Abstract
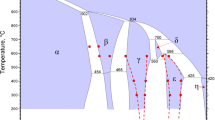
Assessment of literature data, experimental study and thermodynamic modeling of the Pb-As and Cu-Pb-As systems are presented. These chemical systems are of importance for metallic lead refining at 330-500 °C (603-773 K), and for separation of copper from lead in complex polymetallic processes at 1000-1150 °C (1273-1423 K). Few studies are available for the solubility of solid copper arsenide in lead at low temperature. At high temperature, significant discrepancies between available experimentally measured limits of two-liquid miscibility gap exist in the Cu-Pb-As system. Experimental investigation of the present study aimed to fill the gaps and resolve the discrepancies. It consists of equilibration, quenching and electron probe microanalysis. Thermodynamic modeling helped to analyze the results and provided a database of model parameters. Present study is a part of a larger research program aimed at characterization of phase equilibria, heat balance and distribution of elements during complex copper and lead smelting, refining and recycling.













Similar content being viewed by others
References
C.W. Bale, E. Belisle, P. Chartrand, S.A. Decterov, G. Eriksson, A.E. Gheribi, K. Hack, I.H. Jung, Y.B. Kang, J. Melancon, A.D. Pelton, S. Petersen, C. Robelin, J. Sangster, P. Spencer, and M.A. Van Ende, FactSage Thermochemical Software and Databases, 2010–2016, CALPHAD, 2016, 54, p 35-53 (in English)
D. Shishin and E. Jak, Critical Assessment and Thermodynamic Modeling of the Cu-As System, CALPHAD, 2018, 60, p 134-143 (in English)
P.R. Subramanian and D.E. Laughlin, The As-Cu (Arsenic-Copper) System, Bull. Alloy Phase Diagr., 1988, 9(5), p 605-618 (in English)
O. Teppo and P. Taskinen, Assessment of the Thermodynamic Properties of Arsenic-Copper Alloys, Scand. J. Metall., 1991, 20(2), p 141-148 (in English)
R.J. Sinclair, The Extractive Metallurgy of Lead, AusIMM, Carlton, 2009
T.R.A. Davey, Phase Systems Concerned with the Copper Drossing of Lead, Bull. Inst. Min. Metall., 1963, 678, p 553-620
B. Friedrich, F. Toubartz, A. Arnold, Cu-Pb-Me-S-Balances During Lead Refining. in Proceedings of EMC, European Metallurgical Conference, (Germany), (2001), pp. 295-317
Y.P. Novgorodtsev and M.P. Smirnov, Reciprocal Solubility of Components in the Vicinity of the Lead Corner of the Lead-Copper-Arsenic-Sulfur System, Sbornik Nauchnykh Trudov—Gosudarstvennyi Nauchno-Issledovatel’skii Institut Tsvetnykh Metallov, 1967, 26, p 59-75 (in Russian)
H. Hartmann, F. Ensslin, and E. Wunderlich, Decoppering of Lead in the Presence of Tin, Arsenic, and Antimony, Z. Erzbergbau Metallhuettenwes., 1959, 12, p 437-443
A.G. Matyas, Metallurgy of Lead-Copper Dross Smelting, Trans. Inst. Min. Metall. Sect. C, 1977, 86, p 190-194
T. Azakami, M. Hino, and A. Yazawa, The Liquid Miscibility Gap and the Distribution of Silver Between Speiss and Metallic Lead in the Lead-Iron-Arsenic, Lead-Copper-Arsenic and Lead-Iron-Copper-Arsenic Systems at 1200 C, Can. Metall. Q., 1979, 18(4), p 389-394
H. Kleinheisterkamp, Fundamental Equilibria in the Formation of Metallurgical Speisses, Z. Erzbergbau Metallhuettenwes., 1948, 1, p 65-72
J.J. Jacobs, R.H. Maes, and S.R.E. De, The Liquid Miscibility Gaps in the Lead-Copper-Iron-Arsenic System, Trans. Metall. Soc. AIME, 1967, 239(8), p 1166-1171
N. Ritchie, NIST DTSA-II software. https://www.nist.gov/services-resources/software/nist-dtsa-ii. Accessed Aug 2019
