Abstract
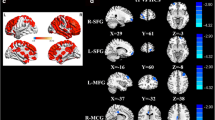
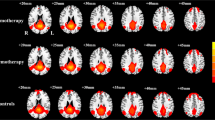
This study aimed to investigate alterations of brain functional network connectivity (FNC) in lung cancer patients after chemotherapy and explore links between these FNC differences and cognitive impairment. Twenty-two lung cancer patients receiving chemotherapy and 26 healthy controls (HCs) underwent resting-state functional MRI (rs-fMRI) and neuropsychological testing. Group independent component analysis (GICA) was applied to rs-fMRI data to extract whole-brain resting state networks (RSNs). Static and dynamic FNC (dFNC) were constructed to reveal RSNs connectivity alterations between lung cancer patients and HCs group, and the correlations between the group differences in RSNs and cognitive performance were analyzed. Our findings revealed that chemotherapeutics can produce widespread connectivity abnormalities in RSNs, mainly focused on default mode network (DMN) and executive control network. Furthermore, the dFNC analysis help identify network configurations of each state and capture more chemotherapy-induced disorders of interactions between and within RSNs, which mainly includes sensorimotor network, attentional network and auditory network. In addition, after chemotherapy, the lung cancer patients spend shorter mean dwell time (MDT) in state 2. The decreased dFNC between DMN [independent component 5 (IC5)] and DMN (IC6) in the lung cancer patients after chemotherapy in state 4 was negatively correlated with Montreal Cognitive Assessment (MoCA) scores (r=-0.447, p=0.042). The dFNC analysis enrich our understanding of the neural mechanisms underlying the chemobrain, and suggested that the temporal dynamics of FNC could be a potential effective method to detect cognitive changes in lung cancer patients receiving chemotherapy.




Similar content being viewed by others
Data availability
Imaging data could be provided upon request.
References
Allen, E. A., Damaraju, E., Plis, S. M., Erhardt, E. B., Eichele, T., & Calhoun, V. D. (2014). Tracking whole-brain connectivity dynamics in the resting state. Cerebral Cortex (New York, N.Y.: 1991), 24(3), 663-76. https://doi.org/10.1093/cercor/bhs352
Amidi, A., & Wu, L. M. (2019). Structural brain alterations following adult non-CNS cancers: a systematic review of the neuroimaging literature. Acta Oncologica (Stockholm, Sweden), 58(5), 522–536. https://doi.org/10.1080/0284186x.2018.1563716
Billiet, T., Emsell, L., Vandenbulcke, M., et al. (2018). Recovery from chemotherapy-induced white matter changes in young breast cancer survivors? Brain Imaging and Behavior, 12(1), 64–77. https://doi.org/10.1007/s11682-016-9665-8
Bromis, K., Gkiatis, K., Karanasiou, I., et al., (2017). Altered brain functional connectivity in small-cell lung cancer patients after chemotherapy treatment: a resting-state fMRI study. Computational and Mathematical Methods in Medicine, 2017, 1403940. https://doi.org/10.1155/2017/1403940
Calhoun, V. D., Adali, T., Pearlson, G. D., & Pekar, J. J. (2001). A method for making group inferences from functional MRI data using independent component analysis. Human Brain Mapping, 14(3), 140–151. https://doi.org/10.1002/hbm.1048
Chen, H., Ding, K., Zhao, J., Chao, H. H., Li, C. R., & Cheng, H. (2019). The dorsolateral prefrontal cortex is selectively involved in chemotherapy-related cognitive impairment in breast cancer patients with different hormone receptor expression. American Journal of Cancer Research, 9(8), 1776–1785
Chen, H. J., Lin, H. L., Chen, Q. F., & Liu, P. F. (2017). Altered dynamic functional connectivity in the default mode network in patients with cirrhosis and minimal hepatic encephalopathy. Neuroradiology, 59(9), 905–914. https://doi.org/10.1007/s00234-017-1881-4
Chen, V. C., Lin, K. Y., Tsai, Y. H., & Weng, J. C. (2020). Connectome analysis of brain functional network alterations in breast cancer survivors with and without chemotherapy. PLoS One1, 15(5), e0232548. https://doi.org/10.1371/journal.pone.0232548
Chen, X., He, X., Tao, L., et al., (2017). The attention network changes in breast cancer patients receiving neoadjuvant chemotherapy: Evidence from an arterial spin labeling perfusion study. Scientific Reports, 17, 742684. https://doi.org/10.1038/srep42684
De Luca, M., Beckmann, C. F., De Stefano, N., Matthews, P. M., & Smith, S. M. (2006). fMRI resting state networks define distinct modes of long-distance interactions in the human brain. NeuroImage, 15(4), 1359–1367. https://doi.org/10.1016/j.neuroimage.2005.08.035
Espinoza, F. A., Anderson, N. E., Vergara, V. M., et al., (2019). Resting-state fMRI dynamic functional network connectivity and associations with psychopathy traits. NeuroImage Clinical, 24, 101970. https://doi.org/10.1016/j.nicl.2019.101970
