgery. Regardless of the opted surgical procedure, there is accumulating evidence showing weight regain after weight loss stabilizes [9,10,11] in a subgroup of patients. Weight regain is a multifactorial condition, which includes hormonal, behavioral, environmental, psychological, and surgical mechanisms [9]. The ratio of weight regain (RWR) may range from 5.7 to 75.6% during 2–6 years after surgery [12] which are possibly associated with recurrence of obesity-related comorbidities, including T2DM [13]. There is not yet an accepted universal cutoff point to define a clinically relevant weight regain [14]. It is possible to speculate that the magnitude of weight regain may be associated with the relapse of metabolic and cardiovascular diseases [15].
Obesity is a well-established cause of chronic low-grade systemic inflammation. Excessive adipose depots, especially centrally located, plays a crucial role in promoting inflammation due to adipose tissue-derived pro-inflammatory cytokines, chemokines, and adipokines [16]. Although bariatric surgery promotes reductions in circulating levels of inflammatory biomarkers [8], little is known about the inflammatory status of these patients with high RWR in the long term [8, 15]. Therefore, this study aims to compare the inflammatory status and the metabolic profile of post-bariatric patients with high RWR with nonsurgical patients with obesity.
Methods
Study Design and Participants
This cross-sectional study enrolled 274 volunteers previously subjected to bariatric surgery in private or public health care units who were without any clinical follow-up. Of those, 40 post-bariatric patients (bariatric group), aged 46 ± 8.7 years, with a body mass index (BMI) of 41.5 ± 11.0 kg/m2 were eligible to participate in the study. Recruitment was performed at our outpatient care unit at their first appointment on it. The nonsurgical group consisted of 40 BMI-, age-, and gender-matched controls who were already attending the clinic and were recruited through the analysis of their medical records. We included only patients who underwent Roux-en-Y gastric bypass (RYGB) surgery exclusively, with a mean weight loss after surgery of approximately 50% of initial body weight, who regained more than 40% of total weight loss, and who did not undergo any other type of procedure or revisional surgery. The exclusion criteria for both groups were pregnancy, smoking, alcoholism, level of physical activity ≥ 150 min/week, a recent history of a major cardiovascular event (acute myocardial infarction, or stroke), chronic kidney disease, heart failure, peripheral arterial disease, chronic liver disease (except for nonalcoholic fatty liver disease), chronic obstructive pulmonary disease, cancer, and HIV infection. All patients had no other infectious disease and were not using any medication with inflammatory or anti-inflammatory effects during or previous to recruitment. No one patient has confirmed COVID-19 infection or had it recently. Recruitment, screening, inclusion/exclusion visits, and data collection occurred between November 2020 and September 2021 at an outpatient’s care unit. During the inclusion visit, the clinical history, and physical, demographic, and anthropometric data were collected and recorded; blood pressure and heart rate were assessed; and also blood samples were collected for further analysis of biochemical, metabolic, and inflammatory profiles. Since these patients belong to a cohort of patients subjected to surgery out of our unit [17], the type of self-reported surgery was confirmed by digestive endoscopy. Figure 1 presents the flowchart of the enrollment, allocation, and follow-up of participants.
Anthropometric, Blood Pressure, and Heart Rate Assessments
An electronic scale and stadiometer (Welmy™ W300A, São Paulo, SP, Brazil) were used to measure the body mass and height. BMI was calculated accordingly. Neck, waist, and hip circumferences were obtained using a flexible measuring tape using standard procedures. Patients in two consecutive medical appointments self-reported preoperative weight and minimum postoperative weight. The excess weight loss (EWL) and RWR were calculated as follows, respectively: preoperative weight − minimum postoperative weight)/(preoperative weight − ideal weight for BMI of 25 kg/m2) × 100% [18] and current weight − minimum weight postoperative)/(preoperative weight − minimum weight postoperative) × 100% [19]. Blood pressure and heart rate were measured using a semiautomated oscillometric device (G-Tech™ BSP11, Hangzhou, Zhejiang, China) following standard recommendations [20].
Biochemical and Metabolic Profile Analysis
Glycated hemoglobin type A1c (HbA1c) and plasma glucose were evaluated by turbidimetric inhibition and glucose oxidase colorimetric methods, respectively. Serum levels of creatinine were assessed by kinetic method (Jaffé method without deproteinization). According to the International Federation of Clinical Chemistry method, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were assayed. Uricase enzymatic/colorimetric method determined levels of uric acid.
