Abstract
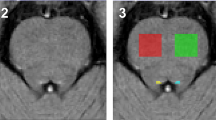
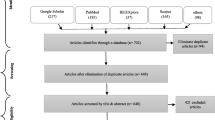
This study aimed to define the most consistent white matter microarchitecture pattern in Parkinson’s disease (PD) reflected by fractional anisotropy (FA), addressing clinical profiles and methodology-related heterogeneity. Web-based publication databases were searched to conduct a meta-analysis of whole-brain diffusion tensor imaging studies comparing PD with healthy controls (HC) using the anisotropic effect size–signed differential mapping. A total of 808 patients with PD and 760 HC coming from 27 databases were finally included. Subgroup analyses were conducted considering heterogeneity with respect to medication status, disease stage, analysis methods, and the number of diffusion directions in acquisition. Compared with HC, patients with PD had decreased FA in the left middle cerebellar peduncle, corpus callosum (CC), left inferior fronto-occipital fasciculus, and right inferior longitudinal fasciculus. Most of the main results remained unchanged in subgroup meta-analyses of medicated patients, early stage patients, voxel-based analysis, and acquisition with 30 diffusion directions. The subgroup meta-analysis of medication-free patients showed FA decrease in the right olfactory cortex. The cerebellum and CC, associated with typical motor impairment, showed the most consistent FA decreases in PD. Medication status, analysis approaches, and the number of diffusion directions have an important impact on the findings, needing careful evaluation in future meta-analyses.
Similar content being viewed by others
References
de Lau LM, Breteler MM. Epidemiology of Parkinson’s disease. Lancet Neurol 2006; 5(6): 525–535
Reichmann H, Brandt MD, Klingelhoefer L. The nonmotor features of Parkinson’s disease: pathophysiology and management advances. Curr Opin Neurol 2016; 29(4): 467–473
Kubicki M, Westin CF, Maier SE, Mamata H, Frumin M, Ersner- Hershfield H, Kikinis R, Jolesz FA, McCarley R, Shenton ME. Diffusion tensor imaging and its application to neuropsychiatric disorders. Harv Rev Psychiatry 2002; 10(6): 324–336
Taylor WD, Hsu E, Krishnan KR, MacFall JR. Diffusion tensor imaging: background, potential, and utility in psychiatric research. Biol Psychiatry 2004; 55(3): 201–207
Le Bihan D, Mangin JF, Poupon C, Clark CA, Pappata S, Molko N, Chabriat H. Diffusion tensor imaging: concepts and applications. J Magn Reson Imaging 2001; 13(4): 534–546
Chen B, Fan G, Sun W, Shang X, Shi S, Wang S, Lv G, Wu C. Usefulness of diffusion-tensor MRI in the diagnosis of Parkinson variant of multiple system atrophy and Parkinson’s disease: a valuable tool to differentiate between them? Clin Radiol 2017; 72 (7): 610.e9–610.e15
Ji L, Wang Y, Zhu D, Liu W, Shi J. White matter differences between multiple system atrophy (parkinsonian type) and Parkinson’s disease: a diffusion tensor image study. Neuroscience 2015; 305: 109–116
Vercruysse S, Leunissen I, Vervoort G, Vandenberghe W, Swinnen S, Nieuwboer A. Microstructural changes in white matter associated with freezing of gait in Parkinson’s disease. Mov Disord 2015; 30 (4): 567–576
Karagulle Kendi AT, Lehericy S, Luciana M, Ugurbil K, Tuite P. Altered diffusion in the frontal lobe in Parkinson disease. AJNR Am J Neuroradiol 2008; 29(3): 501–505
