Abstract
Objective
Transcriptional coactivator p300 has been shown to play a variety of roles in the transcription process and mutation of p300 has been found in certain types of human cancers. However, the expression dynamics of p300 in breast cancer (BC) and its effect on BC patients’ prognosis are poorly understood.
Methods
In the present study, the methods of tissue microarray and immunohistochemistry (IHC) were used to investigate the protein expression of p300 in BCs. Receiver operating characteristic (ROC) curve analysis, Spearman’s rank correlation, Kaplan-Meier plots and Cox proportional hazards regression model were utilized to analyze the data.
Results
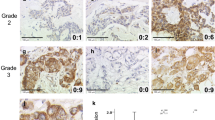
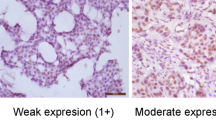
Based on the ROC curve analysis, the cutoff value for p300 high expression was defined when the H score for p300 was more than 105. High expression of p300 could be observed in 105/193 (54.4%) of BCs, in 6/25 (24.0%) of non-malignant breast tissues, respectively (P=0.004). Further correlation analysis showed that high expression of p300 was positively correlated with higher histological grade, advanced clinical stage and tumor recurrence (P<0.05). In univariate survival analysis, a significant association between high expression of p300 and shortened patients’ survival and poor progression-free survival was found (P<0.05). Importantly, p300 expression was evaluated as an independent prognostic factor in multivariate analysis (P<0.05).
Conclusion
Our findings provide a basis for the concept that high expression of p300 in BC may be important in the acquisition of a recurrence phenotype, suggesting that p300 high expression, as examined by IHC, is an independent biomarker for poor prognosis of patients with BC.
Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, et al. Global cancer statistics, 2002. CA Cancer J Clin 2005; 55:74–108.
Liu Y, Ji R, Li J, et al. Correlation effect of EGFR and CXCR4 and CCR7 chemokine receptors in predicting breast cancer metastasis and prognosis. J Exp Clin Cancer Res 2010; 29:16.
Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med 2005; 353:1784–1792.
Goldhirsch A, Wood WC, Gelber RD, et al. Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. Ann Oncol 2007; 18:1133–1144.
Coleman MP, Quaresma M, Berrino F, et al: Cancer survival in five continents: a worldwide population-based study (CONCORD). Lancet Oncol 2008; 9:730–756.
Kundu TK, Palhan VB, Wang Z, et al. Activator-dependent transcription from chromatin in vitro involving targeted histone acetylation by p300. Mol Cell 2000; 6:551–561.
Vo N, Goodman RH. CREB-binding protein and p300 in transcriptional regulation. J Biol Chem 2001; 276:13505–13508.
Goodman RH, Smolik S. CBP/p300 in cell growth, transformation, and development. Genes Dev 2000; 14:1553–1577.
Muraoka M, Konishi M, Kikuchi-Yanoshita R, et al. p300 gene alterations in colorectal and gastric carcinomas. Oncogene 1996; 12:1565–1569.
Gayther SA, Batley SJ, Linger L, et al: Mutations truncating the EP300 acetylase in human cancers. Nat Genet 2000; 24:300–303.
Fan S, Ma YX, Wang C, et al: p300 Modulates the BRCA1 inhibition of estrogen receptor activity. Cancer Res 2002; 62:141–151.
Bandyopadhyay D, Okan NA, Bales E, et al. Down-regulation of p300/CBP histone acetyltransferase activates a senescence checkpoint in human melanocytes. Cancer Res 2002; 62:6231–6239.
Li M, Luo RZ, Chen JW, et al. High expression of transcriptional coactivator p300 correlates with aggressive features and poor prognosis of hepatocellular carcinoma. J Transl Med 2011; 9:5.
Stossi F, Madak-Erdogan Z, Katzenellenbogen BS. Estrogen receptor alpha represses transcription of early target genes via p300 and CtBP1. Mol Cell Biol 2009; 29:1749–1759.
Fermento ME, Gandini NA, Lang CA, et al. Intracellular distribution of p300 and its differential recruitment to aggresomes in breast cancer. Exp Mol Pathol 2010; 88:256–264.
Vleugel MM, Shvarts D, van der Wall E, et al. p300 and p53 levels determine activation of HIF-1 downstream targets in invasive breast cancer. Hum Pathol 2006; 37:1085–1092.
Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 1991; 19:403–410.
Wittekind C. 2010 TNM system: on the 7th edition of TNM classification of malignant tumors. Pathologe 2010; 31:331–332.
Cai MY, Zhang B, He WP, et al. Decreased expression of PinX1 protein is correlated with tumor development and is a new independent poor prognostic factor in ovarian carcinoma. Cancer Sci 2010; 101:1543–1549.
Abubaker J, Bavi P, Al-Haqawi W, et al. PIK3CA alterations in Middle Eastern ovarian cancers. Mol Cancer 2009; 8:51.
Zlobec I, Steele R, Terracciano L, et al. Selecting immunohistochemical cut-off scores for novel biomarkers of progression and survival in colorectal cancer. J Clin Pathol 2007; 60:1112–1116.
Ishihama K, Yamakawa M, Semba S, et al. Expression of HDAC1 and CBP/p300 in human colorectal carcinomas. J Clin Pathol 2007; 60:1205–1210.
Debes JD, Sebo TJ, Lohse CM, et al. p300 in prostate cancer proliferation and progression. Cancer Res 2003; 63:7638–7640.
Karamouzis MV, Konstantinopoulos PA, Papavassiliou AG. Roles of CREB-binding protein (CBP)/p300 in respiratory epithelium tumorigenesis. Cell Res 2007; 17:324–332.
Borrow J, Stanton VP, Jr., Andresen JM, et al: The translocation t(8;16)(p11;p13) of acute myeloid leukaemia fuses a putative acetyltransferase to the CREB-binding protein. Nat Genet 1996; 14:33–41.
Kurebayashi J, Otsuki T, Kunisue H, et al. Expression levels of estrogen receptor-alpha, estrogen receptor-beta, coactivators, and corepressors in breast cancer. Clin Cancer Res 2000; 6:512–518.
De-Carvalho MC, Chimelli LM, Quirico-Santos T. Modulation of fibronectin expression in the central nervous system of Lewis rats with experimental autoimmune encephalomyelitis. Braz J Med Biol Res 1999; 32:583–592.
Hudelist G, Czerwenka K, Kubista E, et al. Expression of sex steroid receptors and their co-factors in normal and malignant breast tissue: AIB1 is a carcinoma-specific co-activator. Breast Cancer Res Treat 2003; 78:193–204.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a grant from the National Natural Science Foundation of China (No.30901709), the National “973” Basic Research Program of China (No. 2010CB529400 and 2010CB912802).
Contributed equally to this study.
Rights and permissions
About this article
Cite this article
Xiao, Xs., Cai, My., Chen, Jw. et al. High expression of p300 in human breast cancer correlates with tumor recurrence and predicts adverse prognosis. Chin. J. Cancer Res. 23, 201–207 (2011). https://doi.org/10.1007/s11670-011-0201-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11670-011-0201-5