Serum levels of triglycerides, total cholesterol (TC), and high-density lipoprotein cholesterol (HDL-c) were assessed by glycerol phosphate oxidase/peroxidase, cholesterol oxidase/peroxidase, and direct colorimetric methods, respectively. These analyses were performed by commercially available kits appropriate for the Automatic Analyser A25 (BioSystems, Barcelona, Spain), according to protocols provided by the kit’s manufacturer (BioSystems, Barcelona, Spain). Low-density lipoprotein cholesterol (LDL-c) was calculated by the Friedewald equation [21].
Analysis of Inflammatory Biomarkers
Blood was harvested into serum tubes to determine levels of interferon-γ (INF-γ), interferon gamma–induced protein-10 (IP-10), monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), interleukin-1β (IL-1 β), interleukin-17 (IL-17), and interleukin-10 (IL-10). Collection tubes were centrifuged over 10 min at 4 °C at 1000 g. Serum was then transferred into cryotubes and stored at – 80 °C until analysis. Serum levels of IFN-γ, IP-10, and MCP-1 were evaluated by Human Quantikine® Immunoassay kits (R&D Systems, MN, USA), sensitivities: 1.28 pg/ml, 4.46 pg/ml, and 10 pg/ml, respectively. Circulating levels of TNF-α, IL-6, IL-1 β, IL-17, and IL-10 were assessed using Quantikine® High Sensitivity Human Immunoassay kits (R&D Systems, MN, USA) with sensitivities: 0.049 pg/ml, 0.09 pg/ml, 0.063 pg/ml, 0.051 pg/ml, and 0.17 pg/ml, respectively. All assays were performed according to directions provided by the kits’ manufacturer. Within-assay coefficient of variation of all analyses was less than 10%.
Statistical Analysis
Data normality was tested by the Shapiro–Wilk test, and results were expressed as mean ± standard deviation (SD) or median (interquartile range). Between-group differences (bariatric vs. nonsurgical groups) were compared by unpaired Student t-test. The chi-square test compared clinical characteristics between groups, and the results were expressed as frequency (n, %). Pearson correlation coefficients analyzed the associations between self-reported information (pre and minimum post-surgery weight). All calculations were performed by NCSS™ statistical software (Kaysville, UT, USA), and statistical significance was set at P ≤ 0.05.
Results
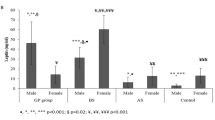
Demographic characteristics, clinical history, and bariatric surgery data of participants are displayed in Table 1. Despite the same BMI, a significantly lower percentage of patients with T2DM and dyslipidemia were detected in the bariatric group than the nonsurgical one (P ≤ 0.05). The groups were similar regarding all variables, except for neck and waist circumferences, significantly lower in those subjected to bariatric surgery (P ≤ 0.04).
Table 2 exhibit the biochemical and metabolic characteristics and inflammatory status of participants. Fasting glucose, HbA1c, and triglycerides were significantly lower in the bariatric compared to nonsurgical group (P ≤ 0.05), while HDL-c was significantly higher (P < 0.001). No difference between groups was detected concerning inflammatory biomarkers (P ≥ 0.10), except for IL-6, which was significantly lower in the bariatric group (P < 0.001). Serum levels of IFN-γ were detected in only three post-bariatric patients (1.491 ± 1.325 pg/ml) and were not used in this analysis.
Discussion
Bariatric surgery is an effective treatment for obesity. Some of our patients subjected to this procedure are still coming back to medical appointment with obesity. Unfortunately, high RWR occurs in a subgroup of patients which is being a clinical challenge for medical management. The inflammatory status and clinical characteristics of patients with obesity with or without bariatric procedure are unknown. In our study, even with obesity, post-bariatric patients showed lower rates of T2DM and dyslipidemia than the nonsurgical group with obesity (15 vs. 48.7% and 30 vs. 51.3%, respectively). The present study noticed more favorable metabolic health in the bariatric group than in the nonsurgical controls matched for BMI, age, and gender, expressed by lower neck and waist circumferences, fasting glucose, HbA1c, triglycerides, and higher HDL-c. Additionally, we originally observed that post-bariatric patients, even still with obesity, had lower circulating IL-6 than BMI-matched patients without any surgical intervention.
