Abstract
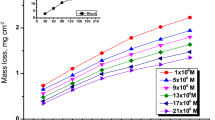
In the present study, corrosion inhibition influence of novel cationic surfactant (CS) with imidazole structure (1-methyl-3-octadecane imidazolium hydrogen sulfate) on low carbon steel in 1 M HCl was investigated by implementing weight loss, potentiodynamic polarization, and electrochemical impedance spectroscopy (EIS) techniques. Increasing the amount of surfactant adequately leads to an increment of the inhibition efficiency of novel CS. According to the obtained results from EIS measurements, inhibition efficiency was about 34% in the presence of 1 ppm surfactant, increasing to about 96.8% at the 25 ppm (near critical micelle concentration) surfactant concentration. Also the effects of temperature and the synergistic effect between surfactant and NaHSO4 salt were studied. The inhibition efficiency increased with the increase of NaHSO4 concentration and reached the maximum value near 0.1 M and experienced a plummet in the temperature range of 30-50 °C. Potentiodynamic polarization measurements revealed that the surfactant acts as mixed-type inhibitors. Results obtained from weight loss, polarization, and impedance measurements are in proper agreement and confirmed the fact that this surfactant is an excellent inhibitor for low carbon steel in 1 M HCl environment. The surface morphology of inhibited and uninhibited metal samples was investigated by atomic force microscope (AFM) and field emission scanning electron microscope (FE-SEM).














Similar content being viewed by others
References
F. Zucchi, G. Trabanelli, and G. Brunoro, The Influence of the Chromium Content on the Inhibitive Efficiency of Some Organic Compounds, Corros. Sci., 1992, 33(7), p 1135–1139
M. Elachouri, M.S. Hajji, S. Kertit, E.M. Essassi, M. Salem, and R. Coudert, Some Surfactants in the Series of 2-(Alkyldimethylammonio) Alkanol Bromides as Inhibitors of the Corrosion of Iron in Acid Chloride Solution, Corros. Sci., 1995, 37(3), p 381–389
I. Jevremovic, M. Singer, S. Nesic, and V. Miskovic, Stankovic, Inhibition Properties of Self-assembled Corrosion Inhibitor Talloil Diethylenetriamine Imidazoline for Mild Steel Corrosion in Chloride Solution Saturated with Carbon Dioxide, Corros. Sci., 2013, 77, p 265–272
X.H. Li, S.D. Deng, H. Fu, G.N. Mu, and N. Zhao, Synergism Between Rare Earth Cerium(IV) Ion and Vanillin on the Corrosion of Steel in H2SO4 Solution: Weight Loss, Electrochemical, UV-VIS, FTIR, XPS, and AFM Approaches, Appl. Surf. Sci., 2008, 254(17), p 5574–5586
X.H. Li, S.D. Deng, G.N. Mu, H. Fu, and F.Z. Yang, Inhibition Effect of Nonionic Surfactant on the Corrosion of Cold Rolled Steel in Hydrochloric Acid, Corros. Sci., 2008, 50(2), p 420–430
S. Bilgic and N. Caliskan, The Effect of N-(1-Toluidine) Salicylaldimine on the Corrosion of Austenitic Chromium–Nickel Steel, Appl. Surf. Sci., 1999, 152(1–2), p 107–114
M.A. Malik, M.A. Hashim, F. Nabi, S.A. Al-Thabaiti, and K. Khan, Review Anti-corrosion Ability of Surfactants: A Review, Int. J. Chem. Sci., 2011, 6(6), p 1927–1948
