Abstract
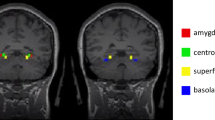
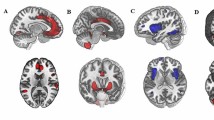
Studies identify the habenula as a key subcortical component in anxiety, with a role in predicting error coding within the evaluative system. However, no clinical reports of generalized anxiety disorder (GAD) describe resting state functional connectivity of habenular circuits. We hypothesized that resting-state functional connectivities of habenula would show differences in neuroanatomical correlates of the evaluative system (prefrontal cortex, habenula) of patients with GAD. We obtained 22 patients with GAD and 21 HCs, matched for gender, age, and years of education. Resting-state functional connectivity of the habenula was assessed using a seed-based template imposed on whole brain MRI, which provided an objective and semi-automated segmentation algorithm in MNI space. Patients with GAD demonstrated enhanced connectivities in the bilateral premotor cortex, right ventrolateral prefrontal cortex, medial frontal cortex, as well as the left orbitofrontal cortex, and reduced connectivities in the left posterior cingulate cortex, and right pulvinar. Moreover, striking differences of abnormal connectivities between groups were observed via analysis of receiver operating characteristic curves (ROC) of statistically significant. These results including ROC curves suggest the potential importance of the habenula in evaluating and deciding to personally relevant reward-related information.







Similar content being viewed by others
Abbreviations
- GAD:
-
generalized anxiety disorder
- HCs:
-
health controls
- VTA:
-
ventral tegmental area
- DSM-5TM:
-
Diagnostic and Statistical Manual of Mental Disorders
- MINI:
-
Mini-International Neuropsychiatric Interview
- HAMA:
-
Hamilton Anxiety Rating Scale
- EPI:
-
echo planar imaging
- FOV:
-
field of view
- TR:
-
repetition time
- TE:
-
echo time
- FA:
-
flip angle
- DICOM:
-
Digital Imaging and Communications in Medicine
- MNI:
-
Montreal Neurological Institute
- FWHM:
-
full width at half maximum
- SPSS21:
-
Statistical Package for the Social Sciences21
- ROC:
-
receiver operating characteristic
- AUC:
-
areas under curves
- PMC:
-
premotor cortex
- vlPFC:
-
ventrolateral prefrontal cortex
- MFC:
-
medial frontal cortex
- OFC:
-
orbitofrontal cortex
- PCC:
-
posterior cingulate cortex
References
Aizawa, H., Amo, R., & Okamoto, H. (2011). Phylogeny and ontogeny of the habenular structure. Frontiers in Neuroscience, 5, 138. https://doi.org/10.3389/fnins.2011.00138.
APA, A. P. A. (2013). Diagnostic and statistical manual of mental disorders (Fifth ed.). American Psychiatric Publishing, p. 189.
Arend, I., Rafal, R., & Ward, R. (2008). Spatial and temporal deficits are regionally dissociable in patients with pulvinar lesions. Brain, 131(8), 2140–2152. https://doi.org/10.1093/brain/awn135.
Ball, T. M., Ramsawh, H. J., Campbell-Sills, L., Paulus, M. P., & Stein, M. B. (2013). Prefrontal dysfunction during emotion regulation in generalized anxiety and panic disorders. Psychological Medicine, 43(7), 1475–1486. https://doi.org/10.1017/s0033291712002383.
Benarroch, E. E. (2015). Habenula: Recently recognized functions and potential clinical relevance. Neurology, 85(11), 992–1000. https://doi.org/10.1212/wnl.0000000000001937.
Calhoon, G. G., & Tye, K. M. (2015). Resolving the neural circuits of anxiety. Nature Neuroscience, 18(10), 1394–1404. https://doi.org/10.1038/nn.4101.
Chan, J., Ni, Y., Zhang, P., Zhang, J., & Chen, Y. (2017). D1-like dopamine receptor dysfunction in the lateral habenula nucleus increased anxiety-like behavior in rat. Neuroscience, 340, 542–550. https://doi.org/10.1016/j.neuroscience.2016.11.005.
Danielmeier, C., Eichele, T., Forstmann, B. U., Tittgemeyer, M., & Ullsperger, M. (2011). Posterior medial frontal cortex activity predicts post-error adaptations in task-related visual and motor areas. The Journal of Neuroscience, 31(5), 1780–1789. https://doi.org/10.1523/jneurosci.4299-10.2011.
Di Bono, M. G., Priftis, K., & Umiltà, C. (2017). Bridging the gap between brain activity and cognition: Beyond the different tales of fMRI data analysis. [Opinion]. Frontiers in Neuroscience, 11(31). https://doi.org/10.3389/fnins.2017.00031.
