Abstract
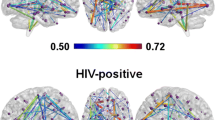
This study examines white matter microstructure using quantitative tractography diffusion magnetic resonance imaging (qtdMRI) in HIV+ individuals from South Africa who were naïve or early in the initiation of antiretroviral therapy. Fiber bundle length (FBL) metrics, generated from qtdMRI, for whole brain and six white matter tracts of interest (TOI) were assessed for 135 HIV+ and 21 HIV− individuals. The association between FBL metrics, measures of disease burden, and neuropsychological performance were also investigated. Results indicate significantly reduced sum of whole brain fiber bundle lengths (FBL, p < 0.001), but not average whole brain FBL in the HIV+ group compared to the HIV− controls. The HIV+ group exhibited significantly shorter sum of FBL in all six TOIs examined: the anterior thalamic radiation, cingulum bundle, inferior and superior longitudinal fasciculi, inferior frontal occipital fasciculus, and the uncinate fasciculus. Additionally, average FBLs were significantly shorter select TOIs including the inferior longitudinal fasciculus, cingulum bundle, and the anterior thalamic radiation. Shorter whole brain FBL sum metrics were associated with poorer neuropsychological performance, but were not associated with markers of disease burden. Taken together these findings suggest HIV affects white matter architecture primarily through reductions in white matter fiber numbers and, to a lesser degree, the shortening of fibers along a bundle path.

Similar content being viewed by others
References
Alexander, A. L., et al. (2011). Characterization of cerebral white matter properties using quantitative magnetic resonance imaging stains. Brain Connectivity, 1(6), 432–446. https://doi.org/10.1089/brain.2011.0071.
Ances, B. M., & Hammoud, D. A. (2014). Neuroimaging of HIV associated neurocognitive disorders (HAND). Current Opinion in HIV and AIDS, 9(6), 545.
Baker, L. M., Cooley, S. A., Cabeen, R. P., Laidlaw, D. H., Joska, J. A., Hoare, J., et al. (2017). Topological organization of whole-brain white matter in HIV infection. Brain Connectivity, 7(2), 115–122. https://doi.org/10.1089/brain.2016.0457.
Baker, L. M., Laidlaw, D. H., Cabeen, R., Akbudak, E., Conturo, T. E., Correia, S., et al. (2016). Cognitive reserve moderates the relationship between neuropsychological performance and white matter fiber bundle length in healthy older adults. Brain Imaging and Behavior. https://doi.org/10.1007/s11682-016-9540-7.
Baker, L. M., Laidlaw, D. H., Conturo, T. E., Hogan, J., Zhao, Y., Luo, X., et al. (2014). White matter changes with age utilizing quantitative diffusion MRI. Neurology, 83(3), 247–252. https://doi.org/10.1212/WNL.0000000000000597.
Beaulieu, C. (2002). The basis of anisotropic water diffusion in the nervous system - a technical review. NMR in Biomedicine, 15(7–8), 435–455. https://doi.org/10.1002/nbm.782.
Behrens, T. E., Berg, H. J., Jbabdi, S., Rushworth, M. F., & Woolrich, M. W. (2007). Probabilistic diffusion tractography with multiple fibre orientations: what can we gain? NeuroImage, 34(1), 144–155. https://doi.org/10.1016/j.neuroimage.2006.09.018.
Behrman-Lay, A. M., Usher, C., Conturo, T. E., Correia, S., Laidlaw, D. H., Lane, E. M., et al. (2015). Fiber bundle length and cognition: a length-based tractography MRI study. Brain Imaging and Behavior, 9(4), 765–775. https://doi.org/10.1007/s11682-014-9334-8.
Biesbroek, J. M., Kuijf, H. J., van der Graaf, Y., Vincken, K. L., Postma, A., Mali, W. P., et al. (2013). Association between subcortical vascular lesion location and cognition: a voxel-based and tract-based lesion-symptom mapping study. The SMART-MR study. PLoS One, 8(4), e60541. https://doi.org/10.1371/journal.pone.0060541.
Bolzenius, J. D., Laidlaw, D. H., Cabeen, R. P., Conturo, T. E., McMichael, A. R., Lane, E. M., et al. (2013). Impact of body mass index on neuronal fiber bundle lengths among healthy older adults. Brain Imaging and Behavior, 7(3), 300–306. https://doi.org/10.1007/s11682-013-9230-7.
Bolzenius, J. D., Laidlaw, D. H., Cabeen, R. P., Conturo, T. E., McMichael, A. R., Lane, E. M., et al. (2015). Brain structure and cognitive correlates of body mass index in healthy older adults. Behavioural Brain Research, 278, 342–347. https://doi.org/10.1016/j.bbr.2014.10.010.