It is widely accepted that substantial weight loss could explain the long-term durability of the metabolic benefits obtained [22]. Previous studies demonstrated high rates of resolution of diabetes and improvement of lipid profile with lower TC and triglycerides, and higher HDL-c in long-term follow-up post-surgery [23, 24]. In our study, the post-bariatric patients had a long time since surgery (10.7 ± 4.3 years) and reached high rates of weight loss (about 85.9 ± 16.8% of EWL). We could speculate that maybe these rates of weight loss can explain some of our findings. Accumulating evidence has shown that post-bariatric weight loss elicits favorable endocrine responses of adipose tissue, increasing adiponectin, and reducing leptin secretions, improving health indicators [25]. Obesity is a complex disease that involves many pathophysiological aspects altogether, favoring the accumulation of energy as adipose tissue. Although far from our desire, even after bariatric surgery, it is common to observe weight regain [10, 17]. Approximately 10–20% of patients regain part of their lost weight at long-term follow-up [6]. Considering cardiovascular diseases, those cases with higher RWR and the current BMI are associated with increased mortality and morbidity [26, 27].
In the present study, our patients presented substantially higher level of weight regain (ranging from 36.8 to 76.2%) than others, which had RWR ranging from 3.9 to 38% during the long-term follow-up (3–7 years or more) after laparoscopic adjustable gastric banding, sleeve gastrectomy, or RYGB [28,29,30]. As previously published, our patients were invited to participate by advertisements [17]. They were all without any medical follow-up before their visit to our public health unit. Their high RWR is possibly also related to it. Therefore, we may speculate that the clinical results are more likely linked to weight loss than regain.
Unexpectedly, IL-6 levels were lower in those subjected to RYGB, even being all obese patients. All other inflammatory biomarkers were similar between bariatric and nonsurgical groups. Accumulating evidence in the literature reinforces the direct relationship between obesity and increase of pro-inflammatory response [16, 31, 32]. On the opposite of this statement, even with obesity again, our post-bariatric patients had lower IL-6 than nonsurgical BMI-matched group. These results add data from prior research, in which it was shown plasma levels of IL-6 were reduced 12 months after bariatric procedures, as RYGB and sleeve gastrectomy [33, 34], suggesting that reductions of IL-6 levels are probably more related to effective weight loss than obesity recidivism.
Interestingly, the waist circumference was lower in the bariatric group, which concurs with previous studies that showed positive correlations between IL-6 and abdominal adiposity [32]. The IL-6 is acknowledged to have pro-inflammatory properties and to be involved in pathogenesis of insulin resistance, T2DM, and atherosclerosis [35, 36]. Therefore, it seems that reduction of IL-6 levels may be related to improvement of metabolic and cardiovascular risk, reflected by decreased fasting glucose, HbA1c, triglycerides, and increased HDL-c.
In our study, it can be speculated that bariatric surgery promoted a long-term sustainable improvement in metabolic profile. It can also be suggested that even in post-bariatric patients with high RWR, effective post-surgery weight loss may prevent the augmenting of IL-6, which is paramount to maintaining their metabolic and cardiovascular integrity. Further research is needed to ratify these results.
Limitation
Although promising, our results must be viewed with caution. Since we did not evaluate pre-surgical clinical conditions and inflammatory status, we have limitations on our statements herein presented. Minimal specificities regarding differences on surgeons’ techniques must be considered as a limitation. However, an effective EWL was successfully achieved. Therefore, surgical failure can be discarded. The cross-sectional design limits cause-and-effect relationships. A healthy age-matched control group would allow a better understanding of the status of chronic inflammation and lack of it is another limitation. Ideally, the clinical long-term follow-up of these patients would lower the potential sources of bias since clinical outcomes may be differently affected by multidisciplinary approaches. Lastly, the different recruitment methods for studied groups are also further selection bias. However, as our medical team followed all patients for at least one year, we believe that careful screening minimized possible interference with the outcomes studied. Therefore, it is possible to believe that the potential abovementioned bias due to secondary diseases or medications on inflammatory markers were eliminated.
Conclusion
Our results have shown that an expressive post-bariatric weight loss, even in those patients with high ratio of weight regain, may maintain the improved metabolic profile. Probably, the inflammatory status, particularly the observed decreased circulating levels of interleukin-6, seemed to play an essential role in our findings. Further studies are needed to better define the possible metabolic protection in post-bariatric patients in the long term.
References
Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19·2 million participants. Lancet. 2016;387(10026):1377–96.
Yumuk V, Tsigos C, Fried M, Schindler K, Busetto L, Micic D, et al. European Guidelines for Obesity Management in Adults. Obes Facts. 2015;8(6):402–24.