D. Shishin, T. Hidayat, J. Chen, P.C. Hayes, and E. Jak, Experimental Investigation and Thermodynamic Modelling of the Distributions of Ag and Au Between Slag, Matte and Metal in the Cu-Fe-O-S-Si System, J. Sustain. Metall., 2018, 5, p 240-249
A.D. Pelton and P. Chartrand, The Modified Quasichemical Model. II—Multicomponent Solutions, Metall. Mater. Trans. A, 2001, 32(6), p 1355-1360
O. Redlich and A.T. Kister, Thermodynamics of Nonelectrolytic Solutions Algebraic Representation of Thermodynamic Properties and the Classification of Solutions, Ind. Eng. Chem., 1948, 40(2), p 345-348
N.A. Gokcen, The As-Pb (Arsenic-Lead) System, Bull. Alloy Phase Diagr., 1990, 11(2), p 120-124
Y. Dessureault, Thermodynamic Modeling of Lead Smelting in the Blast Furnace, Université de Montréal, Montreal, 1994
W. Heike, The Equilibrium Diagram of Lead-Arsenic Alloys and the Melting Point of Arsenic, Intern. Z. Metallog., 1914, 6, p 49-57
O. Bauer and W. Tonn, Alloys of Lead with Arsenic, Z. Metallkd., 1935, 27, p 183-187
S.E. Hutchison and E.A. Peretti, Lead-Rich Portion of the Lead-Arsenic Phase Diagram, J. Less-Common Metals, 1973, 30(2), p 306-308
D.M. Suleimanov, A.N. Mamedov, and A.A. Kuliev, Thermodynamic Properties of Lead-Arsenic Molten Alloys Studied by the Emf. Measurement Method, Izv. Vyssh. Ucheb. Zaved. Khim. Khim. Tekhnol., 1974, 17(3), p 365-367
B. Onderka and J. Wypartowicz, Thermodynamic Activities in the Liquid and the Liquidus Line in the Lead-Arsenic System, Z. Metallkd., 1990, 81(5), p 345-348
E. Zaleska, Electrochemical Investigation of Liquid Lead-Arsenic Solutions, Rocz. Chem., 1974, 48(2), p 195-200
B. Predel and A. Emam, Thermodynamic Properties of Liquid Arsenic-Bismuth and Arsenic-Lead Alloys, Z. Metallk., 1973, 64(9), p 647-652
R.J. McClincy and A.H. Larson, Activity of Arsenic in Dilute Arsenic-Lead Alloys at 703.deg, Trans. Met. Soc. AIME, 1969, 245, p 173
K. Itagaki, T. Shimizu, and M. Hino, Thermodynamic Studies of the Liquid Lead-Arsenic System, Tohoku Daigaku Senko Seiren Kenkyusho Iho, 1978, 34(1), p 45-52
M. Chase, NIST-JANAF Thermochemical Tables, 4th ed., NIST, Gaithersburg, 1998
T. Jantzen and P.J. Spencer, Thermodynamic Assessments of the Cu-Pb-Zn and Cu-Sn-Zn Systems, CALPHAD Comput. Coupling Phase Diagr. Thermochem., 1998, 22(3), p 417-434
A.D. Pelton, S.A. Decterov, G. Eriksson, C. Robelin, and Y. Dessureault, The Modified Quasichemical Model. I—Binary Solutions, Metall. Mater. Trans. B, 2000, 31(4), p 651-659
A.D. Pelton, A General “Geometric” Thermodynamic Model for Multicomponent Solutions, CALPHAD, 2001, 25(2), p 319-328
T. Azakami and M. Hino, Thermodynamic Studies on Behavior of Arsenic in Copper Smelting, Metall. Rev. MMIJ, 1986, 3(3), p 72-86
Acknowledgments
The authors would like to acknowledge the financial and technical support for this research from the consortium of lead producers: Aurubis, Kazzinc Glencore, Umicore, Nystar, Peñoles and Boliden through Australian Research Council Linkage program LP180100028. The authors acknowledge the facilities, the scientific and technical assistance of the Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy and Microanalysis, The University of Queensland. The authors value greatly the scientific discussions with Prof. P.C. Hayes.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shishin, D., Chen, J., Hidayat, T. et al. Thermodynamic Modeling of the Pb-As and Cu-Pb-As Systems Supported by Experimental Study. J. Phase Equilib. Diffus. 40, 758–767 (2019). https://doi.org/10.1007/s11669-019-00764-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-019-00764-6