L.G. Qiu, A.J. Xie, and Y.H. Shen, A Novel Triazole-Based Cationic Gemini Surfactant: Synthesis and Effect on Corrosion Inhibition of Carbon Steel in Hydrochloric Acid, Mater. Chem. Phys., 2005, 91(2–3), p 269–273
M. Mobin and S. Masroor, Adsorption and Corrosion Inhibition Behavior of Schiff Base-Based Cationic Gemini Surfactant on Mild Steel in Formic Acid, J. Dispersion Sci. Technol., 2014, 35(4), p 535–543
A.A. El Maghraby and T.Y. Soror, Efficient Cationic Surfactant as Corrosion Inhibitor for Carbon Steel in Hydrochloric Acid Solutions, Adv. Appl. Sci. Res., 2010, 1(2), p 156–168
M. Motamedi, A.R. Tehrani-Bagha, and M. Mahdavian, Effect of Aging Time on Corrosion Inhibition of Cationic Surfactant on Mild Steel in Sulfamic Acid Cleaning Solution, Corros. Sci., 2013, 70, p 46–54
V.M. Abbasov, H.M. Abd El-Lateef, L.I. Aliyeva, E.E. Qasimov, I.T. Ismayilov, A.H. Tantawy, and S.A. Mamedxanova, Applicability of Novel Anionic Surfactant as a Corrosion Inhibitor of Mild Steel and for Removing Thin Petroleum Films from Water Surface, Am. J. Mater. Sci. Eng., 2013, 1(2), p 18–23
J.R. Kanicky, J.C. Lopez-Montilla, S. Pandey, and D.O. Shah, Surface Chemistry in the Petroleum Industry, Handbook of Applied Surface and Colloid Chemistry, K. Holmberg, Ed., Wiley, Gainesville, FL, 2001, p 251–267
M.J. Rosen, Adsorption of Surface-Active Agents at Interfaces: The Electrical Double Layer, Surfactants and Interfacial Phenomena, M.J. Rosen, Ed., Wiley, Hoboken, NJ, 2004, p 34–73
E. Kowsari and I. Yavari, Green Chemistry-A New Paradigm of Organic Synthesis Using ILs, LAP Academic Publishing, New York, 2011
L.G. Qiu, A.J. Xie, and Y.H. Shen, Understanding the Adsorption of Cationic Gemini Surfactants on Steel Surface in Hydrochloric Acid, Mater. Chem. Phys., 2004, 87(2–3), p 237–240
D. Asefi, M. Arami, and N.M. Mahmoodi, Electrochemical Effect of Cationic Gemini Surfactant and Halide Salts on Corrosion Inhibition of Low Carbon Steel in Acid Medium, Corros. Sci., 2010, 52(3), p 794–800
D. Asefi, M. Arami, and N.M. Mahmoodi, Effect of Nonionic Co-surfactants on Corrosion Inhibition Effect of Cationic Gemini Surfactant, Colloids Surf. A, 2010, 355(1–3), p 183–186
M.A. Hegazy, A Novel Schiff Base-Based Cationic Gemini Surfactants: Synthesis and Effect on Corrosion Inhibition of Carbon Steel in Hydrochloric Acid Solution, Corros. Sci., 2009, 51(11), p 2610–2618
X. Li, S. Deng, H. Fu, and G. Mu, Inhibition Effect of 6-Benzylaminopurine on the Corrosion of Cold Rolled Steel in H2SO4 Solution, Corros. Sci., 2009, 51(3), p 620–634
H. Otmacic and E. Stupnisek-Lisac, Copper Corrosion Inhibitors in Near Neutral Media, Electrochim. Acta, 2003, 48(8), p 985–991
M.A. Migahed, Electrochemical Investigation of the Corrosion Behaviour of Mild Steel in 2 M HCl Solution in Presence of 1-Dodecyl-4-Methoxy Pyridinium Bromide, Mater. Chem. Phys., 2005, 93(1), p 48–53
R. Fuchs-Godec, Effects of Surfactants and Their Mixtures on Inhibition of the Corrosion Process of Ferritic Stainless Steel, Electrochim. Acta, 2009, 54(8), p 2171–2179
L.E. Zerpa, J.L. Salager, C.A. Koh, E. Dendy, and A.K. Sum, Surface Chemistry and Gas Hydrates in Flow Assurance, Ind. Eng. Chem. Res., 2011, 50(1), p 188–197