Chiang PL, Chen HL, Lu CH, Chen PC, Chen MH, Yang IH, Tsai NW, Lin WC. White matter damage and systemic inflammation in Parkinson’s disease. BMC Neurosci 2017; 18(1): 48
Chen MH, Chen PC, Lu CH, Chen HL, Chao YP, Li SH, Chen YW, Lin WC. Plasma DNA mediate autonomic dysfunctions and white matter injuries in patients with Parkinson’s disease. Oxid Med Cell Longev 2017; 2017: 7371403
Zhang K, Yu C, Zhang Y, Wu X, Zhu C, Chan P, Li K. Voxel-based analysis of diffusion tensor indices in the brain in patients with Parkinson’s disease. Eur J Radiol 2011; 77(2): 269–273
Luo C, Song W, Chen Q, Yang J, Gong Q, Shang HF. White matter microstructure damage in tremor-dominant Parkinson’s disease patients. Neuroradiology 2017; 59(7): 691–698
Worker A, Blain C, Jarosz J, Chaudhuri KR, Barker GJ, Williams SC, Brown RG, Leigh PN, Dell’Acqua F, Simmons A. Diffusion tensor imaging of Parkinson’s disease, multiple system atrophy and progressive supranuclear palsy: a tract-based spatial statistics study. PLoS One 2014; 9(11): e112638
Agosta F, Canu E, Stojković T, Pievani M, Tomić A, Sarro L, Dragašević N, Copetti M, Comi G, Kostić VS, Filippi M. The topography of brain damage at different stages of Parkinson’s disease. Hum Brain Mapp 2013; 34(11): 2798–2807
Schwarz ST, Abaei M, Gontu V, Morgan PS, Bajaj N, Auer DP. Diffusion tensor imaging of nigral degeneration in Parkinson’s disease: a region-of-interest and voxel-based study at 3 T and systematic review with meta-analysis. Neuroimage Clin 2013; 3: 481–488
Atkinson-Clement C, Pinto S, Eusebio A, Coulon O. Diffusion tensor imaging in Parkinson’s disease: review and meta-analysis. Neuroimage Clin 2017; 16: 98–110
Cochrane CJ, Ebmeier KP. Diffusion tensor imaging in parkinsonian syndromes: a systematic review and meta-analysis. Neurology 2013; 80(9): 857–864
Albrecht F, Ballarini T, Neumann J, Schroeter ML. FDG-PET hypometabolism is more sensitive than MRI atrophy in Parkinson’s disease: a whole-brain multimodal imaging meta-analysis. Neuroimage Clin 2019; 21: 101594
Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009; 6(7): e1000097
Pan P, Zhan H, Xia M, Zhang Y, Guan D, Xu Y. Aberrant regional homogeneity in Parkinson’s disease: a voxel-wise meta-analysis of resting-state functional magnetic resonance imaging studies. Neurosci Biobehav Rev 2017; 72: 223–231
Shepherd AM, Matheson SL, Laurens KR, Carr VJ, Green MJ. Systematic meta-analysis of insula volume in schizophrenia. Biol Psychiatry 2012; 72(9): 775–784
Radua J, Mataix-Cols D. Voxel-wise meta-analysis of grey matter changes in obsessive-compulsive disorder. Br J Psychiatry 2009; 195(5): 393–402
Radua J, Mataix-Cols D, Phillips ML, El-Hage W, Kronhaus DM, Cardoner N, Surguladze S. A new meta-analytic method for neuroimaging studies that combines reported peak coordinates and statistical parametric maps. Eur Psychiatry 2012; 27(8): 605–611
Radua J, Rubia K, Canales-Rodríguez EJ, Pomarol-Clotet E, Fusar- Poli P, Mataix-Cols D. Anisotropic kernels for coordinate-based meta-analyses of neuroimaging studies. Front Psychiatry 2014; 5: 13
Wise T, Radua J, Nortje G, Cleare AJ, Young AH, Arnone D. Voxelbased meta-analytical evidence of structural disconnectivity in major depression and bipolar disorder. Biol Psychiatry 2016; 79(4): 293–302