Diekhof, E. K., & Gruber, O. (2010). When desire collides with reason: Functional interactions between anteroventral prefrontal cortex and nucleus accumbens underlie the human ability to resist impulsive desires. The Journal of Neuroscience, 30(4), 1488–1493. https://doi.org/10.1523/JNEUROSCI.4690-09.2010.
Drabant, E. M., Kuo, J. R., Ramel, W., Blechert, J., Edge, M. D., Cooper, J. R., et al. (2011). Experiential, autonomic, and neural responses during threat anticipation vary as a function of threat intensity and neuroticism. Neuroimage, 55(1), 401–410. https://doi.org/10.1016/j.neuroimage.2010.11.040.
Ely, B. A., Xu, J., Goodman, W. K., Lapidus, K. A., Gabbay, V., & Stern, E. R. (2016). Resting-state functional connectivity of the human habenula in healthy individuals: Associations with subclinical depression. Human Brain Mapping, 37(7), 2369–2384. https://doi.org/10.1002/hbm.23179.
Erpelding, N., Sava, S., Simons, L. E., Lebel, A., Serrano, P., Becerra, L., & Borsook, D. (2014). Habenula functional resting-state connectivity in pediatric CRPS. Journal of Neurophysiology, 111(2), 239–247. https://doi.org/10.1152/jn.00405.2013.
Fan, L., Li, H., Zhuo, J., Zhang, Y., Wang, J., Chen, L., et al. (2016). The human Brainnetome atlas: A new brain atlas based on connectional architecture. Cerebral Cortex, 26(8), 3508–3526. https://doi.org/10.1093/cercor/bhw157.
Fiorillo, C. D. (2013). Two dimensions of value: Dopamine neurons represent reward but not aversiveness. Science, 341(6145), 546–549. https://doi.org/10.1126/science.1238699.
Guo, X., Meng, Z., Huang, G., Fan, J., Zhou, W., Ling, W., et al. (2016). Meta-analysis of the prevalence of anxiety disorders in mainland China from 2000 to 2015. Scientific Reports, 6, 28033. https://doi.org/10.1038/srep28033.
Herkenham, M., & Nauta, W. J. (1977). Afferent connections of the habenular nuclei in the rat. A horseradish peroxidase study, with a note on the fiber-of-passage problem. The Journal of Comparative Neurology, 173(1), 123–146. https://doi.org/10.1002/cne.901730107.
Hetu, S., Luo, Y., Saez, I., D'Ardenne, K., Lohrenz, T., & Montague, P. R. (2016). Asymmetry in functional connectivity of the human habenula revealed by high-resolution cardiac-gated resting state imaging. Human Brain Mapping, 37(7), 2602–2615. https://doi.org/10.1002/hbm.23194.
Hikosaka, O. (2010). The habenula: From stress evasion to value-based decision-making. Nature Reviews. Neuroscience, 11(7), 503–513. https://doi.org/10.1038/nrn2866.
Hoffman, D. L., Dukes, E. M., & Wittchen, H.-U. (2008). Human and economic burden of generalized anxiety disorder. Depression and Anxiety, 25(1), 72–90. https://doi.org/10.1002/da.20257.
Hong, S., Jhou, T. C., Smith, M., Saleem, K. S., & Hikosaka, O. (2011). Negative reward signals from the lateral Habenula to dopamine neurons are mediated by Rostromedial tegmental nucleus in primates. Journal of Neuroscience, 31(32), 11457–11471. https://doi.org/10.1523/jneurosci.1384-11.2011.
Jacinto, L. R., Mata, R., Novais, A., Marques, F., & Sousa, N. (2017). The habenula as a critical node in chronic stress-related anxiety. Experimental Neurology, 289, 46–54.
Kang, S., Li, J., Bekker, A., & Ye, J. H. (2018). Rescue of glutamate transport in the lateral habenula alleviates depression- and anxiety-like behaviors in ethanol-withdrawn rats. Neuropharmacology, 129, 47–56. https://doi.org/10.1016/j.neuropharm.2017.11.013.
Kelley, W. M., Macrae, C. N., Wyland, C. L., Caglar, S., Inati, S., & Heatherton, T. F. (2002). Finding the self? An event-related fMRI study. Journal of Cognitive Neuroscience, 14(5), 785–794. https://doi.org/10.1162/08989290260138672.
Kim, J. W., Naidich, T. P., Joseph, J., Nair, D., Glasser, M. F., O'Halloran, R., ... Xu, J. (2018). Reproducibility of myelin content-based human habenula segmentation at 3 Tesla. doi: https://doi.org/10.1002/hbm.24060.