Buch, S., Chivero, E. T., Hoare, J., Jumare, J., Nakasujja, N., Mudenda, V., et al. (2016). Proceedings from the NIMH symposium on “NeuroAIDS in Africa: neurological and neuropsychiatric complications of HIV”. Journal of Neurovirology, 22(5), 699–702.
Cabeen, R. P., Bastin, M. E., & Laidlaw, D. H. (2016). Kernel regression estimation of fiber orientation mixtures in diffusion MRI. NeuroImage, 127, 158–172. https://doi.org/10.1016/j.neuroimage.2015.11.061.
Catani, M., & Thiebaut de Schotten, M. (2012). Atlas of human brain connections. Oxford, Oxford University Press.
Chang, L., Wong, V., Nakama, H., Watters, M., Ramones, D., Miller, E. N., et al. (2008). Greater than age-related changes in brain diffusion of HIV patients after 1 year. Journal of Neuroimmune Pharmacology, 3(4), 265–274. https://doi.org/10.1007/s11481-008-9120-8.
Correa, D. G., Zimmermann, N., Tukamoto, G., Doring, T., Ventura, N., Leite, S. C., et al. (2016). Longitudinal assessment of subcortical gray matter volume, cortical thickness, and white matter integrity in HIV-positive patients. Journal of Magnetic Resonance Imaging. https://doi.org/10.1002/jmri.25263.
Correia, S., Lee, S. Y., Voorn, T., Tate, D. F., Paul, R. H., Zhang, S., et al. (2008). Quantitative tractography metrics of white matter integrity in diffusion-tensor MRI. NeuroImage, 42(2), 568–581. https://doi.org/10.1016/j.neuroimage.2008.05.022.
de Almeida, S. M., Ribeiro, C. E., de Pereira, A. P., Badiee, J., Cherner, M., Smith, D., et al. (2013). Neurocognitive impairment in HIV-1 clade C- versus B-infected individuals in Southern Brazil. Journal of Neurovirology, 19(6), 550–556. https://doi.org/10.1007/s13365-013-0215-5.
Fischl, B. (2012). FreeSurfer. NeuroImage, 62(2), 774–781. https://doi.org/10.1016/j.neuroimage.2012.01.021.
Heaps, J. M., Joska, J., Hoare, J., Ortega, M., Agrawal, A., Seedat, S., et al. (2012). Neuroimaging markers of human immunodeficiency virus infection in South Africa. Journal of Neurovirology, 18(3), 151–156. https://doi.org/10.1007/s13365-012-0090-5.
Heaton, R. K., Clifford, D. B., Franklin, D. R. Jr., Woods, S. P., Ake, C., Vaida, F., et al. (2010). HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER study. Neurology, 75(23), 2087–2096. https://doi.org/10.1212/WNL.0b013e318200d727.
Hoare, J., Fouche, J. P., Phillips, N., Joska, J. A., Donald, K. A., Thomas, K., & Stein, D. J. (2015). Clinical associations of white matter damage in cART-treated HIV-positive children in South Africa. Journal of Neurovirology, 21(2), 120–128. https://doi.org/10.1007/s13365-014-0311-1.
Hoare, J., Fouche, J. P., Spottiswoode, B., Joska, J. A., Schoeman, R., Stein, D. J., & Carey, P. D. (2010). White matter correlates of apathy in HIV-positive subjects: a diffusion tensor imaging study. The Journal of Neuropsychiatry and Clinical Neurosciences, 22(3), 313–320. https://doi.org/10.1176/appi.neuropsych.22.3.313. https://doi.org/10.1176/jnp.2010.22.3.313.
Hoare, J., Fouche, J. P., Spottiswoode, B., Sorsdahl, K., Combrinck, M., Stein, D. J., et al. (2011). White-matter damage in clade C HIV-positive subjects: a diffusion tensor imaging study. The Journal of Neuropsychiatry and Clinical Neurosciences, 23(3), 308–315. https://doi.org/10.1176/appi.neuropsych.23.3.308. https://doi.org/10.1176/jnp.23.3.jnp308.
Hoare, J., Westgarth-Taylor, J., Fouche, J. P., Combrinck, M., Spottiswoode, B., Stein, D. J., & Joska, J. A. (2013). Relationship between apolipoprotein E4 genotype and white matter integrity in HIV-positive young adults in South Africa. European Archives of Psychiatry and Clinical Neuroscience, 263(3), 189–195. https://doi.org/10.1007/s00406-012-0341-8.