M.E. Achouri, N.M. Infante, F. Izquierdo, S. Kertit, H.M. Gouttaya, and B. Nciri, Synthesis of Some Cationic Gemini Surfactants and Their Inhibitive Effect on Iron Corrosion in Hydrochloric Acid Medium, Corros. Sci., 2001, 43(1), p 19–35
D. Asefi, M. Arami, A.A. Sarabi, and N.M. Mahmoodi, The Chain Length Influence of Cationic Surfactant and Role of Nonionic Co-surfactants on Controlling the Corrosion Rate of Steel in Acidic Media, Corros. Sci., 2009, 51(8), p 1817–1821
F.M. Menger and J.S. Keiper, Gemini Surfactants, Angew. Chem. Int. Ed., 2000, 39, p 1906–1920
H. Ma, S. Chen, B. Yin, S. Zhao, and X. Liu, Impedance Spectroscopic Study of Corrosion Inhibition of Copper by Surfactants in the Acidic Solutions, Corros. Sci., 2003, 45(5), p 867–882
M.A. Hegazy and M.F. Zaky, Inhibition Effect of Novel Nonionic Surfactants on the Corrosion of Carbon Steel in Acidic Medium, Corros. Sci., 2010, 52(4), p 1333–1341
V. Branzoi, F. Branzoi, and M. Baibarac, The Inhibition of the Corrosion of Armco Iron in HCl Solutions in the Presence of Surfactants of the Type of N-Alkyl Quaternary Ammonium Salts, Mater. Chem. Phys., 2000, 65(3), p 288–297
F.A. Ansari and M.A. Quraishi, Inhibitive Performance of Gemini Surfactants as Corrosion Inhibitors for Mild Steel in Formic Acid, Portugaliae Electrochim. Acta, 2010, 28(5), p 321–335
H. Eivaz Mohammadloo, A.A. Sarabi, A.A. Sabbagh Alvani, H. Sameie, and R. Salimi, Nano-ceramic Hexafluorozirconic Acid Based Conversion Thin Film: Surface Characterization and Electrochemical Study, Surf. Coat. Technol., 2012, 206(19–20), p 4132–4139
M.A. Deyab, Effect of Cationic Surfactant and Inorganic Anions on the Electrochemical Behavior of Carbon Steel in Formation Water, Corros. Sci., 2007, 49(5), p 2315–2328
S.Y. Arman, R. Naderi, and B. PouryosefiMarkhalia, Effect of DC Trend Removal and Window Functioning Methods on Correlation Between Electrochemical Noise Parameters and EIS Data of Stainless Steel in an Inhibited Acidic Solution, RSC Adv., 2014, 4(73), p 39045–39057
X. Wang, H. Yang, and F. Wnag, A Cationic Gemini-Surfactant as Effective Inhibitor for Mild Steel in HCl Solutions, Corros. Sci., 2010, 52(4), p 1268–1276
X. Li, L. Tang, H. Liu, G. Mu, and G. Liu, Influence of Halide Ions on Inhibitive Performance of Cetyl Trimethyl Ammonium Bromide in Various Concentrations of Phosphoric Acid for Cold Rolled Steel, Mater. Lett., 2008, 62(15), p 2321–2324
I.B. Obot, N.O. Obi-Egbedi, and S.A. Umoren, The Synergistic Inhibitive Effect and Some Quantum Chemical Parameters of 2,3-Diaminonaphthalene and Iodide Ions on the Hydrochloric Acid Corrosion of Aluminium, Corros. Sci., 2009, 51(2), p 276–282
A. Fiala, A. Chibani, A. Darchen, A. Boulkamh, and K. Djebbar, Investigations of the Inhibition of Copper Corrosion in Nitric Acid Solutions by Ketene Dithioacetal Derivatives, Appl. Surf. Sci., 2007, 253(24), p 9347–9356
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tabatabaei, F.S., Sarabi, A.A., Kowsari, E. et al. Electrochemical and Morphological Study of Steel in 1 M HCl in the Presence of Task Specific Liquid. J. of Materi Eng and Perform 24, 3433–3443 (2015). https://doi.org/10.1007/s11665-015-1630-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-015-1630-y