Kringelbach, M. L. (2005). The human orbitofrontal cortex: Linking reward to hedonic experience. Nature Reviews Neuroscience, 6, 691. https://doi.org/10.1038/nrn1747.
Levy, B. J., & Wagner, A. D. (2011). Cognitive control and right ventrolateral prefrontal cortex: Reflexive reorienting, motor inhibition, and action updating. Annals of the New York Academy of Sciences, 1224(1), 40–62. https://doi.org/10.1111/j.1749-6632.2011.05958.x.
Makovac, E., Meeten, F., Watson, D. R., Herman, A., Garfinkel, S. N., H, D. C., & Ottaviani, C. (2016). Alterations in amygdala-prefrontal functional connectivity account for excessive worry and autonomic dysregulation in generalized anxiety disorder. Biological Psychiatry, 80(10), 786–795. https://doi.org/10.1016/j.biopsych.2015.10.013.
Maner, J. K., Richey, J. A., Cromer, K., Mallott, M., Lejuez, C. W., Joiner, T. E., & Schmidt, N. B. (2007). Dispositional anxiety and risk-avoidant decision-making. Personality and Individual Differences, 42(4), 665–675. https://doi.org/10.1016/j.paid.2006.08.016.
Markovic, V., Agosta, F., Canu, E., Inuggi, A., Petrovic, I., Stankovic, I., et al. (2017). Role of habenula and amygdala dysfunction in Parkinson disease patients with punding. Neurology, 88(23), 2207–2215. https://doi.org/10.1212/wnl.0000000000004012.
Mathuru, A. S., & Jesuthasan, S. (2013). The medial habenula as a regulator of anxiety in adult zebrafish. Front Neural Circuits, 7, 99. https://doi.org/10.3389/fncir.2013.00099.
Matsumoto, M., & Hikosaka, O. (2009). Representation of negative motivational value in the primate lateral habenula. Nature Neuroscience, 12(1), 77–84. https://doi.org/10.1038/nn.2233.
Menon, V. (2011). Large-scale brain networks and psychopathology: A unifying triple network model. Trends in Cognitive Sciences, 15(10), 483–506. https://doi.org/10.1016/j.tics.2011.08.003.
Miu, A. C., Heilman, R. M., & Houser, D. (2008). Anxiety impairs decision-making: Psychophysiological evidence from an Iowa gambling task. Biological Psychology, 77(3), 353–358. https://doi.org/10.1016/j.biopsycho.2007.11.010.
Moon, C. M., Kim, G. W., & Jeong, G. W. (2014). Whole-brain gray matter volume abnormalities in patients with generalized anxiety disorder: Voxel-based morphometry. Neuroreport, 25(3), 184–189. https://doi.org/10.1097/wnr.0000000000000100.
Peterson, D. A., Lotz, D. T., Halgren, E., Sejnowski, T. J., & Poizner, H. (2011). Choice modulates the neural dynamics of prediction error processing during rewarded learning. Neuroimage, 54(2), 1385–1394. https://doi.org/10.1016/j.neuroimage.2010.09.051.
Pobbe, R. L., & Zangrossi, H., Jr. (2008). Involvement of the lateral habenula in the regulation of generalized anxiety- and panic-related defensive responses in rats. Life Sciences, 82(25–26), 1256–1261. https://doi.org/10.1016/j.lfs.2008.04.012.
Proulx, C. D., Hikosaka, O., & Malinow, R. (2014). Reward processing by the lateral habenula in normal and depressive behaviors. Nature Neuroscience, 17(9), 1146–1152. https://doi.org/10.1038/nn.3779.
Qiao, J., Li, A., Cao, C., Wang, Z., Sun, J., & Xu, G. (2017). Aberrant functional network connectivity as a biomarker of generalized anxiety disorder. Frontiers in Human Neuroscience, 11, 626. https://doi.org/10.3389/fnhum.2017.00626.
Raichle, M. E. (2015). The brain's default mode network. Annual Review of Neuroscience, 38, 433–447. https://doi.org/10.1146/annurev-neuro-071013-014030.
Ridderinkhof, K. R., Nieuwenhuis, S., & Braver, T. S. (2007). Medial frontal cortex function: An introduction and overview. Cognitive, Affective, & Behavioral Neuroscience, 7(4), 261–265.
Song, X. W., Dong, Z. Y., Long, X. Y., Li, S. F., Zuo, X. N., Zhu, C. Z., et al. (2011). REST: A toolkit for resting-state functional magnetic resonance imaging data processing. PLoS One, 6(9), e25031. https://doi.org/10.1371/journal.pone.0025031.