Hoare, J., Westgarth-Taylor, J., Fouche, J. P., Spottiswoode, B., Paul, R., Thomas, K., et al. (2012). A diffusion tensor imaging and neuropsychological study of prospective memory impairment in South African HIV positive individuals. Metabolic Brain Disease, 27(3), 289–297. https://doi.org/10.1007/s11011-012-9311-0.
Holm, S. (1979). A simple sequentially rejective multiple test procedure. Scandinavian Journal of Statistics, 6(2), 65–70.
Jenkinson, M., Beckmann, C. F., Behrens, T. E., Woolrich, M. W., & Smith, S. M. (2012). Fsl. Neuroimage, 62(2), 782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015.
Jenkinson, M., & Smith, S. (2001). A global optimisation method for robust affine registration of brain images. Medical Image Analysis, 5(2), 143–156.
Joska, J. A., Fincham, D. S., Stein, D. J., Paul, R. H., & Seedat, S. (2010). Clinical correlates of HIV-associated neurocognitive disorders in South Africa. AIDS and Behavior, 14(2), 371–378. https://doi.org/10.1007/s10461-009-9538-x.
Joska, J. A., Westgarth-Taylor, J., Hoare, J., Thomas, K. G., Paul, R., Myer, L., & Stein, D. J. (2012). Neuropsychological outcomes in adults commencing highly active anti-retroviral treatment in South Africa: a prospective study. BMC Infectious Diseases, 12, 39. https://doi.org/10.1186/1471-2334-12-39.
Joska, J. A., Westgarth-Taylor, J., Myer, L., Hoare, J., Thomas, K. G., Combrinck, M., et al. (2011). Characterization of HIV-associated neurocognitive disorders among individuals starting antiretroviral therapy in South Africa. AIDS and Behavior, 15(6), 1197–1203. https://doi.org/10.1007/s10461-010-9744-6.
Leemans, A., & Jones, D. K. (2009). The B-matrix must be rotated when correcting for subject motion in DTI data. Magnetic Resonance in Medicine, 61(6), 1336–1349. https://doi.org/10.1002/mrm.21890.
Masters, M. C., & Ances, B. M. (2014). Role of neuroimaging in HIV-associated neurocognitive disorders. In Seminars in neurology (Vol. 34, No. 01, pp. 89–102). New York: Thieme Medical Publishers.
Mori, S., & van Zijl, P. C. (2002). Fiber tracking: principles and strategies - a technical review. NMR in Biomedicine, 15(7–8), 468–480. https://doi.org/10.1002/nbm.781.
Nath, A. (2015). Eradication of human immunodeficiency virus from brain reservoirs. Journal of Neurovirology, 21(3), 227–234.
Oishi, K., Faria, A., Jiang, H., Li, X., Akhter, K., Zhang, J., et al. (2009). Atlas-based whole brain white matter analysis using large deformation diffeomorphic metric mapping: application to normal elderly and Alzheimer’s disease participants. NeuroImage, 46(2), 486–499.
Ortega, M., Heaps, J. M., Joska, J., Vaida, F., Seedat, S., Stein, D. J., et al. (2013). HIV clades B and C are associated with reduced brain volumetrics. Journal of Neurovirology, 19(5), 479–487. https://doi.org/10.1007/s13365-013-0202-x.
Paul, R. H., Brickman, A. M., Navia, B., Hinkin, C., Malloy, P. F., Jefferson, A. L., et al. (2005). Apathy is associated with volume of the nucleus accumbens in patients infected with HIV. The Journal of Neuropsychiatry and Clinical Neurosciences, 17(2), 167–171. https://doi.org/10.1176/jnp.17.2.167.
Paul, R. H., Joska, J. A., Woods, C., Seedat, S., Engelbrecht, S., Hoare, J., et al. (2014). Impact of the HIV Tat C30C31S dicysteine substitution on neuropsychological function in patients with clade C disease. Journal of Neurovirology, 20(6), 627–635. https://doi.org/10.1007/s13365-014-0293-z.
Paul, R. H., Phillips, S., Hoare, J., Laidlaw, D. H., Cabeen, R., Olbricht, G. R., et al. (2017). Neuroimaging abnormalities in clade C HIV are independent of Tat genetic diversity. Journal of Neurovirology, 23(2), 319–328. https://doi.org/10.1007/s13365-016-0503-y.
Radloff, L. S. (1991). The use of the center for epidemiologic studies depression scale in adolescents and young adults. Journal of Youth and Adolescence, 20(2), 149–166. https://doi.org/10.1007/BF01537606.
Ragin, A. B., Wu, Y., Gao, Y., Keating, S., Du, H., Sammet, C., et al. (2015). Brain alterations within the first 100 days of HIV infection. Annals of Clinical and Translational Neurology, 2(1), 12–21.
