Abstract
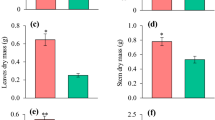
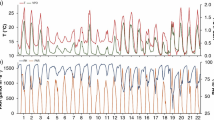
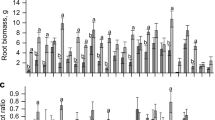
Plantations of eucalypts as short-rotation tree crops are rapidly expanding in tropical and sub-tropical regions, including southern China, where the soils are acidic and available phosphorus (P) is limited. We investigated seedling growth, dry matter accumulation, and the dynamics of photosynthetic rate and chlorophyll content of seven Eucalyptus species/hybrids (E. dunnii, E. grandis, E. grandis × E. camaldulensis, E. urophylla × E. camaldulensis, E. urophylla × E. tereticornis, E. grandis × E. tereticornis, E. urophylla × E. grandis) in response to different levels of P supply (0, 6, 12 and 18 mg·kg−1 KH2PO4). The photosynthetic rate and the chlorophyll content significantly declined as the P supply declined in almost a linear fashion for all species as the P stress period extended. In the absence of P supply, height growth of seedlings of all species was significantly impaired, while root collar diameter growth and whole plant dry matter accumulation was not affected by the level of P supply in most of the species. Significant inter-species variations in growth, dry matter accumulation and photosynthetic rate in response to P supply were detected. Eucalyptus dunnii had the lowest growth performance across all levels of P supply while E. Urophylla × E. tereticornis showed superior growth performance. From a practical point of view, E. Urophylla × E. tereticornis is suggested as a candidate hybrid for planting on slightly P-deficient sites in southern China while E. dunnii, being a slow-growing species, is not suitable for short-rotation plantation. On plantation sites where severe P deficiency exists, P fertilization needs to be considered to boost rapid growth of seedlings so as to meet the management objectives of short-rotation plantation.
Similar content being viewed by others
References
Baldwin JC, Athikkattuvalasu SK, Kashchandra GR. 2001. LEPS2, a phosphorus starvation-induced novel acid phosphatase from tomato. Plant Physiology, 125: 728–737.
Bariola PA, Christie JH, Crispin BT, Michael TV, Vanita DJ, Pamela JG. 1994. The Arabidopsis ribonuclease gene RNS1 is tightly controlled in response to phosphate limitation. The Plant Journal, 6: 673–685.
Chen HJ, Li YQ, Chen DD, Zhang Y, Wu LM, Ji JS. 1996. Soil phosphorus fractions and their availability in Chinese fir plantations in south China, Forest Research, 9: 121–126.
Chen HJ. 2003. Phosphatase activity and P fractions in soils of an 18-year-old Chinese fir (Cunninghamia lanceolata) plantation. Forest Ecology and Management, 178: 301–310.
Davis CS. 2002. Statistical methods for the analysis of repeated measurements. London: Springer, p. 203–298.
Grove TS, Thomson BD, Malajczuk N. 1996. Nutritional physiology of eucalypts: uptake, distribution and utilization. Nutrition of eucalypts. Australia: CSIRO, pp. 77–108.
Guo SL, Yan XF, Bai B, Yu S. 2005. Responses of larch seedling’s photosynthetic characteristics to nitrogen and phosphorus deficiency. Chinese Journal of Applied Ecology, 16: 589–594.
Jacob J, Lawlor DW. 1991. Stomatal and mesophyll limitations of photosynthesis in phosphate deficient sunflower, maize and wheat plants. Journal of Experimental Botany, 42(8): 1003–1011.
Kirschbaum MUF, Tompkins D. 1990. Photosynthetic responses to phosphorus nutrition in Eucalyptus grandis seedlings. Functional Plant Biology, 17: 527–535.
Laclau JP, Bouillet JP, Ranger J. 2000. Dynamics of biomass and nutrient accumulation in a clonal plantation of Eucalyptus in Congo. Forest Ecology and Management, 128: 181–196.
Lewis JD, Griffin KL, Thomas RB, Strain BR. 1994. Phosphorus supply affects the photosynthetic capacity of loblolly pine grown in elevated carbon dioxide. Tree physiology, 14: 1229–1244.
Li LH, Qiu XH, Li XH, Wang SP, Lian XM. 2009. The expression profile of genes in rice roots under low phosphorus stress. Science in China Series C: Life Sciences, 52: 1055–1064.
Liu JZ, Li ZS, Li JY. 1994. Utilization of plant potentialities to enhance the bio-efficiency of phosphorus in soil. Chinese Journal of Eco-Agriculture, 1: 18–25.