Radua J, Grau M, van den Heuvel OA, Thiebaut de Schotten M, Stein DJ, Canales-Rodríguez EJ, Catani M, Mataix-Cols D. Multimodal voxel-based meta-analysis of white matter abnormalities in obsessive-compulsive disorder. Neuropsychopharmacology 2014; 39(7): 1547–1557
Chen S, Chan P, Sun S, Chen H, Zhang B, Le W, Liu C, Peng G, Tang B, Wang L, Cheng Y, Shao M, Liu Z, Wang Z, Chen X, Wang M, Wan X, Shang H, Liu Y, Xu P, Wang J, Feng T, Chen X, Hu X, Xie A, Xiao Q. The recommendations of Chinese Parkinson’s disease and movement disorder society consensus on therapeutic management of Parkinson’s disease. Transl Neurodegener 2016; 5 (1): 12
Jones DK. The effect of gradient sampling schemes on measures derived from diffusion tensor MRI: a Monte Carlo study. Magn Reson Med 2004; 51(4): 807–815
Guan X, Huang P, Zeng Q, Liu C, Wei H, Xuan M, Gu Q, Xu X, Wang N, Yu X, Luo X, Zhang M. Quantitative susceptibility mapping as a biomarker for evaluating white matter alterations in Parkinson’s disease. Brain Imaging Behav 2019; 13(1): 220–231
Wen MC, Heng HSE, Lu Z, Xu Z, Chan LL, Tan EK, Tan LCS. Differential white matter regional alterations in motor subtypes of early drug-naive Parkinson’s disease patients. Neurorehabil Neural Repair 2018; 32(2): 129–141
Péran P, Barbagallo G, Nemmi F, Sierra M, Galitzky M, Traon AP, Payoux P, Meissner WG, Rascol O. MRI supervised and unsupervised classification of Parkinson’s disease and multiple system atrophy. Mov Disord 2018; 33(4): 600–608
Rektor I, Svátková A, Vojtíšek L, Zikmundová I, Vaníček J, Király A, Szabó N. White matter alterations in Parkinson’s disease with normal cognition precede grey matter atrophy. PLoS One 2018; 13 (1): e0187939
Acosta-Cabronero J, Cardenas-Blanco A, Betts MJ, Butryn M, Valdes-Herrera JP, Galazky I, Nestor PJ. The whole-brain pattern of magnetic susceptibility perturbations in Parkinson’s disease. Brain 2017; 140(1): 118–131
Zanigni S, Evangelisti S, Testa C, Manners DN, Calandra-Buonaura G, Guarino M, Gabellini A, Gramegna LL, Giannini G, Sambati L, Cortelli P, Lodi R, Tonon C. White matter and cortical changes in atypical parkinsonisms: a multimodal quantitative MR study. Parkinsonism Relat Disord 2017; 39: 44–51
Vervoort G, Leunissen I, Firbank M, Heremans E, Nackaerts E, Vandenberghe W, Nieuwboer A. Structural brain alterations in motor subtypes of Parkinson’s disease: evidence from probabilistic tractography and shape analysis. PLoS One 2016; 11(6): e0157743
Agosta F, Canu E, Stefanova E, Sarro L, Tomić A, Špica V, Comi G, Kostić VS, Filippi M. Mild cognitive impairment in Parkinson’s disease is associated with a distributed pattern of brain white matter damage. Hum Brain Mapp 2014; 35(5): 1921–1929
Kamagata K, Motoi Y, Tomiyama H, Abe O, Ito K, Shimoji K, Suzuki M, Hori M, Nakanishi A, Sano T, Kuwatsuru R, Sasai K, Aoki S, Hattori N. Relationship between cognitive impairment and white-matter alteration in Parkinson’s disease with dementia: tractbased spatial statistics and tract-specific analysis. Eur Radiol 2013; 23(7): 1946–1955
Kim HJ, Kim SJ, Kim HS, Choi CG, Kim N, Han S, Jang EH, Chung SJ, Lee CS. Alterations of mean diffusivity in brain white matter and deep gray matter in Parkinson’s disease. Neurosci Lett 2013; 550: 64–68
Melzer TR, Watts R, MacAskill MR, Pitcher TL, Livingston L, Keenan RJ, Dalrymple-Alford JC, Anderson TJ. White matter microstructure deteriorates across cognitive stages in Parkinson disease. Neurology 2013; 80(20): 1841–1849