Calle EE, Thun MJ, Petrelli JM, Rodriguez C, Heath CW Jr. Body-mass index and mortality in a prospective cohort of U.S. adults. N Engl J Med. 1999;341(15):1097–105.
Must A, Spadano J, Coakley EH, Field AE, Colditz G, Dietz WH. The disease burden associated with overweight and obesity. JAMA. 1999;282(16):1523–9.
Bray GA, Heisel WE, Afshin A, Jensen MD, Dietz WH, Long M, et al. The science of obesity management: an endocrine society scientific statement. Endocr Rev. 2018;39(2):79–132.
Sjöström CD, Lissner L, Wedel H, Sjöström L. Reduction in incidence of diabetes, hypertension and lipid disturbances after intentional weight loss induced by bariatric surgery: the SOS Intervention Study. Obes Res. 1999;7(5):477–84.
Reges O, Greenland P, Dicker D, Leibowitz M, Hoshen M, Gofer I, et al. Association of bariatric surgery using laparoscopic banding, Roux-en-Y gastric bypass, or laparoscopic sleeve gastrectomy vs usual care obesity management with all-cause mortality. JAMA. 2018;319(3):279–90.
Askarpour M, Khani D, Sheikhi A, Ghaedi E, Alizadeh S. Effect of bariatric surgery on serum inflammatory factors of obese patients: a systematic review and meta-analysis. Obes Surg. 2019;29(8):2631–47.
Yanos BR, Saules KK, Schuh LM, Sogg S. Predictors of lowest weight and long-term weight regain among Roux-en-Y gastric bypass patients. Obes Surg. 2015;25(8):1364–70.
Karmali S, Brar B, Shi X, Sharma AM, de Gara C, Birch DW. Weight recidivism post-bariatric surgery: a systematic review. Obes Surg. 2013;23(11):1922–33.
Peterli R, Wölnerhanssen BK, Peters T, Vetter D, Kröll D, Borbély Y, et al. Effect of laparoscopic sleeve gastrectomy vs laparoscopic Roux-en-Y gastric bypass on weight loss in patients with morbid obesity: the SM-BOSS randomized clinical trial. JAMA. 2018;319(3):255–65.
Lauti M, Kularatna M, Hill AG, MacCormick AD. Weight regain following sleeve gastrectomy-a systematic review. Obes Surg. 2016;26(6):1326–34.
Jiménez A, Casamitjana R, Flores L, Viaplana J, Corcelles R, Lacy A, et al. Long-term effects of sleeve gastrectomy and Roux-en-Y gastric bypass surgery on type 2 diabetes mellitus in morbidly obese subjects. Ann Surg. 2012;256(6):1023–9.
Lee PC, Dixon JB, Sim PY, Lim CH. Treatment options for poor responders to bariatric surgery. Curr Obes Rep. 2020;9(3):364–72.
King WC, Hinerman AS, Belle SH, Wahed AS, Courcoulas AP. Comparison of the performance of common measures of weight regain after bariatric surgery for association with clinical outcomes. JAMA. 2018;320(15):1560–9.
Kawai T, Autieri MV, Scalia R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am J Physiol Cell Physiol. 2021;320(3):C375–91.
Romagna EC, Lopes KG, Mattos DMF, Farinatti P, Kraemer-Aguiar LG. Physical activity level, sedentary time, and weight regain after bariatric surgery in patients without regular medical follow-up: a cross-sectional study. Obes Surg. 2021;31(4):1705–13.
Arterburn DE, Telem DA, Kushner RF, Courcoulas AP. Benefits and risks of bariatric surgery in adults: a review. JAMA. 2020;324(9):879–87.
Cooper TC, Simmons EB, Webb K, Burns JL, Kushner RF. Trends in weight regain following Roux-en-Y gastric bypass (RYGB) bariatric surgery. Obes Surg. 2015;25(8):1474–81.
Pickering TG, Hall JE, Appel LJ, Falkner BE, Graves J, Hill MN, et al. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension. 2005;45(1):142–61.
Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). Jama. 2001;285(19):2486–97.
Adams TD, Davidson LE, Litwin SE, Kim J, Kolotkin RL, Nanjee MN, et al. Weight and metabolic outcomes 12 years after gastric bypass. N Engl J Med. 2017;377(12):1143–55.
Buchwald H, Estok R, Fahrbach K, Banel D, Jensen MD, Pories WJ, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122(3):248-56.e5.