Salminen, L. E., Schofield, P. R., Lane, E. M., Heaps, J. M., Pierce, K. D., Cabeen, R., et al. (2013). Neuronal fiber bundle lengths in healthy adult carriers of the ApoE4 allele: a quantitative tractography DTI study. Brain Imaging and Behavior, 7(3), 274–281. https://doi.org/10.1007/s11682-013-9225-4.
Scaravilli, F., Bazille, C., & Gray, F. (2007). Neuropathologic contributions to understanding AIDS and the central nervous system. Brain Pathology, 17(2), 197–208. https://doi.org/10.1111/j.1750-3639.2007.00047.x.
Sheehan, D. V., Lecrubier, Y., Sheehan, K. H., Amorim, P., Janavs, J., Weiller, E., et al. (1998). The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. The Journal of Clinical Psychiatry, 59(Suppl 20), 22–33; quiz 34–57.
Seider, T. R., Gongvatana, A., Woods, A. J., Chen, H., Porges, E. C., Cummings, T., et al. (2016). Age exacerbates HIV-associated white matter abnormalities. Journal of Neurovirology, 22(2), 201–212.
Smith, S. M. (2002). Fast robust automated brain extraction. Human Brain Mapping, 17(3), 143–155. https://doi.org/10.1002/hbm.10062.
Tate, D., Paul, R. H., Flanigan, T. P., Tashima, K., Nash, J., Adair, C., et al. (2003). The impact of apathy and depression on quality of life in patients infected with HIV. AIDS Patient Care and STDs, 17(3), 115–120. https://doi.org/10.1089/108729103763807936.
Tate, D. F., Conley, J., Paul, R. H., Coop, K., Zhang, S., Zhou, W., et al. (2010). Quantitative diffusion tensor imaging tractography metrics are associated with cognitive performance among HIV-infected patients. Brain Imaging and Behavior, 4(1), 68–79. https://doi.org/10.1007/s11682-009-9086-z.
Thompson, P. M., & Jahanshad, N. (2015). Novel neuroimaging methods to understand how HIV affects the brain. Current HIV/AIDS Reports, 12(2), 289–298.
Wright, P., Heaps, J., Shimony, J. S., Thomas, J. B., & Ances, B. M. (2012). The effects of HIV and combination antiretroviral therapy on white matter integrity. AIDS (London, England), 26(12), 1501.
Yushkevich, P. A., Piven, J., Hazlett, H. C., Smith, R. G., Ho, S., Gee, J. C., & Gerig, G. (2006). User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. NeuroImage, 31(3), 1116–1128. https://doi.org/10.1016/j.neuroimage.2006.01.015.
Zappala, G., Thiebaut de Schotten, M., & Eslinger, P. J. (2012). Traumatic brain injury and the frontal lobes: what can we gain with diffusion tensor imaging? Cortex, 48(2), 156–165. https://doi.org/10.1016/j.cortex.2011.06.020.
Zhang, H., Yushkevich, P. A., Alexander, D. C., & Gee, J. C. (2006). Deformable registration of diffusion tensor MR images with explicit orientation optimization. Medical Image Analysis, 10(5), 764–785. https://doi.org/10.1016/j.media.2006.06.004.
Zhang, H., Avants, B. B., Yushkevich, P. A., Woo, J. H., Wang, S., McCluskey, L. F., et al. (2007). High-dimensional spatial normalization of diffusion tensor images improves the detection of white matter differences: an example study using amyotrophic lateral sclerosis. IEEE Transactions on Medical Imaging, 26(11), 1585–1597. https://doi.org/10.1109/TMI.2007.906784.
Zhang, H., Awate, S. P., Das, S. R., Woo, J. H., Melhem, E. R., Gee, J. C., & Yushkevich, P. A. (2010). A tract-specific framework for white matter morphometry combining macroscopic and microscopic tract features. Medical Image Analysis, 14(5), 666–673.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Funding for this study was provided by the National Institute of Mental Health to Robert H. Paul R01MH085604.
Conflict of interest
Authors have no conflicts of interest to declare.
Ethical approval
All procedures were approved by the institutional review boards for human subjects’ research at the University of Missouri-St. Louis and the University of Cape Town in accordance with the ethical standards national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Heaps-Woodruff, J.M., Joska, J., Cabeen, R. et al. White matter fiber bundle lengths are shorter in cART naive HIV: an analysis of quantitative diffusion tractography in South Africa. Brain Imaging and Behavior 12, 1229–1238 (2018). https://doi.org/10.1007/s11682-017-9769-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-017-9769-9