Hattori T, Orimo S, Aoki S, Ito K, Abe O, Amano A, Sato R, Sakai K, Mizusawa H. Cognitive status correlates with white matter alteration in Parkinson’s disease. Hum Brain Mapp 2012; 33(3): 727–739
Kamagata K, Zalesky A, Hatano T, Ueda R, Di Biase MA, Okuzumi A, Shimoji K, Hori M, Caeyenberghs K, Pantelis C, Hattori N, Aoki S. Gray matter abnormalities in idiopathic Parkinson’s disease: evaluation by diffusional kurtosis imaging and neurite orientation dispersion and density imaging. Hum Brain Mapp 2017; 38(7): 3704–3722
Rosskopf J, Müller HP, Huppertz HJ, Ludolph AC, Pinkhardt EH, Kassubek J. Frontal corpus callosum alterations in progressive supranuclear palsy but not in Parkinson’s disease. Neurodegener Dis 2014; 14(4): 184–193
Ziegler E, Rouillard M, André E, Coolen T, Stender J, Balteau E, Phillips C, Garraux G. Mapping track density changes in nigrostriatal and extranigral pathways in Parkinson’s disease. Neuroimage 2014; 99: 498–508
Stoodley CJ, Schmahmann JD. Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex 2010; 46(7): 831–844
Wu T, Hallett M. The cerebellum in Parkinson’s disease. Brain 2013; 136(3): 696–709
Schweder PM, Hansen PC, Green AL, Quaghebeur G, Stein J, Aziz TZ. Connectivity of the pedunculopontine nucleus in parkinsonian freezing of gait. Neuroreport 2010; 21(14): 914–916
Canu E, Agosta F, Sarasso E, Volontè MA, Basaia S, Stojkovic T, Stefanova E, Comi G, Falini A, Kostic VS, Gatti R, Filippi M. Brain structural and functional connectivity in Parkinson’s disease with freezing of gait. Hum Brain Mapp 2015; 36(12): 5064–5078
Tessa C, Lucetti C, Diciotti S, Paoli L, Cecchi P, Giannelli M, Baldacci F, Ginestroni A, Vignali C, Mascalchi M, Bonuccelli U. Hypoactivation of the primary sensorimotor cortex in de novo Parkinson’s disease: a motor fMRI study under controlled conditions. Neuroradiology 2012; 54(3): 261–268
Hall JM, Ehgoetz Martens KA, Walton CC, O’Callaghan C, Keller PE, Lewis SJ, Moustafa AA. Diffusion alterations associated with Parkinson’s disease symptomatology: a review of the literature. Parkinsonism Relat Disord 2016; 33: 12–26
Sobhani S, Rahmani F, Aarabi MH, Sadr AV. Exploring white matter microstructure and olfaction dysfunction in early Parkinson disease: diffusion MRI reveals new insight. Brain Imaging Behav 2019; 13(1): 210–219
Catani M, Howard RJ, Pajevic S, Jones DK. Virtual in vivo interactive dissection of white matter fasciculi in the human brain. Neuroimage 2002; 17(1): 77–94
Cronin-Golomb A. Parkinson’s disease as a disconnection syndrome. Neuropsychol Rev 2010; 20(2): 191–208
Hu X, Zhang J, Jiang X, Zhou C, Wei L, Yin X, Wu Y, Li J, Zhang Y, Wang J. Decreased interhemispheric functional connectivity in subtypes of Parkinson’s disease. J Neurol 2015; 262(3): 760–767
Luo C, Guo X, Song W, Zhao B, Cao B, Yang J, Gong Q, Shang HF. Decreased resting-state interhemispheric functional connectivity in Parkinson’s disease. BioMed Res Int 2015; 2015: 692684
Li J, Yuan Y, Wang M, Zhang J, Zhang L, Jiang S, Wang X, Ding J, Zhang K. Decreased interhemispheric homotopic connectivity in Parkinson’s disease patients with freezing of gait: a resting state fMRI study. Parkinsonism Relat Disord 2018; 52: 30–36
Zheng Z, Shemmassian S, Wijekoon C, Kim W, Bookheimer SY, Pouratian N. DTI correlates of distinct cognitive impairments in Parkinson’s disease. Hum Brain Mapp 2014; 35(4): 1325–1333