Feng, Y., Wang, Y. F., Zheng, L. J., Shi, Z., Huang, W., & Zhang, L. J. (2020). Network-level functional connectivity alterations in chemotherapy treated breast cancer patients: a longitudinal resting state functional MRI study. Cancer Imaging: the Official Publication of the International Cancer Imaging Society, 16(1), 73. https://doi.org/10.1186/s40644-020-00355-6
Fiorenzato, E., Strafella, A. P., Kim, J., et al., (2019). Dynamic functional connectivity changes associated with dementia in Parkinson’s disease. Brain: a Journal of Neurology, 1(9), 2860–2872. https://doi.org/10.1093/brain/awz192
Fox, M. D., & Raichle, M. E. (2007). Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nature Reviews. Neuroscience, 8(9), 700–711. https://doi.org/10.1038/nrn2201
Fu, Z., Caprihan, A., Chen, J., et al., (2019). Altered static and dynamic functional network connectivity in Alzheimer’s disease and subcortical ischemic vascular disease: shared and specific brain connectivity abnormalities. Human Brain Mapping, 1(11), 3203–3221. https://doi.org/10.1002/hbm.24591
Galea, M., & Woodward, M. (2005). Mini-Mental State Examination (MMSE). The Australian Journal of Physiotherapy, 51(3), 198
Hu, L., Chen, H., Su, W., et al., (2020). Aberrant static and dynamic functional connectivity of the executive control network in lung cancer patients after chemotherapy: a longitudinal fMRI study. Brain Imaging and Behavior, 14(3), 927–940. https://doi.org/10.1007/s11682-020-00287-6
Janelsins, M. C., Kesler, S. R., Ahles, T. A., & Morrow, G. R. (2014). Prevalence, mechanisms, and management of cancer-related cognitive impairment. International Review of Psychiatry (Abingdon, England), 26(1), 102–113. https://doi.org/10.3109/09540261.2013.864260
Kaasa, S., Olsnes, B. T., Thorud, E., & Host, H. (1988). Reduced short-term neuropsychological performance in patients with nonsmall-cell lung cancer treated with cisplatin and etoposide. Antibiotics and Chemotherapy, 41, 226–231
Kesler, S. R., Adams, M., Packer, M., et al., (2017). Disrupted brain network functional dynamics and hyper-correlation of structural and functional connectome topology in patients with breast cancer prior to treatment. Brain and Behavior, 7(3), e00643. https://doi.org/10.1002/brb3.643
Lepage, C., Smith, A. M., Moreau, J., et al., (2014). A prospective study of grey matter and cognitive function alterations in chemotherapy-treated breast cancer patients. Springerplus, 3, 444. https://doi.org/10.1186/2193-1801-3-444
Lewis, N., Lu, H., Liu, P., et al., (2020). Static and dynamic functional connectivity analysis of cerebrovascular reactivity: An fMRI study. Brain and Behavior, 10(6), e01516. https://doi.org/10.1002/brb3.1516
Li, M., & Caeyenberghs, K. (2018). Longitudinal assessment of chemotherapy-induced changes in brain and cognitive functioning: A systematic review. Neuroscience and Biobehavioral Reviews, 92, 304–317. https://doi.org/10.1016/j.neubiorev.2018.05.019
Li, X., Chen, H., Lv, Y., et al., (2018). Diminished gray matter density mediates chemotherapy dosage-related cognitive impairment in breast cancer patients. Scientific Reports, 14(1), 13801. https://doi.org/10.1038/s41598-018-32257-w
Liegeois, R., Li, J., Kong, R., et al., (2019). Resting brain dynamics at different timescales capture distinct aspects of human behavior. Nature Communications, 24(1), 2317. https://doi.org/10.1038/s41467-019-10317-7
Malhi, G. S., Das, P., Outhred, T., Bryant, R. A., & Calhoun, V. (2019). Resting-state neural network disturbances that underpin the emergence of emotional symptoms in adolescent girls: resting-state fMRI study. The British Journal of Psychiatry: the Journal of Mental Science, 215(3), 545–551. https://doi.org/10.1192/bjp.2019.10
McDonald, B. C., Conroy, S. K., Ahles, T. A., West, J. D., & Saykin, A. J. (2012). Alterations in brain activation during working memory processing associated with breast cancer and treatment: a prospective functional magnetic resonance imaging study. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology, 10(20), 2500–2508. https://doi.org/10.1200/jco.2011.38.5674
Menning, S., de Ruiter, M. B., Veltman, D. J., et al., (2017). Changes in brain activation in breast cancer patients depend on cognitive domain and treatment type. PLoS One1, 12(3), e0171724. https://doi.org/10.1371/journal.pone.0171724
Menning, S., de Ruiter, M. B., Veltman, D. J., et al., (2018). Changes in brain white matter integrity after systemic treatment for breast cancer: a prospective longitudinal study. Brain Imaging and Behavior, 12(2), 324–334. https://doi.org/10.1007/s11682-017-9695-x
Nasim, F., Sabath, B. F., & Eapen, G. A. (2019). Lung cancer. The Medical clinics of North America, 103(3), 463–473. https://doi.org/10.1016/j.mcna.2018.12.006