Mulligan DR, Sands R. 1988. Dry matter, phosphorus and nitrogen partitioning in three Eucalyptus species grown under a nutrient deficit. New phytologist, 109: 21–28.
Raghothama KG. 1999. Phosphate acquisition. Annual review of plant biology, 50: 665–693.
Scholz RW, Ulrich AE, Eilittä M, Roy A. 2013. Sustainable use of phosphorus: A finite resource. Science of the Total Environment, 461–462: 799–803.
Shenoy VV, Kalagudi GM. 2005. Enhancing plant phosphorus use efficiency for sustainable cropping. Biotechnology advances, 23: 501–513.
Shepherd M, Bartle J, Lee DJ, Brawner J, Bush D, Turnbull P, Macdonel P, Brown TR, Simmons B, Henry R. 2011. Eucalypts as a biofuel feedstock. Biofuels, 2: 639–657.
Singh KD. 2013. Global forest resources assessments. In: Capacity building for the planning, assessment and systematic observations of forests. Environmental Science and Engineering. Springer. p. 203–211.
Thomas DS, Montagu KD, Conroy JP. 2006a. Leaf inorganic phosphorus as a potential indicator of phosphorus status, photosynthesis and growth of Eucalyptus grandis seedlings, Forest Ecology and Management, 223: 267–274.
Thomas DS, Montagu KD, Conroy JP. 2006b. Why does phosphorus limitation increase wood density in Eucalyptus grandis seedlings? Tree physiology, 26: 35–42.
Turnbull TL, Warren CR, Adams MA. 2007. Novel mannose-sequestration technique reveals variation in subcellular orthophosphate pools do not explain the effects of phosphorus nutrition on photosynthesis in Eucalyptus globulus seedlings. New phytologist, 176: 849–861.
Wang CN, Cao FX, Huang YF, Li J. 2004. Poly-usage of eucalyptus and its sub-metabolized production. Non-wood Forest Research, 22(3): 57–59.
Warren CR. 2011. How does P affect photosynthesis and metabolite profiles of Eucalyptus globulus? Tree physiology, 31: 727–739.
Wu C, Sun HL, Wang ZQ. 2005. Change of potential photosynthesis rates in Fraxinus mandshurica seedlings supplied with different phosphorus levels, Journal of Anhui Agricultural University, 33: 2028–2031.
Wu PF, Ma XQ, Tigabu M, Wang C, Liu AQ, Oden PC. 2011a. Root morphological plasticity and biomass production of two Chinese fir clones with high phosphorus efficiency under low phosphorus stress. Canadian Journal of Forest Research, 41: 228–234.
Wu PF, Tigabu M, Ma XQ, Oden PC, He YL, Yu XT, He ZY. 2011b. Variations in biomass, nutrient contents and nutrient use efficiency among Chinese fir provenances. Silvae Genetica, 60: 95–105.
Xu D, Dell B, Malajczuk N, Gong M. 2001. Effects of P fertilization and ectomycorrhizal fungal inoculation on early growth of eucalypt plantations in southern China. Plant and soil, 233: 47–57.
Xu D. 1997. Effects of different phosphate levels on growth and nutrient uptake in different provenances of Eucalyptus urophylla S. T. Blake, Tropical and Subtropical Soil Science, 6(2): 76–81.
Yu YC, Yu J, Shan QH, Fang L, Jiang DF. 2008. Organic acid exudation from the roots of Cunninghamia lanceolata and Pinus massoniana seedlings under low phosphorus stress. Frontiers of Forestry in China, 3: 117–120.
Zar Jh. 1996. Biostatistical analysis (3rd edition). London: Prentice-Hall, p. 123–178.
Zhang XZ. 1986. Determination of chlorophyll content in plant tissue. Liaoning Agricultural Science and Technology, 3: 19–21.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding: This research is supported by Forestry Bureau of China (Grant No. 2011-4-59, No. 201304303), National Natural Science Foundation of China (Grant No. 31100472) and the Training Program Foundation for University Distinguished Young Talents of Fujian Province, China (Grant No. JA12091).
Corresponding editor: Zhu Hong
Rights and permissions
About this article
Cite this article
Wu, Pf., Ma, Xq., Tigabu, M. et al. Comparative growth, dry matter accumulation and photosynthetic rate of seven species of Eucalypt in response to phosphorus supply. Journal of Forestry Research 25, 377–383 (2014). https://doi.org/10.1007/s11676-014-0465-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-014-0465-y