Gorges M, Müller HP, Liepelt-Scarfone I, Storch A, Dodel R; LANDSCAPE Consortium, Hilker-Roggendorf R, Berg D, Kunz MS, Kalbe E, Baudrexel S, Kassubek J, Kassubek J. Structural brain signature of cognitive decline in Parkinson’s disease: DTI-based evidence from the LANDSCAPE study. Ther Adv Neurol Disorder 2019; 12: 1756286419843447
Vasconcellos LF, Pereira JS, Adachi M, Greca D, Cruz M, Malak AL, Charchat-Fichman H. Volumetric brain analysis as a predictor of a worse cognitive outcome in Parkinson’s disease. J Psychiatr Res 2018; 102: 254–260
Chan LL, Ng KM, Rumpel H, Fook-Chong S, Li HH, Tan EK. Transcallosal diffusion tensor abnormalities in predominant gait disorder parkinsonism. Parkinsonism Relat Disord 2014; 20(1): 53–59
Galantucci S, Agosta F, Stankovic I, Petrovic I, Stojkovic T, Kostic V, Filippi M. Corpus callosum damage and motor function in Parkinson’s disease (P2.006). Neurology 2014; 82(10 Supplement): P2.006
Catani M, Jones DK, Donato R, Ffytche DH. Occipito-temporal connections in the human brain. Brain 2003; 126(9): 2093–2107
Fox CJ, Iaria G, Barton JJ. Disconnection in prosopagnosia and face processing. Cortex 2008; 44(8): 996–1009
Ffytche DH. The hodology of hallucinations. Cortex 2008; 44(8): 1067–1083
Ross ED. Sensory-specific amnesia and hypoemotionality in humans and monkeys: gateway for developing a hodology of memory. Cortex 2008; 44(8): 1010–1022
Catani M. From hodology to function. Brain 2007; 130(3): 602–605
Catani M, Mesulam M. The arcuate fasciculus and the disconnection theme in language and aphasia: history and current state. Cortex 2008; 44(8): 953–961
Rudrauf D, Mehta S, Grabowski TJ. Disconnection’s renaissance takes shape: formal incorporation in group-level lesion studies. Cortex 2008; 44(8): 1084–1096
Haghshomar M, Dolatshahi M, Ghazi Sherbaf F, Sanjari Moghaddam H, Shirin Shandiz M, Aarabi MH. Disruption of inferior longitudinal fasciculus microstructure in Parkinson’s disease: a systematic review of diffusion tensor imaging studies. Front Neurol 2018; 9: 598
Lee E, Lee JE, Yoo K, Hong JY, Oh J, Sunwoo MK, Kim JS, Jeong Y, Lee PH, Sohn YH, Kang SY. Neural correlates of progressive reduction of bradykinesia in de novo Parkinson’s disease. Parkinsonism Relat Disord 2014; 20(12): 1376–1381
Wang M, Jiang S, Yuan Y, Zhang L, Ding J, Wang J, Zhang J, Zhang K, Wang J. Alterations of functional and structural connectivity of freezing of gait in Parkinson’s disease. J Neurol 2016; 263(8): 1583–1592
Tan SYZ, Keong NCH, Selvan RMP, Li H, Ooi LQR, Tan EK, Chan LL. Periventricular white matter abnormalities on diffusion tensor imaging of postural instability gait disorder parkinsonism. AJNR Am J Neuroradiol 2019; 40(4): 609–613
Wu JY, Zhang Y, Wu WB, Hu G, Xu Y. Impaired long contact white matter fibers integrity is related to depression in Parkinson’s disease. CNS Neurosci Ther 2018; 24(2): 108–114
Duncan GW, Firbank MJ, Yarnall AJ, Khoo TK, Brooks DJ, Barker RA, Burn DJ, O’Brien JT. Gray and white matter imaging: a biomarker for cognitive impairment in early Parkinson’s disease? Mov Disord 2016; 31(1): 103–110
Reijnders JS, Ehrt U, Weber WE, Aarsland D, Leentjens AF. A systematic review of prevalence studies of depression in Parkinson’s disease. Mov Disord 2008; 23(2): 183–189, quiz 313