Nasreddine, Z. S., Phillips, N. A., Bedirian, V., et al., (2005). The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society, 53(4), 695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
Pergolizzi, D., Root, J. C., Pan, H., et al., (2019). Episodic memory for visual scenes suggests compensatory brain activity in breast cancer patients: a prospective longitudinal fMRI study. Brain Imaging and Behavior. https://doi.org/10.1007/s11682-019-00038-2
Piccirillo, J. F., Hardin, F. M., Nicklaus, J., et al., (2015). Cognitive impairment after chemotherapy related to atypical network architecture for executive control. Oncology, 88(6), 360–368. https://doi.org/10.1159/000370117
Qiu, Y., Guo, Z., Han, L., et al., (2018). Network-level dysconnectivity in patients with nasopharyngeal carcinoma (NPC) early post-radiotherapy: longitudinal resting state fMRI study. Brain Imaging and Behavior, 12(5), 1279–1289. https://doi.org/10.1007/s11682-017-9801-0
Rosazza, C., & Minati, L. (2011). Resting-state brain networks: literature review and clinical applications. Neurological Sciences: Official Journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology, 32(5), 773–785. https://doi.org/10.1007/s10072-011-0636-y
Siegel, R. L., Miller, K. D., Jemal, A. (2020). Cancer statistics. CA: a Cancer Journal for Clinicians, 70(1), 7-30. https://doi.org/10.3322/caac.21590
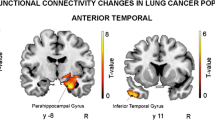
Simo, M., Rifa-Ros, X., Vaquero, L., et al., (2018). Brain functional connectivity in lung cancer population: an exploratory study. Brain Imaging and Behavior, 12(2), 369–382. https://doi.org/10.1007/s11682-017-9697-8
Sousa, H., Almeida, S., Bessa, J., & Pereira, M. G. (2020). The developmental trajectory of cancer-related cognitive impairment in breast cancer patients: a systematic review of longitudinal neuroimaging studies. Neuropsychology Review, 30(3), 287–309. https://doi.org/10.1007/s11065-020-09441-9
Stinear, C. M., Coxon, J. P., & Byblow, W. D. (2009). Primary motor cortex and movement prevention: where Stop meets Go. Neuroscience and Biobehavioral Reviews, 33(5), 662–673. https://doi.org/10.1016/j.neubiorev.2008.08.013
Tao, L., Lin, H., Yan, Y., et al., (2017). Impairment of the executive function in breast cancer patients receiving chemotherapy treatment: a functional MRI study. European Journal of Cancer Care, 26(6). https://doi.org/10.1111/ecc.12553
You, J., Hu, L., Zhang, Y., et al., (2020). Altered dynamic neural activity in the default mode network in lung cancer patients after chemotherapy. Medical Science Monitor: International Medical Journal of Experimental and Clinical Research, 18, 26921700. https://doi.org/10.12659/msm.921700
Zhang, G., Li, Y., & Zhang, J. (2019). Tracking the dynamic functional network interactions during goal-directed auditory tasks by brain state clustering. Frontiers in Neuroscience, 13, 1220. https://doi.org/10.3389/fnins.2019.01220
Zhang, Y., Chen, Y. C., Hu, L., et al., (2020). Chemotherapy-induced functional changes of the default mode network in patients with lung cancer. Brain Imaging and Behavior, 14(3):847–856. https://doi.org/10.1007/s11682-018-0030-y
Funding
This work was supported by the Natural Science Foundation of Jiangsu Higher Education Institutions (No. 18KJB320007), Nanjing Outstanding Youth Fund (No. JQX19006), and 333 High-level Talents Training Project of Jiangsu Province (No. BRA2019122).
Author information
Authors and Affiliations
Contributions
LH. and SD. are co-first authors of this paper, they design the experiment, analyze the data and draft the paper for the work. YZ., JY., SS., and PW. help to acquire the clinical and fMRI data. WX. helps to revise the paper critically for important intellectual content. XY. and YCC. are co-corresponding authors of this paper, they did the financial support, review and final approval of the paper to be published. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
The current study was approved by the Research Ethics Committee of the Nanjing Medical University.
Consent to participate
All participants provided written informed consent before undergoing MR imaging.
Conflict of interest
The authors declare that there is no potential conflict of interests regarding the publication of this paper.
Consent to publish
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 555 KB)
Rights and permissions
About this article
Cite this article
Hu, L., Ding, S., Zhang, Y. et al. Dynamic functional network connectivity reveals the brain functional alterations in lung cancer patients after chemotherapy. Brain Imaging and Behavior 16, 1040–1048 (2022). https://doi.org/10.1007/s11682-021-00575-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-021-00575-9