Stephenson-Jones, M., Floros, O., Robertson, B., & Grillner, S. (2012). Evolutionary conservation of the habenular nuclei and their circuitry controlling the dopamine and 5-hydroxytryptophan (5-HT) systems. Proceedings of the National Academy of Sciences of the United States of America, 109(3), E164–E173. https://doi.org/10.1073/pnas.1119348109.
Strawn, J. R., Wehry, A. M., Chu, W. J., Adler, C. M., Eliassen, J. C., Cerullo, M. A., et al. (2013). Neuroanatomic abnormalities in adolescents with generalized anxiety disorder: A voxel-based morphometry study. Depression and Anxiety, 30(9), 842–848. https://doi.org/10.1002/da.22089.
Strotmann, B., Kogler, C., Bazin, P. L., Weiss, M., Villringer, A., & Turner, R. (2013). Mapping of the internal structure of human habenula with ex vivo MRI at 7T. Frontiers in Human Neuroscience, 7, 878.
Strotmann, B., Heidemann, R. M., Anwander, A., Weiss, M., Trampel, R., Villringer, A., & Turner, R. (2014). High-resolution MRI and diffusion-weighted imaging of the human habenula at 7 tesla. Journal of Magnetic Resonance Imaging, 39(4), 1018–1026. https://doi.org/10.1002/jmri.24252.
Torrisi, S., Nord, C. L., Balderston, N. L., Roiser, J. P., Grillon, C., & Ernst, M. (2017). Resting state connectivity of the human habenula at ultra-high field. Neuroimage, 147, 872–879. https://doi.org/10.1016/j.neuroimage.2016.10.034.
Ullsperger, M., Danielmeier, C., & Jocham, G. (2014). Neurophysiology of performance monitoring and adaptive behavior. Physiological Reviews, 94(1), 35–79. https://doi.org/10.1152/physrev.00041.2012.
Wang, W., Hou, J., Qian, S., Liu, K., Li, B., Li, M., . . . Sun, G. (2016). Aberrant regional neural fluctuations and functional connectivity in generalized anxiety disorder revealed by resting-state functional magnetic resonance imaging. Neuroscience Letters, 624, 78–84. doi: https://doi.org/10.1016/j.neulet.2016.05.005.
Wikenheiser, A. M., & Schoenbaum, G. (2016). Over the river, through the woods: Cognitive maps in the hippocampus and orbitofrontal cortex. Nature Reviews. Neuroscience, 17(8), 513–523. https://doi.org/10.1038/nrn.2016.56.
Yang, Y., & Raine, A. (2009). Prefrontal structural and functional brain imaging findings in antisocial, violent, and psychopathic individuals: A meta-analysis. Psychiatry Research, 174(2), 81–88. https://doi.org/10.1016/j.pscychresns.2009.03.012.
Acknowledgements
This work was supported by the Nanjing Brain Hospital Affiliated to Nanjing Medical University. Also, the protocol for the research project has been approved by a suitably constituted Ethics Committee of the Nanjing Brain Hospital Affiliated to Nanjing Medical University. All co-authors listed have approved the manuscript that is enclosed and there is no financial interest to report. The views expressed in this paper are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the U.S. government.
Funding
This study was funded by National Natural Science Foundation of China (Grant 81571344, 81201064, 81871344); Natural Science Foundation of Jiangsu Province (Grant BK20161109); the Natural Science Foundation of the Higher Education Institutions of Jiangsu Province, China (Grant 18KJB190003); key research and development program (Social Development) project of Jiangsu province (Grant BE20156092015).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Author Yuan Zhong has received research grants from National Natural Science Foundation of China (Grant 81871344) and Natural Science Foundation of the Higher Education Institutions of Jiangsu Province, China (Grant 18KJB190003). Author Chun Wang has received has received research grants from National Natural Science Foundation of China (Grant 81571344, 81201064) and Natural Science Foundation of Jiangsu Province (Grant BK20161109). Author Ning Zhang has received has received research grants from key research and development program (Social Development). project of Jiangsu province (Grant BE20156092015).
Author Zijuan Ma, Yuan Zhong, Christina S. Hines, Yun Wu, Yuting Li, Manlong Pang, Jian Li, Chiyue Wang, Peter T. Fox, Ning Zhang, Chun Wang declares that he/she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 61 kb)
Rights and permissions
About this article
Cite this article
Ma, Z., Zhong, Y., Hines, C.S. et al. Identifying generalized anxiety disorder using resting state habenular circuitry. Brain Imaging and Behavior 14, 1406–1418 (2020). https://doi.org/10.1007/s11682-019-00055-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-019-00055-1