Yu J, Lam CLM, Lee TMC. White matter microstructural abnormalities in amnestic mild cognitive impairment: a metaanalysis of whole-brain and ROI-based studies. Neurosci Biobehav Rev 2017; 83: 405–416
Bromis K, Calem M, Reinders AATS, Williams SCR, Kempton MJ. Meta-analysis of 89 structural MRI studies in posttraumatic stress disorder and comparison with major depressive disorder. Am J Psychiatry 2018; 175(10): 989–998
Hawkes CH, Del Tredici K, Braak H. Parkinson’s disease: a dual-hit hypothesis. Neuropathol Appl Neurobiol 2007; 33(6): 599–614
Nigro S, Riccelli R, Passamonti L, Arabia G, Morelli M, Nisticò R, Novellino F, Salsone M, Barbagallo G, Quattrone A. Characterizing structural neural networks in de novo Parkinson disease patients using diffusion tensor imaging. Hum Brain Mapp 2016; 37(12): 4500–4510
Zatorre RJ, Jones-Gotman M. Human olfactory discrimination after unilateral frontal or temporal lobectomy. Brain 1991; 114(Pt 1A): 71–84
Doty RL. Olfactory dysfunction in Parkinson disease. Nat Rev Neurol 2012; 8(6): 329–339
Berman BD, Smucny J, Wylie KP, Shelton E, Kronberg E, Leehey M, Tregellas JR. Levodopa modulates small-world architecture of functional brain networks in Parkinson’s disease. Mov Disord 2016; 31(11): 1676–1684
Dean DC 3rd, Sojkova J, Hurley S, Kecskemeti S, Okonkwo O, Bendlin BB, Theisen F, Johnson SC, Alexander AL, Gallagher CL. Alterations of myelin content in Parkinson’s disease: a crosssectional neuroimaging study. PLoS One 2016; 11(10): e0163774
Degirmenci B, Yaman M, Haktanir A, Albayrak R, Acar M, Caliskan G. The effects of levodopa use on diffusion coefficients in various brain regions in Parkinson’s disease. Neurosci Lett 2007; 416(3): 294–298
Smith SM, Jenkinson M, Johansen-Berg H, Rueckert D, Nichols TE, Mackay CE, Watkins KE, Ciccarelli O, Cader MZ, Matthews PM, Behrens TE. Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. Neuroimage 2006; 31(4): 1487–1505
Zalesky A. Moderating registration misalignment in voxelwise comparisons of DTI data: a performance evaluation of skeleton projection. Magn Reson Imaging 2011; 29(1): 111–125
Nortje G, Stein DJ, Radua J, Mataix-Cols D, Horn N. Systematic review and voxel-based meta-analysis of diffusion tensor imaging studies in bipolar disorder. J Affect Disord 2013; 150(2): 192–200
Acknowledgements
This study was supported by the National Natural Science Foundation (Nos. 81621003, 81761128023, 81220108013, 81227002, and 81030027), the Program for Innovative Research Team in University (No. IRT16R52) of China, the Professorship Award (No. T2014190) of China, and the CMB Distinguished Professorship Award (No. F510000/G16916411) administered by the Institute of International Education.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Xueling Suo, Du Lei, Wenbin Li, Lei Li, Jing Dai, Song Wang, Nannan Li, Lan Cheng, Rong Peng, Graham J Kemp, and Qiyong Gong declare that they have no conflict of interest. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5). Informed consent was obtained from all patients for being included in the study.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Suo, X., Lei, D., Li, W. et al. Altered white matter microarchitecture in Parkinson’s disease: a voxel-based meta-analysis of diffusion tensor imaging studies. Front. Med. 15, 125–138 (2021). https://doi.org/10.1007/s11684-019-0725-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11684-019-0725-5