Ikramuddin S, Korner J, Lee WJ, Thomas AJ, Connett JE, Bantle JP, et al. Lifestyle intervention and medical management with vs without Roux-en-Y gastric bypass and control of hemoglobin A1c, LDL cholesterol, and systolic blood pressure at 5 years in the diabetes surgery study. JAMA. 2018;319(3):266–78.
Appachi S, Kelly KR, Schauer PR, Kirwan JP, Hazen S, Gupta M, et al. Reduced cardiovascular risk following bariatric surgeries is related to a partial recovery from “adiposopathy.” Obes Surg. 2011;21(12):1928–36.
Pischon T, Boeing H, Hoffmann K, Bergmann M, Schulze MB, Overvad K, et al. General and abdominal adiposity and risk of death in Europe. N Engl J Med. 2008;359(20):2105–20.
Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, et al. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet. 2009;373(9669):1083–96.
Sjöström L, Lindroos AK, Peltonen M, Torgerson J, Bouchard C, Carlsson B, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351(26):2683–93.
Courcoulas AP, King WC, Belle SH, Berk P, Flum DR, Garcia L, et al. Seven-year weight trajectories and health outcomes in the Longitudinal Assessment Of Bariatric Surgery (LABS) study. JAMA Surg. 2018;153(5):427–34.
Clapp B, Wynn M, Martyn C, Foster C, O’Dell M, Tyroch A. Long term (7 or more years) outcomes of the sleeve gastrectomy: a meta-analysis. Surg Obes Relat Dis. 2018;14(6):741–7.
Fernández-Sánchez A, Madrigal-Santillán E, Bautista M, Esquivel-Soto J, Morales-González A, Esquivel-Chirino C, et al. Inflammation, oxidative stress, and obesity. Int J Mol Sci. 2011;12(5):3117–32.
Thorand B, Baumert J, Döring A, Herder C, Kolb H, Rathmann W, et al. Sex differences in the relation of body composition to markers of inflammation. Atherosclerosis. 2006;184(1):216–24.
Viana EC, Araujo-Dasilio KL, Miguel GP, Bressan J, Lemos EM, Moyses MR, et al. Gastric bypass and sleeve gastrectomy: the same impact on IL-6 and TNF-α. Prospect Clin Trial Obes Surg. 2013;23(8):1252–61.
Swarbrick MM, Stanhope KL, Austrheim-Smith IT, Van Loan MD, Ali MR, Wolfe BM, et al. Longitudinal changes in pancreatic and adipocyte hormones following Roux-en-Y gastric bypass surgery. Diabetologia. 2008;51(10):1901–11.
Collaboration IRGCERF, Sarwar N, Butterworth AS, Freitag DF, Gregson J, Willeit P, et al. Interleukin-6 receptor pathways in coronary heart disease: a collaborative meta-analysis of 82 studies. Lancet (London, England). 2012;379(9822):1205–13.
Pradhan AD, Manson JE, Rifai N, Buring JE, Ridker PM. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA. 2001;286(3):327–34.
Funding
This study received funding from Coordination for the Improvement of Higher Education Personnel (CAPES, process 88882.463218/2019–01, recipient KGL) and Carlos Chagas Filho Foundation for the Research Support in the State of Rio de Janeiro (FAPERJ, process 250304, recipient LGK-A).
Author information
Authors and Affiliations
Contributions
The original study design was made by KGL and LGK-A. The project administration and supervision were performed by LGK-A. KGL, ECR, DSS, MCTB, PRF, JES, and KGL recruited the patients and collected the data. KGL, MGS, and LGK-A analyzed the data; interpreted the results; and drafted the manuscript. KGL, ECR, JES, EB, MGS, and LGK-A revised the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All procedures performed involving the participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the local Ethics Committee (CAAE: 16425419.8.0000.5259) and registered in ClinicalTrials.gov (NCT04193397).
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
• High weight regain may occur in post-bariatric patients without medical follow-up.
• Metabolic protection occurs in post-bariatric patients even with weight regain.
• Decreased interleukin-6 was observed in post-bariatric patients with high weight regain.
Rights and permissions
About this article
Cite this article
Lopes, K.G., Romagna, E.C., da Silva, D.S. et al. Metabolic and Inflammatory Profiles of Post-Bariatric Patients with Weight Recidivism. OBES SURG 32, 1849–1855 (2022). https://doi.org/10.1007/s11695-022-06025-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-022-06025-9